Uncategorized
Every bit of help is welcome when controlling exotic species—at least we have a few exotics eating others. Domestic cats are obvious predatory exotics here in BC, whether they’re fattened pets out prowling for even more food or feral and eating just to survive.
Domestic chickens (Gallus gallus) also eat whatever they can catch, so any stray lizard wandering into a chicken run had better run really fast. Do chickens care if wall lizards taste like chicken?
House sparrows (Passer domesticus) are known to eat hatchling common wall lizards (Podarcis muralis), and now we can add the European starling (Sturnus vulgaris) to the list of exotic species eating common wall lizards too.
This European starling has a taste for exotic food, photographed by Jeremy Gatten, May 30, 2018, in Central Saanich.
Now I find myself in a funny spot. I like lizards, but I don’t like invasive species. Yet I find myself rooting for an invasive species because it is eating wall lizards.
The Early Bird post
Many photos have circulated in the last year showing an American kestrel feeding on common wall lizards. It is so nice to see a predator with a taste for exotic food.
But falcons aren’t the only lizard predators out there. Young lizards are taken by a wide range of animals, from spiders—yes, hatchling lizards get caught in webs and eaten by spiders—and snakes to raccoons, domestic cats and dogs, some larger birds like the great blue heron, and a wide range of perching birds.

American robin with a fast-food snack. Photo courtesy of Kathy Lamb.
This weekend I received a nice photo of an American robin who has also decided that young wall lizards are fast food worth the effort.
Spotted towhee with a sizable saurian snack. Photo courtesy of Jenny Whitfield.
Spotted towhees, house sparrows, dark-eyed juncos, Steller’s jays and a few other birds also love lizard lunches. But even with passerine predation, the wall lizard population is still beyond control.
American robin and its tetrapod treat. Photo courtesy of Kathy Lamb.
If you see something hunting and catching common wall lizards, please take photos and document the event. Records can be added to iNaturalist and emailed to me at the Royal BC Museum.
The Yard Birds post
Why wouldn’t you want a garden full of birds and their songs? My city garden is alive with birds—not rock pigeons, European starlings and house sparrows, but species native to this region.

Anna’s hummingbirds, golden-crowned sparrows, dark-eyed juncos, bushtits, bewick’s wrens, spotted towhees, American robins, Cooper’s hawks, merlins . . . the list goes on. A savannah sparrow spent a few sheltered days in our garden recuperating after a window strike. The long grass and other plants gave it plenty of shelter from predatory eyes.

Forget grass—go for complexity.
Small trees, bushes and ground cover all make a multilayered environment that is great habitat for a range of birds. Plant flowers for pollinating insects. Leave debris on the ground to provide shelter for other arthropods and prevent rain compaction of the soil.

The more complex the ground cover, the less you may need to water—the soil itself will be shielded from direct sunlight and will keep moist. If you are lucky, toads and salamanders will move in.
Cultivate seed-bearing plants that birds can harvest in addition to plants that you can enjoy, both visually and on the dinner table. In our garden, there is a fair degree of sharing with our avian neighbours. Our thornless blackberries, kale flowers and borage are a hit with the house finches.
Have a look at this video series. Obviously, based on the birds (cardinals—wow!), this is from eastern Canada (Ontario). The closest comparisons I have seen in my garden to the bold colours of a cardinal are a western tanager and northern oriole.

The same garden design principles apply everywhere—complexity, minimum disturbance, a water source(?). Wherever you are in North America, you can have a garden full of birds, colour, song and natural entertainment.
Fishy Photo post
While cleaning out files for the move of our offices (and the entire Royal BC Museum collection), I found an old black and white photograph. Most photos I have found are not useful given today’s publication standards. But this black and white photo by Barry Campbell, taken September 8, 1975, shows a basking shark off the north end of Flores Island, not far from Obstruction Island, along the west side of Vancouver Island.
The photographer said he had seen up to eight sharks at a time in this area—I can’t imagine seeing eight of the planet’s second largest shark. This species has been all but eliminated from British Columbia. Seeing one is newsworthy today; if someone said they’d seen eight, I would not believe them.
There is another note attached to the photograph, dating to 2002. The author of this newer note states that the fins are from a dolphin or some other whale. However, the photo clearly shows the shark swimming away from the photographer with the tail in the foreground, and further away is the dorsal fin flopped over to the left. The subterminal notch in the tail, which delineates the upper lobe, is clearly visible. The dorsal seems to be lighter coloured, but this could simply be the angle light is hitting the dorsal fin at relative to the tail. The shark was estimated to be about 7.6 metres long (back then he used the old measurement of feet, and this shark was about 25 of those).
I wasn’t fortunate enough to be on the CCGS Vector the year they saw a basking shark up near Langara Island, but someday I hope to see one of these massive plankton predators in person.
Anoles Appear post
Our garden centres carry tropical plants as well as temperate plants for your garden. Even hardware stores like Canadian Tire, Rona, Lowes and Home Depot carry a fair selection of plants. Indoor plants often come from way south of Vancouver Island, and in the last few years, there has been a noticeable increase in people reporting the presence of stow-away lizards—usually the brown anole (Anolis sagrei). In the southern USA, the brown anole is at a population density such that accidental transport is commonplace, even expected.
In 2020, I received several emails about brown anoles in plants bought from stores in BC. The appearance of lizards as stowaways likely is not new, but people’s awareness of exotic and potentially invasive species has certainly improved. The lizards generally appear as hatchlings, after being shuttled north of the international border as single eggs in potting soil.
These days, people can easily report the strange things that crawl out of their new house plants. Records are added to iNaturalist to document the trade in plants as a vector for herpetile dispersal. We even received a cuban treefrog (RBCM 2541) in August 2020, caught at a Rona outlet in Nanaimo. While these are not “wild” records in the strict sense in iNaturalist, they are important records of assisted dispersal.
In 2020, a male brown anole appeared in Duncan on Vancouver Island in a plant bought at the Canadian Tire outlet in that city. The record is in iNaturalist. This one was small, but not a hatchling as I had suspected. I had assumed lizards arrive as eggs, deposited one at a time in the soil in plant pots. This male was thin, and it could have been in Canadian Tire in Duncan for some time. Once it was caught, it was fed and housed in luxury accommodations, and now has been dropped off to keep me company.
Brown anoles have variable thermal tolerance, with those ranging further north having a lower critical thermal minimum. In other words, those which invaded northern regions have evolved tolerance for cooler temperatures—not bad for a species originating in the Caribbean. I wonder when climate change and a warming BC coast will intersect with the minimum temperatures needed for brown anole survival? I bet they can already survive here in a greenhouse or garden centre with some measure of climate control.
Because of the cold snap in the USA in 2021 and previous polar vortex events, there was strong selection for cold-tolerant lizards. We now know of anoles that can survive down to 4.4C (Stroud et al. 2020). This means that future stowaways may come from cold-hardy stock, assuming cold tolerance is persistent as a new trait. Tolerance of colder temperatures increases the chance brown anoles could survive here in southwestern BC in the right location. The rest of Canada still is too cold to support anoles outside of a cozy warm greenhouse.
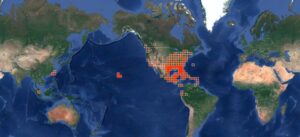
Brown Anoles are native to Cuba, the Bahamas and satellite islands, but they have become globe-hopping invaders, with a significant track record of establishment and spread in the USA since the first appearance in the Florida Keys (Garman 1887, in Lever 2003).
Kraus (2009) and Lever (2003) lists the following transport vectors for brown anoles, with additions by me based on iNaturalist records where the source of the lizard was specified (Canadian records in red):
Pet Trade: Canary Islands and all Hawaiian Islands.
Cargo stow away: St. Vincent, Grenada, Ontario, and the US states of Maine, Florida, Georgia, Texas, Pennsylvania, Alabama, Arkansas and Louisiana.
Intentional release: Florida
Unknown: Mexico, Belize, Bermuda, and the US state of South Carolina.
Nursery Trade: Cayman Islands, Taiwan, British Columbia, Alberta, Ontario, and the US states of Florida, Georgia, Texas, California, Arizona, Colorado, Vermont, Connecticut, Pennsylvania, New York, Delaware, Maryland, North Carolina, South Carolina, Michigan, Iowa, Kentucky, Indiana, Wisconsin, Mississippi, Illinois, Kansas, Oklahoma, Alabama, Arkansas, Louisiana, Ohio, South Dakota, Tennessee, Washington and Virginia.
Clearly the trade in ornamental plants is the biggest source of brown anoles, and we can expect more of these exotic lizards in the future. Fortunately they tend to ship one at a time, are unlikely to find mates, and records in iNaturalist across most of the United States represent single lizards, not established populations. But don’t be surprised if you shop at a nursery someday and see these guys flitting about in search of insects. If a male and female do end up in the same greenhouse, and there are plenty of plants where eggs can be deposited, a population could easily become established. Their colonization pattern is almost like our own, surviving in a biosphere, waiting for the rest of a planet to be terraformed. But in this case, lizards only have to wait for climate warming to give them the chance at freedom.
References: Kraus 2009. Lever 2003.
The Royal BC Museum and BC Archives are preparing collections for a move to the new Collections and Research Building that will be constructed in Colwood in 2024 (see the announcement in Victoria News). This process involves surveying collections, re-housing items that may not currently be housed with enough protection for transportation outside of the storage areas and conserving artifacts and records that are not stable enough to be moved.
The Issue of Broken Glass Plates
One thing that has come through the Paper Conservation Lab a lot is broken glass-plate photographs. Glass plates were used as a support for photographic slides and negatives early in the history of photography and continued to be used well into the first half of the twentieth century. Many have undergone treatment in the past couple of years (see my previous blog post, Fragments of Glass: Repairing a WWI Glass Plate Negative), but these treatments are time-consuming. Too time-consuming for the size of the backlog measured against the staff resources currently available. This means that some plates may need to be moved prior to being treated. How do you move sharp, fragile shards of glass-plate photographs in a manner that keeps the plates and those handling them safe, while also maintaining the intellectual links these plates have to the rest of their respective collections?
The solution must account for plates of varying sizes that currently exist in two or more shards of varying sizes. All of the shards will need to be kept together so that pieces don’t get lost, and each shard should be secured in place to prevent shuffling during transit. Many broken plates are currently stored in envelopes. In these cases, the individual pieces have been permitted to grind up against each other and move around, creating an opportunity for the sharp edges of the glass to cut up the delicate photographic emulsion layer–a situation that would only become exacerbated by the vibrations associated with a move. It has also led to some shards becoming shuffled and mixed up.

Fig 3. Collection manager Vicky Karas using the transmitted-light table to sort through shards of glass plate negatives to reassemble broken plates preparation for rehousing
The Re-Housing Solution
To provide flexible yet sufficient support and protection for broken glass plates for the collection move, these broken plates are being moved into custom-made pincushion trays. The base of this
tray is Coroplast, a corrugated plastic board of archival quality. The edges are one-inch Ethafoam strips with a quarter-inch Ethafoam sheet in the center. The edges and inset surface of these trays are then lined with Tyvek to create a smooth surface for the plates. The Ethafoam is attached to the Coroplast with hot glue and the Tyvek is held in place without the use of any adhesives—it is pressed into the inner edge creases of the tray and under the edge strips, then folded up over the top edges of the tray and tucked into an incision that runs around the sunken portion of the tray.

Figure 4. Construction of pin-cushion tray
The glass-plate shards are set into the tray, one piece at a time, beginning in one of the tray corners. Each shard is held in place with a small block of Volara foam, which is then pinned in place with a one-inch stainless steel dressmaker’s pin. Once enough pins have been applied to hold this first shard in place, the next shard is added and held in place with more pinned foam blocks until all shards are pinned in place.
These trays are 26 cm x 23.5 cm in the centre, which means they can accommodate the majority of plate holdings. For smaller plates, two can fit in a tray. The external dimensions of the Coroplast base fit within a standard-size record storage box. The hand cut-outs on either side allow for the trays to lifted in and out easily and the one-inch Ethafoam edges provide support for subsequent trays when stacked within the box. Trays are numbered so that their position can be precisely noted and tracked while they remain separate from their original locations and replaced after treatment in the future. (Each tray also has an “a” side and a “b” side, to accommodate instances where there are two plates to a tray.)
The pincushion trays that have been created for this project will provide excellent protection for each shard as they move from one collections storage building to another and await conservation treatment. The downside of this new temporary storage system is that it drastically increases the amount of space these plates are taking up—and in the context of most archival and museum institutions, space comes at a high premium. In this instance however, the museum is moving to a new facility with larger collections stores, so the increase in the footprint of this collection is not a major concern. It is also a temporary storage system.
In the years to come, these plates will be treated and returned to their original storage locations as time permits, and these boxes of trays will not be needed. However, the flexibility of these pincushion trays means that if any new glass plates are acquired broken, there is an existing storage method available and already assembled to provide temporary protection and support while they await treatment.
Acknowledgements
I would like to extend a big thank you to Vicky Karas who has put a great deal of time, care and effort into this re-housing project already . . . and there’s still more to come.
Thank you to Lesley Golding, collections manager for the BC Archives photographic holdings, for her insight during the development of this temporary storage plan and her expertise in regards to location tracking and control.
Free Fly Frog Food post
When I was a kid, I had a range of pets—mostly reptiles and amphibians; no surprise there. To feed my frogs, toads and lizards, I either snuck around the garden with a large aquarium dipnet and trapped flies (which were on our dog’s poop), or I trapped them with a more passive technique.
Today (February 12, 2021) I answered an email about yet another brown anole (Anolis sagrei) that had arrived on some tropical plants, this time at a Canadian Tire outlet in Duncan, BC.
The anole’s new owners are going to set up a terrarium for this little lizard, and why not. They are charming terrarium inhabitants. I gave the owners advice on how to feed and water the lizard, and I decided to detail the trap I made as a kid so they could harvest flies as free food for their new pet.
Here was my trap, made from household items that, when I was a kid, went in the trash (this was pre-recycling).
1) Cut a pop bottle in half and recycle the cap.

2) Cut slots in the top half of the bottle so it fits inside the lower half of the bottle.
3) Cut a hole in the bottom of the bottle (here the hole is in one of the nubbins—corrugations—at the bottom). Flies will find their way in, but when you come to pick up the trap, they likely will not find this hole and will fly upwards.

4) Bait the trap and put the bottom halves together.

5) Tape a parmesan cheese container over the top. Why I use a parmesan cheese container will be explained in a second.
6) Wait.
7) When flies have entered, pick up the entire apparatus and use your thumb to prevent escape out the hole in the corugation.
8) Flies likely will go up and into the parmesan jar when they are disturbed.
9) Remove the tape carefully, and in one smooth motion, lift the parmesan container and screw its cap (this one is red) in place. Bugs will be nicely contained.
10) Now you can simply place the container in your terrarium with the lid open (this is why I used the parmesan container—it has a convenient flap to open, with fly-sized openings). If you are worried about flies escaping before you close the terrarium, then put the parmesan container in the fridge to cool the flies. Then once flies are cool, put the container in the aquarium and let the flies warm up—and watch your pets devour their dipteran dinner.
Chilliwacked post
Common wall lizards, Podarcis muralis, were first detected in Chilliwack in late June 2020 by Frances and Georgina Wetmore.

Photo courtesy of Alex Wetmore.
The report came to me in late September to verify the identification, and without a doubt, the photo captured a common wall lizard. After a few quick emails, Christian Lodders pinpointed the epicenter of the colony along Kathleen Drive in Chilliwack. Several gardens in the area now are known to have wall lizards—in other words, they are firmly established. Newly hatched wall lizards were seen this autumn.
How did they get from Vancouver Island (or Denman Island) to Chilliwack? Who knows. Lizards could have been shipped by accident as eggs in a plant pot. One nest creates an instant population of 5–10 lizards, genetic bottleneck notwithstanding. Maybe a gravid female was a stowaway in some camping gear? Mr. Lodders talked to homeowners in the area, and there is no indication anyone intentionally released lizards.
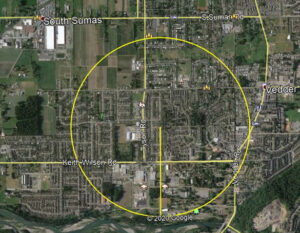
As on Vancouver Island, the end result is the same for Chilliwack—if lizards are not eradicated, they will spread in the area. Under their own power, lizards can disperse one kilometre in 10 years (as outlined in the yellow circle above), and with assistance from humans, their range can expand far more rapidly in the area. With a little help, they could make their way to Washington, and that state would have two wall lizard species: Italian wall lizards (Podarcis siculus) are already established on Orcas Island.
Abstract
Premise of the study: Many arctic-alpine species have vast geographic ranges, but these may encompass substantial gaps whose origins are poorly understood. Here we address the phylogeographic history of Silene acaulis, a perennial cushion plant with a circumpolar distribution except for a large gap in Siberia.
Methods: We assessed genetic variation in a range-wide sample of 103 populations using plastid DNA (pDNA) sequences and AFLPs (amplified fragment length polymorphisms). We constructed a haplotype network and performed Bayesian phylogenetic analyses based on plastid sequences. We visualized AFLP patterns using principal coordinate analysis, identified genetic groups using the program structure, and estimated genetic diversity and rarity indices by geographic region.
Key results: The history of the main pDNA lineages was estimated to span several glaciations. AFLP data revealed a distinct division between Beringia/North America and Europe/East Greenland. These two regions shared only one of 17 pDNA haplotypes. Populations on opposite sides of the Siberian range gap (Ural Mountains and Chukotka) were genetically distinct and appear to have resulted from postglacial leading-edge colonizations. We inferred two refugia in North America (Beringia and the southern Rocky Mountains) and two in Europe (central-southern Europe and northern Europe/East Greenland). Patterns in the East Atlantic region suggested transoceanic long-distance dispersal events.
Conclusions: Silene acaulis has a highly dynamic history characterized by vicariance, regional extinction, and recolonization, with persistence in at least four refugia. Long-distance dispersal explains patterns across the Atlantic Ocean, but we found no evidence of dispersal across the Siberian range gap.
Keywords: AFLP; Caryophyllaceae; Silene acaulis; arctic-alpine; disjunct distribution; phylogeography; psbD-trnT(GGU) spacer; refugia; rpL32-trnL(UAG) spacer; trnL(UAA) intron; trnL(UAA)-trnF(GAA) spacer.
In June of 2018, I was invited to participate in a BioBlitz at the Hakai Institute on Calvert Island. From the initial planning stages, it was clear that this was going to be an exciting and scientifically rich journey. The organizers (Dr. Brian Starzomski, Sara Wickham, and Gillian Sadlier-Brown) invited me to help them to survey the island for “all terrestrial life.” With such a daunting task at hand, I was relieved to discover that a number of other experts, including other entomologists, botanists, lichenologists, ornithologists, and general naturalists, had also been invited.
Following a 1.5 hour float plane flight north from Campbell River, we arrived at Calvert Island. Situated about halfway between the northern tip of Vancouver Island and the southern tip of Haida Gwaii, the island truly represents the heart of the Great Bear Rainforest. From the first moments at the facility, we began our task of identifying as many of the species inhabiting the island as possible. The next seven days were spent traversing to the beaches that make up the northwest coast of the island, hiking to inland hilltops, and scouring lagoons at low tide. In all locations, I collected insects, while others collected insects, but also plants, spiders, lichens, and observations of vertebrates. Evenings were spent carefully preparing the specimens gathered each day. A lab full of equipment and enthusiastic staff made it possible to identify specimens, photograph them, and extract tissue. The extracted tissue will be sent to the Centre for Biodiversity Genomics at the University of Guelph for DNA barcoding.
The week was beautiful and tiring, but extremely productive. The insect and spider specimens, over 1300 in total, have made their way to the Entomology Collection at the Royal BC Museum. They are being further identified to species where possible. This single week of field work will produce a valuable snapshot of the insects and spiders inhabiting a special portion of British Columbia’s central coast. They could help us to answer any number of questions about the patterns of biodiversity in our province and the forces that affect those patterns. Stay tuned here for future analyses of these data.

A beautiful coastal habitat, alive with insect diversity. North Beach, Calvert Island. Photo by Joel Gibson.
On March 11, 2011, a magnitude-9 earthquake occurred off the coast of Japan, which triggered a monumental tsunami. The tsunami created massive destruction, taking lives, collapsing buildings, roads, railways and a dam and triggering a series of nuclear accidents. Tsunami debris continues to wash up on the shores of North America and Hawaii, seven years later. The Royal BC Museum has become the repository and permanent home for the biological material collected from this tsunami debris.
Fifty boxes of organisms collected from tsunami marine debris, both wet and dry, were shipped to the Royal BC Museum. This included some debris that was comprised of man-made objects (substrates), to which marine life was still attached. This sea life was primarily marine invertebrates such as barnacles, bivalves, bryozoans, and hydrozoans. The substrate materials included pieces of fibreglass, masonry, plastics, rubber, Styrofoam and wood, from objects such as docks, boats, buoys, household items and buildings. I agreed to carry out a conservation assessment of the dry material and to advise staff on the best way to store it.
The purpose of storing the Japanese tsunami debris collection at the Royal BC Museum is the long term preservation of the marine life. Research work will be carried out to identify any invasive species that have made their way here from Japan. The stability and the longevity of the substrates will influence the long term preservation of the attached sea life. If the substrates degrade, break apart and disintegrate, the attached colonies and individual specimens will be lost.
I assessed the collection and found that the dry, synthetic, substrate materials were deteriorated and dirty. All were discoloured, bleached, brittle and/or cracked. Some of the substrates were missing sections. There were no plans to remove residual sea salt and sand from the substrates, as this would damage the attached marine life.
I consulted with our senior conservator Kasey Lee, on the best storage conditions for plastics and rubber, as she had just returned from attending a Conservation of Plastics workshop at the Canadian Conservation Institute. The Conservation experts recommended that plastic materials be stored under freezing conditions.
I proposed to Dr. Henry Choong, our curator of Invertebrate Zoology and Heidi Gartner, our Collection Manager of Invertebrate Zoology, that the dry tsunami debris material be stored in a chest freezer, at minus 20◦ C. This would halt the deterioration processes of the plastics, and prevent the loss of marine life. It would also reduce the off-gassing of volatile acids from substrate materials such as wood and rubber. This would preclude the development of Byne’s disease, the chemical and physical breakdown of carbonate-based materials such as shells, due to exposure to acids.
I also suggested that, as a precaution, unbuffered acid-free tissue paper or card be added to the storage containers. This would serve as a humidity buffer because the deterioration processes of some plastics are accelerated in the presence of moisture. As rubber will continue to oxidize in freezing conditions, to slow down its deterioration rate, I also recommended that an oxygen scavenger be added to the sealed storage boxes containing the rubber substrates.
I provided advice to contractor Pascale Archibald on proper storage containers and supplied her with some of the conservation – approved storage materials to house the tsunami debris collection. She wrapped individual objects and cushioned them in unbuffered, acid-free tissue paper in clear polystyrene Durphy boxes. Pascale also made custom storage mounts for the irregular-shaped substrates, using corrugated polyethylene known as Coroplast and polyethylene foam called Ethafoam.
Re-housing of the dry tsunami debris material for long term storage is now completed. The substrates are supported without damaging the attached sea life. An oxygen scavenger will be added to the containers with the rubber substrates and all of the newly-housed dry material will be moved into a chest freezer in the Invertebrate lab.

The green plastic lid is now stored on a cushioned mount made of Ethafoam and polyester fiberfill in a custom-built Coroplast box.
Identification of several of the plastics will be carried out with FTIR analysis (Fourier-transform infrared spectroscopy) at the Canadian Conservation Institute.
A temporary display of the Japanese tsunami debris collection can be viewed in the Royal BC Museum Pocket gallery in Cliff Carl Hall, until Sunday, October 14, 2018.
In June of 2018, I was invited to Calvert Island by the Hakai Institute to take part in the Hakai Seagrass BioBlitz.
The Hakai seagrass surveys, part of an ongoing long-term research project, focus on unravelling subtidal patterns of microbial ecology, with a focus on several ecologically important marine macroalgae and seagrass (Macrocystis, Nereocystis, and Zostera). Sampling was done offshore with divers, and also in surfgrass habitat on the rocky shore.
The seagrass biomass provides food, habitat, and nursery areas for numerous adult and juvenile vertebrates and invertebrates. A single acre of seagrass may support thousands of fish, and millions of small invertebrates. Because seagrasses support such high biodiversity, and because of their sensitivity to changes in water quality, they have become recognized as important indicator species that reflect the overall health of coastal ecosystems.
During our survey in June, we sampled focal species on various nearshore and intertidal localities around Calvert Island. This work is part of other surveys done at local and regional spatial scales and along the Central Coast in conjunction with the Hakai long-term seagrass and kelp monitoring programs conducted by the Salomon Lab at Simon Fraser University. Our team on the survey comprised experts from the Beaty Museum, Florida Museum of Natural History, Smithsonian, Friday Harbor Labs, and of course, the Royal BC Museum.
Specimens collected include polychaetes (worms), diatoms, amphipods, hydromedusae (small jellies), and hydroids. I am a hydrozoan specialist, so my task was identifying and studying the hydrozoans (hydroids and hydromedusae) found living on the seagrass.
All specimens were photographed, databased, and sent to be barcoded. The Tula foundation provided the funding for the barcoding through the Barcode of Life Data System-BOLD, University of Guelph. Cnidarian samples (hydroids, etc) will be sent to the Smithsonian for DNA work, as they require different treatment.
The Royal BC Museum’s participation reflects the institution’s important role as a member of the larger biodiversity research community- increased taxonomic resolution of the flora and fauna associated with seagrass helps us understand the structure and dynamics of the seagrass habitats. Another goal of this year’s, and other bioblitzs, is to generate an exhaustive field guide to the algae and invertebrates that can be found in seagrass habitats around Calvert Island.

The drive to Golden
Stories of the past inform and shape our understanding of the present. But what happened to the stories less shared, either in public institutions or private sphere? The Royal BC Museum as a public institution is committed to exploring, preserving, and sharing British Columbia (BC)’s diverse heritage. With guidance from the Advisory Committee and in partnership with the South Asian Studies Institute (SASI) at the University of the Fraser Valley (UFV), our work on the Punjabi Canadian Legacy Project (PCLP) Phase 1 in 2015-2017 resulted in the development of new collections, new online educational tools, and community consultations throughout the province. This work was made possible by the generous support from the H.Y. Louie Co. Ltd, as well as the strong institutional support from host institutions around BC.
In the 2015-2017 Phase 1 consultations, communities have expressed the wish to create strong, diverse legacy projects, including K-12 and public education materials, travelling exhibitions to traditional and non-traditional public exhibition spaces, community engagement and development, and other communications. The Advisory Committee are currently helping to shape a fundraising plan for these projects.

The site in Golden, BC believed to be the location of the first Sikh temple in North America in the early twentieth century.
Following the consultations feedback, currently we are working on the Phase 2 of the PCLP with a modest BC Museums Association Canada 150 fund administered through the UFV, ending in March 2018. In response to community feedback on further outreach, the SASI and the Royal BC Museum collaborate to hold community workshops to continue engagement and collect family history. From summer to winter 2017, we completed workshops in Golden, Prince George, Paldi/Duncan, Kelowna, Vancouver, Surrey, and Abbotsford with support from the host institutions. The project coordinator stayed in each region to collect oral history after the workshops. By March 2018 we estimate to complete 80 interviews. The stories we have collected so far in the project reveal amazing connections of the Punjabi Canadian communities across the province and overseas.
Over the course of this work, our community connections opened the project team’s eyes to community resilience and networks. In July 2017, families with former ties to Golden, BC, a Rocky Mountain town where communities believe to be home to the first gurdwara in North America, held a reunion in Surrey. The event gathered 200 people from around the province who moved away from Golden for work or children’s education. The project team attended this reunion, and met some of the families again in Golden as they scheduled their family annual trips to Golden at the time of our community workshop.
In Golden, we heard about the first Sikh wedding in 1972 from the broom and bride, Swarn and Balbir Patara. At the time there was no Sikh temple in the area. What communities believe to be the first Sikh temple in North America in the early twentieth century left no relics in Golden. They had to ship the utensils, religious items and even a priest from Paldi near Lake Cowichan to perform the wedding ceremony.

Left: Swarn and Balbir Patara, the first Punjabi couple to get married in Golden with a Sikh wedding in 1972, speaking to us about the local history in front of the plaque in Golden, BC. Right: Swarn and Balbir Patara’s wedding in 1972.

The two local hosts, Davo Mayo, the President of the Paldi Gurdwara and Kathryn Gagnon, Manager/Curator of the Cowichan Valley Museum & Archives, welcomed the attendees at the Community Workshop at the Paldi Sikh Temple in Paldi, BC in October 2017.
In Paldi, we heard again about the traditions in the early days, stories that people shared in other communities as well. At the time, as the community population was small, people travelled to gather at different gurdwaras for holiday celebrations and commemorations together. For example, the Independence Day of India was celebrated at Masachi Lake Gurdwara, and Vaisakhi was celebrated in the Victoria Gurdwara. For these gatherings, all the community members got together in the temple from different cities, and to accommodate them, a lot of mattresses were stored at the temple premises. Many today remember it vividly and would love to keep this tradition alive.
In the amazing journey of working on this project, the connections among many families and communities through generations and places that we have learnt along the way have been heartwarming and great motivations for us to continue this legacy building project.
For more general info on the Punjabi Canadian Legacy Project click here. To see the Phase 2 project updates, click here. A short video on the Paldi workshop can be found here.

Community Workshop at the Golden Sikh Temple in Golden, BC in July, 2017.

Community Workshop at the Ross Street Komagata Maru Museum at the Khalsa Diwan Society of Vancouver in November, 2017.
In July 2017 the Royal BC Museum loaned John Lennon’s 1965 Rolls Royce Phantom V to Rolls Royce Motorcars Ltd for an exhibition at Bonhams of London, UK. I had the distinct pleasure of overseeing the delivery of the Lennon Rolls Royce on that trip.
So how do you drive a three ton car with a custom paint job that is fragile and irreplaceable to Britain? The answer is you don’t. You fly, of course.
The Lennon Rolls Royce is more like a moveable artifact than a car, with most of its value associated with its history and aesthetics rather than its monetary collector value. There’s nothing like it in the world. Accordingly, care and planning for the loan were critical. Even a small ding or mechanical failure means a permanent change to the artifact, one that might require expensive restoration work, or more likely, will stay with the car for the rest of its life. The car is not in perfect condition now, but most of the blemishes are actually associated with the history of the car before it arrived at the Museum. John Lennon used the car extensively in the late ‘60’s and later lent it out to other well-known rockers including the Rolling Stones, the Moody Blues and Bob Dylan. Oh, the stories it could tell.
My epic adventure with the Lennon Rolls Royce began with loading the vehicle into an enclosed trailer for the trip to the mainland, where it would be crated for its overseas flight. It rode the ferry without notice, tucked safely inside its enclosed transport. So the first leg involved a truck trailer and a boat.
Once on the mainland, the Rolls Royce was unloaded from the trailer under its own power, measured and examined, and a crate design was finalized. A carpenter set to work building a reinforced platform that looked like it was built to support an office building. In the end, it is likely that the crate weighed almost as much as the car itself.

When the crate base was ready, I drove the Rolls Royce up a precarious ramp onto the platform. This was no easy feat, as the car is right-hand drive, sports a choke, various vintage knobs and levers, and is not driven regularly, so tends to run a bit rough. There were a few anxious moments when it actually ran out of gas on the ramp, and I feared that the engine-assist breaks would fail to keep me (and the car) from rolling backward into the nearest obstacle. Fortunately the hand brake worked and we were able to tow it up the rest of the way onto the crate base.
The car was then strapped down over and propped up under the axels, just in case the tires deflated in flight, allowing the car to sink and the straps to go slack. The crate was made only slightly larger than the car, with no pads or cushioning as these would abrade the paint as a result of vibration during movement.
The Rolls Royce then flew into Stansted Airport, north of London, UK. The countryside around that part of Britain is amazingly familiar, with the same blackberries in bloom, chestnut trees laden with spikey balls, and, surprisingly to me, fields of Canola (I hail from the prairies). We met the Rolls Royce in the cargo hangar after it cleared customs. I would have loved to see the looks on the faces and hear the banter of those customs agents, but that business took place behind closed doors. The vehicle literally rolled out along a conveyor belt, was lifted off by a humungous fork lift, and deposited into a shower-curtained lorry (when in Rome…). It fit, with at least an inch to spare. See, it’s all about the planning.
Then we were off again through the English countryside, to a tiny little town between fields, churches and tudor-style inns to the outfit that would uncrate the Rolls Royce. When we arrived, the team immediately set to work unbuttoning the crate with the expectation of children on Christmas morning. They knew what was inside and several took a few moments to call their friends and loved ones to brag about their task. Everyone took pictures. I fended off overly-enthusiastic bystanders who wanted to climb in.
Once again there was an adrenaline moment when the improvised ramp split as the Rolls Royce was driven down off the crate base. A piercing crack was heard, followed by the sight of the car dropping a few centimeters. As rubber ages, it becomes stiff and brittle and the tires on the Rolls Royce were definitely on their last… well, legs. A blow out would be disastrous. Just the jarring of the slight fall could have dislodged a brittle joint or loose paint. Fortunately no damage was done and the ramp was reinforced before the front wheels met the same fate.
The next leg of the trip involved driving the vehicle onto a special car-carrying lorry. Fortunately this was a slick job with nothing left to guesswork and ingenuity. The truck actually disgorged its box onto the level pavement where the vehicle could be easily driven inside using fold out metal ramps. Then the driver could take his leave through the overhead side door. These people were professionals with all the best equipment. I left the driving to them.
Once safely loaded into the car-transport lorry, we embarked on another long drive through the English countryside, destined for P & A Wood, official Rolls Royce service company. They had been contracted to put new tires on the Lennon Rolls Royce, and to give it a thorough inspection by trained mechanics, experts that we don’t have on staff at the Royal BC Museum. I marvelled at how the lorry navigated the tight traffic circles, passing through one quaint town after another. My driver was kind enough to explain to me the processes for thatching roofs and creating the decorative pargetting on the plaster cottages we passed. The sheep and cows raised inquisitive heads and the ubiquitous pigeons dared us to hit them as they scavenged at the roadside.
After an hour or so, we finally reached our destination, a very impressively appointed set of newer buildings sporting Rolls Royce/Bentley showrooms, parts stores, and two service shops. We unloaded the Rolls Royce on the street, almost causing a couple of accidents as passing motorists craned to have a look, and drove into a spotless building that looked more like a showroom than a service department. The Lennon Rolls Royce was deposited amongst its kind, with Fred Astaire’s 1927 Rolls Royce Phantom I just ahead of us. I felt a sense of awe, but surprising to me, so did the various workers inside, who were thrilled to see the famous John Lennon Phantom V up close. The digital shutters began snapping again all around me. It was and will always be a privilege to be in the company of such a world famous piece of history.
I left England shortly after depositing the Lennon Rolls Royce with P & A Wood, headed home to my office job at the Royal BC Museum. The Curator, Lorne Hammond, would take my place in London a few days later, assisting with the installation and media events at Bonhams. The rest will be Rolls Royce history. It appears that they have finally forgiven John Lennon for daring to mess with their refined brand image. Time has a way of sorting out those things. But the excitement around the Lennon Rolls Royce appears to be timeless.
B.C. is home to more than 3,000 flowering plant species; the richest flora in Canada. This botanical exuberance is our legacy of a complex geological history coupled with a varied landscape and climate. The result is the occurrence of many rare species in our province.
At the Royal British Columbia Museum Herbarium, we keep dried and pressed examples of the province’s plants including at least one specimen of each of the rare species. These identified and labelled specimens are accompanied by information about place and date of collection, collector, and often details of the local environment. The specimens serve as proof, or vouchers of the species in B.C. and as a reference for comparison of newly collected material.
Rare plants can be classified into four groups:
1. Those that occur at a single or few localities, each population with few individuals.
2. Those that occur at several localities and are locally common.
3. Those that occur in many areas, but in low numbers.
4. Those that occur in a restricted area but are abundant
Rare plants, in many cases, are endangered plants because compared to widespread species even minor disturbance can cause them to disappear or be seriously affected. However, some rare species on distant difficult-to-reach mountains may not be endangered, whereas large populations in areas under intense influence from human activity can be seriously endangered. Climate change is one disruptive phenomenon that will reach all plants.
My work focuses on the environmental history of the province and how that history might explain the distribution of rare species. Furthermore, lessons learned from ancient history provide insights into the potential fate of our flora, including rare species, in the event of major climatic warming associated with the “Greenhouse Effect”.
With the potential for warming of about 2-4 degrees Celsius, in the mean annual temperature, we can expect major changes in vegetation and consequently major impact on rare species. In this context all rare species on whatever scale whether local (such as Galiano Island), regional or provincial must be considered potentially endangered. The reason for this concern, is that we do not know how plant species will respond to climate change. We do know that change will effect plants somehow. Rare species are most sensitive because even the smallest impact may destroy a population; and because a species is rare, once it disappears, there will be no reservoir in British Columbia from which it can recover. The greatest concern is for plants that are not only rare and impacted by climatic warming but also under stress from direct human activities such as logging, agriculture or urban development; For all rare plant species we must consider reducing these added stresses to help them survive the broader assault of Global warming.
As climate change proceeds we may expect some rare or endangered species to benefit and expand. These would include species of dry open habitants such as the garry oak (Quercus garryana) woodland and meadows of south east Vancouver Island and adjacent Gulf Islands. This region contain a very high concentration of rare plants such as the endemic Macoun’s meadow-foam (Limnanthes macounii), bristly manzanita (Arctostaphylos columbiana), golden Indian Paintbrush, (Castilleja levisecta) a balsamroot (Balsamorhiza deltoidea) and many others. The rare and endangered species of the arid lands of the southern Okanagan – Thompson and Kootenay may benefit; provided we conserve sufficient habitat for them and provide corridors for their migration. These species thrive under hot dry setting and could spread northward and up-slope as forests and woodland succumb to drought. Good examples include the Mariposa lilies (Calochortus spp).
The losers will be plant species of moist and cool or cold settings; inhabitants of the alpine zone and wetlands. Eventually, forests will spread up-slope, eliminating open alpine habitats and species especially on southern low elevation alpine areas. In some places weedy species such as knapweed, may expand into pristine subalpine and alpine zones as live stock carry seeds through expanding grasslands.
Wetlands in all parts of the province, especially dry regions such as the Gulf Islands and adjacent Vancouver Island and the southern interior, will be at greatest risk. Studies of bog and lake cores from these areas clearly reveal that water levels, water chemistry, and as a result, plant communities change markedly as climate alters. For example, in our area many smaller lakes and ponds were neutral to alkaline, precipitating the limey sediment called marl. Some medium-sized lakes were completely dry in Interior B.C. where the mean annual temperature was about 2 C warmer. Once suitable conditions for a wetland plant disappear, the plant disappears. Unlike terrestrial plants, wetland plants cannot disperse up-slope or up-valley along a corridor or gradient of suitable habitat. Somehow they must jump to the next suitable wetland before the one in which they live dries up. Combine natural change of wetlands with increased demand for water by livestock, moist sites for agriculture, drinking water, irrigation and invasion by introduced species such as Purple loosestrife and you have a prescription for very difficult times for endangered wetland plant species.
Each local area should know what rare and endangered species occur there and where they grow. Learn how to recognize your rare plant residents. Consider adopting the plants and their locality and monitor the population for increases or decreases. Develop local policies and strategies to minimize the impact on these special plants and places. Take responsibility for conserving the natural legacy of thousands of years of history; some of those rare species may become crucial elements of the new vegetation that is to come.
*Article modified from original printed in the Active Page Galiano Monthly Magazine, January 1991.
Protected: In a Manner of Speaking download
As many of you will have noticed, there is a new archives collection search database. It uses a system called AtoM (Access to Memory) which is also used by City of Vancouver Archives, UBC Rare Books and Special Collections, SFU Archives, the World Bank and others. It is quite different from the old “blue and white site” with which we have become familiar, and with which – with all its faults – we have become comfortable. However, the old search site, dating back to the early years of the internet, will soon be gone, having survived longer than most websites.
Detailed instructions for the new system are available as well as a brief search guide, but for those who have used the old site, the following search tips may be useful.
>Use * (instead of ?) as a wild card (e.g. judg* to retrieve judge, judgement, judgment, judging, etc.), and ? (instead of #) for single character substitution (e.g. ver?g?n will find Veregin, Verigan, Verigen as well as the more common spelling of Verigin).
>Use AND (all caps) to combine terms (e.g. alma AND russell) and quotation marks to specify an exact match (e.g. “alma russell”) Otherwise the search will be treated as an OR, i.e. match any terms, search. For example, alma russell will retrieve descriptions with Alma Russell as a phrase and with both Alma and Russell somewhere in the description (not adjacent), but also records with only Alma and with only Russell.
>With the exception of the Identifier field in Advanced Search, search terms can be in upper or lowercase. When using Boolean operators (AND, OR, NOT) – which must be in uppercase – enter the entire search statement in uppercase if you wish to avoid having to shift from lower to upper case, e.g. ALMA AND RUSSELL AND WAR.
>Enclose any term or number with a hyphen or other special character in quotation marks, e.g. “MS-0055”, “I-00204”, “e/c/w96a”. Otherwise the hyphen, slash, etc. will be ignored. For example, if MS-0055 is searched without quotation marks (other than in the Identifier field), the results will include records descriptions with only ms (or MS) and only 0055 present as well as those with both.
>The Identifier field option in Advanced search is unique in that it is not only case sensitive but it does not require quotation marks to search a number with hyphens designated as an identifier (e.g. call number, accession number, item number). A string of such numbers can be entered with spaces in between to search on any of the terms entered, e.g. MS-0054 MS-0055 G-02580 G-02581 G-02582. Note that not all numbers are considered identifiers, e.g. HP photo numbers are only searchable in a basic or Any Field search.
>Use the General material designation filter in Advanced search to limit results to one type of record, e.g. textual, sound, etc. By default the system searches all types (except library material and vital event records which are in separate databases).
Limit search results to series level descriptions to view textual records at the same level as the blue and white site. If you know the MS or GR numbers, use the Identifier search option in Advanced Search which will also yield only the series level description. Similarly, a PR number will produce only the fonds level description.
>When viewing an individual series level record description use the Quick Search feature in the left-hand column to search for specific lower-level record descriptions. If the series level description has an attached finding aid (in the Notes area), click on the link to open the PDF and use CTRL-F to search for specific files or items.
Next: Basic search vs Advanced search
By-Catch post
When most people think about by-catch, they think of shrimp fisheries and the thousands of tons of sea life which gets caught along with shrimp. The desired species are kept, and most everything else is dumped back into the sea or onshore.
By-catch can be a great source of new specimens for museum collections. When I go out with the Department of Fisheries and Oceans on deep sea trips I am able to sample the catch and preserve what I want. Some specimens are new to BC, some are just cool – like Brown Catsharks. We keep some specimens because they have an interesting story – like the Dreamer that had swallowed a huge wad of packing tape.
By-catch from my samples and individually donated specimens also benefits other museum researchers. Melissa Frey and I carefully dissected a large copepod (a species of Pennella) that had attached to the back of the eye of a Louvar. I get the fish, the invertebrates collection gets the massive ectoparasitic copepod (I win – both in non-creepiness and biomass). Sometimes we save other parasites (flukes to leeches) from aquatic vertebrates for the “aquatic” invertebrate collection, as well as mites and ticks from reptiles, to fleas from mammals, and some funky flies (Family Streblidae) which are ectoparasites of bats. These go into the entomology collection.
This week, the entomologists returned fire with another specimen – this time from their sampling program at Hotsprings Cove, Maquinna Provincial Park. Their insect traps catch the occasional shrew, toad and salamander, and today I received this tiny Western Red-backed Salamander for the amphibian and reptile collection.
 Again I win – a cute salamander for the collection and all I had to do was walk down stairs and say, “Thank you very much”. Such is the nature of museum collaboration.
Again I win – a cute salamander for the collection and all I had to do was walk down stairs and say, “Thank you very much”. Such is the nature of museum collaboration.
PEACE post
This past summer I had ‘one of those moments’ – a moment that where you are suddenly aware that you are on the brink of something special – and it happened at an unexpected place: a car rest stop.
View of the Peace River from the car rest stop.
Yes, I had this ‘moment’ while at a car rest stop, at the top of a hill, overlooking the Peace River. I was struck by the beauty of the river and surrounding area. However, the feeling was compounded by the fact that I was also about to take part in my first ‘bioblitz’.
A bioblitz is an event that takes place over a short period of time where scientists, naturalists, and volunteers attempt to identify as many species as possible in a specific area. Myself, and 5 colleagues from the Royal British Columbia Museum (RBCM), were about to join a bioblitz organized by the Biological Survey of Canada (BSC) and the Yellowstone to Yukon (Y2Y) Conservation Initiative. Over a five days period we would be joining at least 20 other participants in documenting the biodiversity of the Peace River area that lies between Hudson’s Hope and Fort St John along the Peace River. The area was chosen because 1) it is an area that would change drastically with the construction and flooding caused by the BC Hydro Site C hydroelectric dam and 2) because relatively little is known about the flora and fauna in the area. As a biology nerd, I was excited to be participating in such a large collaboration in such an important area.
The RBCM participants were Claudia Copley (Entomology Collection Manager), Darren Copley (Bird and Mammal Preparator), Erica Wheeler (Botany Collection Manager), Ken Marr (Botany Curator), and Kristiina Ovaska (Invertebrate Research Associate). The other participants included researchers from all over the province (and few from outside the province!) that work for a range of private and professional (both governmental and NGOs) organizations and businesses. However, the best part of the bioblitz was that we were joined, supported, and guided by local naturalists, land owners and First Nations.
Darren and Claudia Copley scanning the skies for birds.
Erica Wheeler collecting plant specimens with the help of a local land owner.
Kristiina Ovaska, RBCM Invertebrate Research Associate.
A collaborative field team from the RBCM, Ministry of Environment, and the local community.
Local young naturalist.
Local land owners sharing their knowledge of the area.
Through the incredible collaboration of Y2Y and BSC, researchers were connected with local First Nations, land owners, and boat owners who shared their time, knowledge, and resources – thereby, allowing researchers to visit unique and difficult to reach habitats. During the five day bioblitz we able to explore an incredible diversity of habitats and sampled wetlands, sloughs, rivers, creek slopes, cascading water seeps, rotting tree falls, and forest leaf litter.
Sampling wetlands.
Sampling along the river edge.
A stunning and unique environment along the Peace River.
I was thrilled to get to work with our Research Associate, Kristiina Ovaska, in the Peace. Not only is Kristiina an expert on terrestrial gastropods and amphibians but she is a talented photographer. Here is a small sampling of the amazing photos she took of the local biodiversity:
Caught!
Freshwater snail.
Terrestrial slug.
Spider.
In short, it was an incredible week. My feeling, my ‘moment’, at the car rest stop on the first day was more than justified. I had an amazing week learning from, and working with, incredible researchers and naturalists. However, I was most touched by the connections made with the local community and First Nations. They are a group of people that are very passionate and committed to wanting to preserve the Peace River in its current state. The habitats, the plants and animals, and the people of this place are deeply connected.
On a Dark Path post
Even before I was hired as a museum curator, I was asked to write 28 species entries for some obscure fishes in Animal: the definitive visual guide to the world’s wildlife. I had to take painfully dry science and distil it into 45 or so words that anyone could understand. That set me on a dark path (cue sound effects) to the hidden vaults of museum collections and my laboratory. Who doesn’t want a laboratory full of bones?
The skeleton of an Olive Ridley Sea turtle in my laboratory.
Museum work dwells in obscurity, and a great proportion of BC citizens have no idea that RBCM staff publish academic research on par with university professors. We have an annual research day where museum staff tell other staff-members what is going on here – because we often tend to work in isolation and forget to trumpet our own successes. Short story is: Yes we do research – but to quote a friend’s daughter, “It’s not rocket science.” Some people question the value of museum research since it is not generating food, medicine or some other product essential to the Canadian economy. But not everything has an immediate practical value.
A North Pacific Argentine (Argentina sialis) from BC blasts off into academic obscurity. Modified from: http://www.forwallpaper.com/wallpaper/takeoff-launch-rocket-fire-smoke-baikonur-240897.html
People spend hours debating whether Pluto is a planet or not, and we spend billions of dollars on rockets and probes to examine our distant neighbours. We won’t colonise that planetoid/planet – so why spend so much energy debating its celestial designation? We debate it because people love to learn about new things – and let’s face it, people love a good argument.
Pure science has its own unique value – it is unlike other fields of study. If we look back, there was a time when electricity was considered a mere curiosity with no practical application. That is hilarious from a 2015 vantage point, as I bask in fluorescent light, the hum of the light’s respective ballasts in my ears, with several electronic gadgets at my fingertips. Much of what we do in museum research and collections development appears to have no practical value. We collect and study objects with no idea how they will be used by future generations. Perhaps the Louvar and Finescale Triggerfish are the vanguard to future drastic changes to the Pacific Ocean’s ecology. Perhaps not. I have no idea whether anyone will ever look at those fish again.
We commonly get accused of stamp collecting – merely trying to collect one of everything just to say we have a complete set. Perhaps my collection of Star Wars cards predisposed me to a life as a museum scientist. Now that I think of it, we actually have packs of Star Wars cards in the RBCM’s Human History collection. I wonder if the museums packages still have the horrible tasting chewing gum? But I digress…
Instead of being satisfied with one of everything in Natural History (a philatelic approach), we keep collecting. We collect year to year from different areas, males, females, young, old, eggs, tadpoles, oddities, etc., and in all Natural History disciplines, the one commonality is that we do specimen based research. We don’t have labs full of machines that go ‘ping’, unlike university biology departments – which to me look more like chemistry labs these days. But at a museum, we still have preserved specimens – plants, crabs, fish, fungi, lichen, birds, whales… This is a taxonomists playground. University researchers come to us when they need material for study (DNA, hair samples, or to make measurements from historic specimens) – just today I had a request for feather samples from White-crowned Sparrows. Museums also are as close to a time machine as you can get outside of science fiction. At museums you can hold an animal or plant collected 100 or more years ago. Nowhere else provides this sort of service to science and society.
But who cares? What is the point of discovering a new fish in BC waters? Or unravelling a species complex of beetles or stickleback? For me, the research generates knowledge for the simple pleasure of knowing stuff. I get a research paper published and maintain a publication record as a fish nerd – and that is enough for me. Do you benefit from scientific introspection (AKA navel-gazing)? I say yes.
People did well enough before the Victorian era, even though the present diversity of nature was far from understood. But ships in the 1700s and 1800s carried frenzied explorers to far corners of the world, in a grand age of biological stamp-collection. The race was on to catalogue nature and this continues even today. Without a proper understanding of species boundaries, all other branches of biological science and medical experimentation collapse. There is no sense comparing the effects of a drug on Norway Rats relative to a control group of un-medicated gerbils. It may seem simplistic to say that, but if we don’t understand species, we could be comparing quite different organisms rendering scientific results meaningless.
As a practical example of your reliance on science, try figure out what species of fish are in your local grocery store. Sure the labels says “sole”, but which one? Is that a local species? Or imported from Europe? Could one of them represent an endangered species? Are all the fish in the same tray from the same species? You may not be interested in a museum article on the identification of a single fish, or the RBCM’s contribution to DNA barcoding, but you may be ecologically conscious and want to make informed dietary decisions. Without proper identification, or if the basic taxonomy is wrong, we all flounder in ignorance. Even your grocery store choices depend on basic museum science.
Beyond practicalities on an individual level, does the museum as an organisation benefit from obscure research? Again yes. Active study maintains the institution’s reputation as research facility. It shows we are engaged, and that we continue to discover new things. The research helps build the collection – a biological library which generates even more scientific study. We stockpile museum specimens and information for the benefit of society. Museum researchers generate primary literature for academic discussion, but we also filter volumes of information to create concise content for public consumption. We bridge the gap between hard-nosed science and the hard-working public. A museum fails when it isolates collections and researchers from the public. What is the point of a museum collection if no one sees it, no one studies it, and the information is not shared.
 There is no point collecting specimens only to lock them away forever.
There is no point collecting specimens only to lock them away forever.
How can you keep current with our scientific advances and collection development? Our work is available globally in scientific journals. However, peer-reviewed science is mostly held in university libraries, and online access to scientific articles can be an expensive cure for insomnia. We also make our work available in a more casual format. You can visit in person and stroll through our galleries, take a collection tour, view one of our travelling exhibits, and we are also just a click away with blog articles, museum magazine articles, and web portal content. There are many ways the RBCM bridges the gap between pure academic research and the interests of every-day museum visitors. You don’t expect to learn about Pluto at the RBCM, but you do come here to learn about British Columbia and connect with our vibrant research community. We are irrelevant without you.
Clean Baleen post
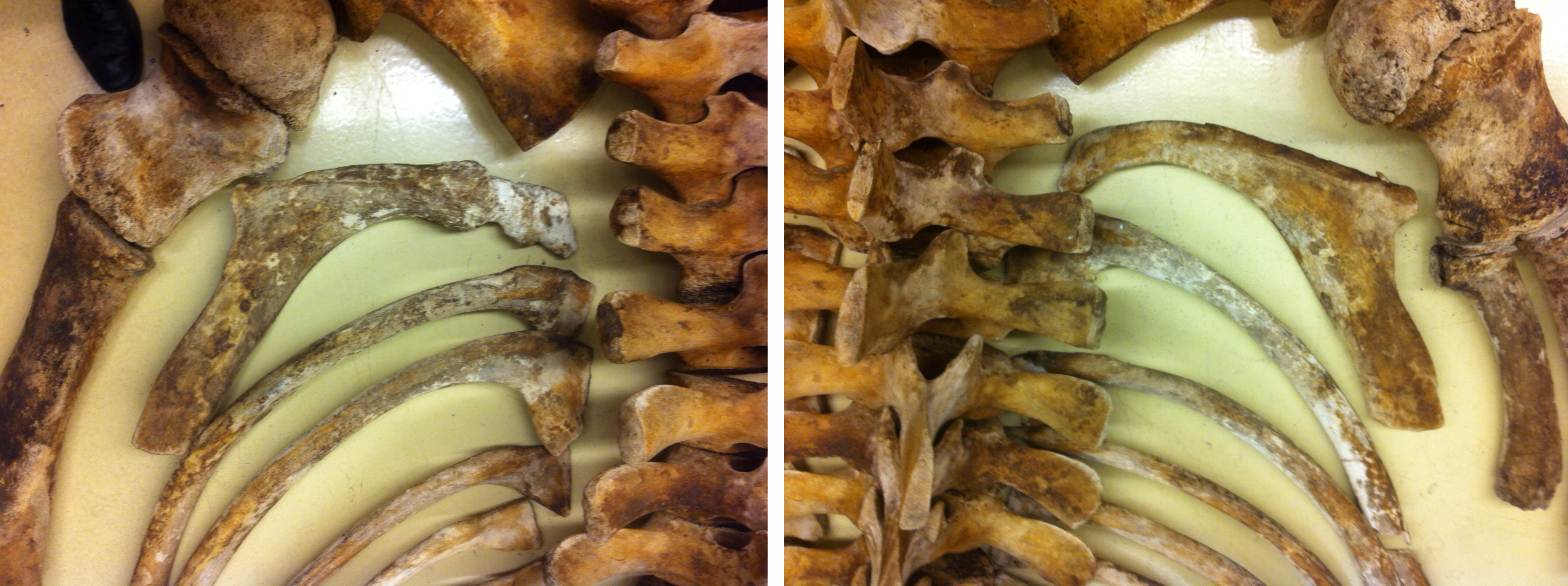
![Whale-005[1]](http://staff.royalbcmuseum.bc.ca/wp-content/uploads/2015/09/Whale-0051.jpg) On April 20th, 2015, a 9 meter Grey Whale washed up on Long Beach, Vancouver Island. On April 23rd, a small army of researchers and volunteers performed a necropsy and cleaned most of the meat off the bones. The bones are now buried and once clean, will be added to the Royal BC Museum’s research collection.
On April 20th, 2015, a 9 meter Grey Whale washed up on Long Beach, Vancouver Island. On April 23rd, a small army of researchers and volunteers performed a necropsy and cleaned most of the meat off the bones. The bones are now buried and once clean, will be added to the Royal BC Museum’s research collection.
One item that did not stay with the whale’s bones was its baleen. Baleen is essentially the same epidermal material as your fingernails and acts as a sieve to allow the whale to strain water from edible morsels like crustaceans. You can see the baleen in the mouth in both of these photos, although in the second photo, most baleen had already been removed. Several organisations wanted material for public display, and so I decided to experiment with a method of slow dehydration to preserve strips of baleen.
The baleen itself was removed right down to the gum-line along the jaw – and it pulled out very cleanly. I left cut sections of baleen at room temperature in a standard picnic cooler overnight. The next day I washed the sand and other debris out of the baleen and then immersed each piece in 95% ethanol. The pieces stayed in ethanol for 5 months (almost to the day), with the hope that the alcohol would draw out water from the remaining tissues.
To make a short story long: it worked.
 The baleen was removed from the ethanol and left to air dry over a few days. I had thought I would have to use heavy nails to nail each piece to a board to prevent the tissue from curling, but it proved unnecessary. The gum-line did curl slightly, but not enough to ruin the look of each piece.
The baleen was removed from the ethanol and left to air dry over a few days. I had thought I would have to use heavy nails to nail each piece to a board to prevent the tissue from curling, but it proved unnecessary. The gum-line did curl slightly, but not enough to ruin the look of each piece.
 In these three photos you can see the baleen in lingual view (the side facing the tongue), labial (the side facing the lip) and the underside of the gumline which shows fine slots and canals which housed the cells producing baleen tissue.
In these three photos you can see the baleen in lingual view (the side facing the tongue), labial (the side facing the lip) and the underside of the gumline which shows fine slots and canals which housed the cells producing baleen tissue.
Now the baleen chunks can be left at room temperature and over time the slight alcohol-fishy-smell should dissipate. Once permits are secured for each organisation, I’ll be able to ship the sections of baleen to local nature centers so that people can see first hand how fascinating baleen really is.
Parents and Mentors post
I am sure my parents just shook their heads when I started bringing home reptiles and amphibians, but they did not discourage me. That is what matters. They suffered the smells that wafted from the various cages, had to deal with escaped snakes, lizards and newts (newts always were found dried-out in mid-stride…), and took me to the hospital after a large python tried to kill me. But I don’t think they had any idea that my interest in animals would lead to a worthwhile career.
 Chasing snakes and frogs in the Interlake of Manitoba (Norris Lake).
Chasing snakes and frogs in the Interlake of Manitoba (Norris Lake).
In winter my Dad took me out to buy goldfish, and in summer, we’d go to nearby ponds to catch frogs to feed to my pet snakes. We’d also do our annual pilgrimage to the snake dens at Narcisse, in Manitoba’s interlake region. I was a lucky kid. My parents let me vanish into swamps with ice cream buckets to hunt frogs and snakes all day. I wonder what proportion of kids today are so free. Perhaps there is a game now where kids get to catch and keep frogs and snakes. Sim-Terrarium anyone?
 Granite Lake, west of Kenora, Ontario – one of many snakes I caught as a kid, along with Jamie Coyle and my brother Ian.
Granite Lake, west of Kenora, Ontario – one of many snakes I caught as a kid, along with Jamie Coyle and my brother Ian.
These memories came flooding back because my friend Lea also has started her son on a similar path. Lea has allowed her son to have a pet praying mantis and a chamaeleon… who knows what is next. My guess is a Bearded Dragon will be next or a Leopard Gecko. A few decades from now perhaps he will have my job here at the RBCM.
 Canada goose bones from the RBCM collection (RBCM 16187).
Canada goose bones from the RBCM collection (RBCM 16187).
Last week I helped a young lad identify a bone he had found. Obviously he had ideas about the bone’s identity, and had wondered whether it was a skull or not. But after a short visit to the Ornithology collection, I was able to show that the bone he found was the sternum from a Canada Goose. Then I explained where muscles attached that allowed the bird to flap its wings – and how to find the same muscles on the next chicken or turkey they roasted. Perhaps he will become a prominent comparative anatomist. And he too will thank his parents for helping foster an interest in nature.
 Parents – try not to freak out when your kid brings one of these home.
Parents – try not to freak out when your kid brings one of these home.
Perhaps I am to these kids, what Bill Preston was to me. Bill was the herpetologist at the Manitoba Museum, and as the author of the Amphibians and Reptiles of Manitoba, he made a great impression on me. Bill’s book (now very worn) kept me interested in amphibians and reptiles and I eventually replaced him at the Manitoba Museum as their Vertebrate Zoologist. If we spark the imaginations of kids and get them outside to build our next generation of naturalists, who knows where they will end up. I was thrilled to see so many kids at the RBCM’s two commemorative beach walks this summer – my youngest daughter included.
 Anna and I during the June 14th beach seine at Willows Beach (photo by Chris O’Connor, RBCM)
Anna and I during the June 14th beach seine at Willows Beach (photo by Chris O’Connor, RBCM)
We also hosted some amazing kids this year at our Gold Rush day camps. They certainly will remember the vats and jars of pickled specimens in my lab. These kids are the future scientists, politicians, lawyers, entrepreneurs, and regardless of which career they choose (we can’t all be lucky enough to work at a museum), kids need exposure to nature. It doesn’t matter if kids like lichen, liverworts, lepidoptery, or leopard frogs, our job as parents and mentors is to make sure today’s kids appreciate and value their connection to the earth.
Learning about soil (photo by my wife, Jeannette Bedard).
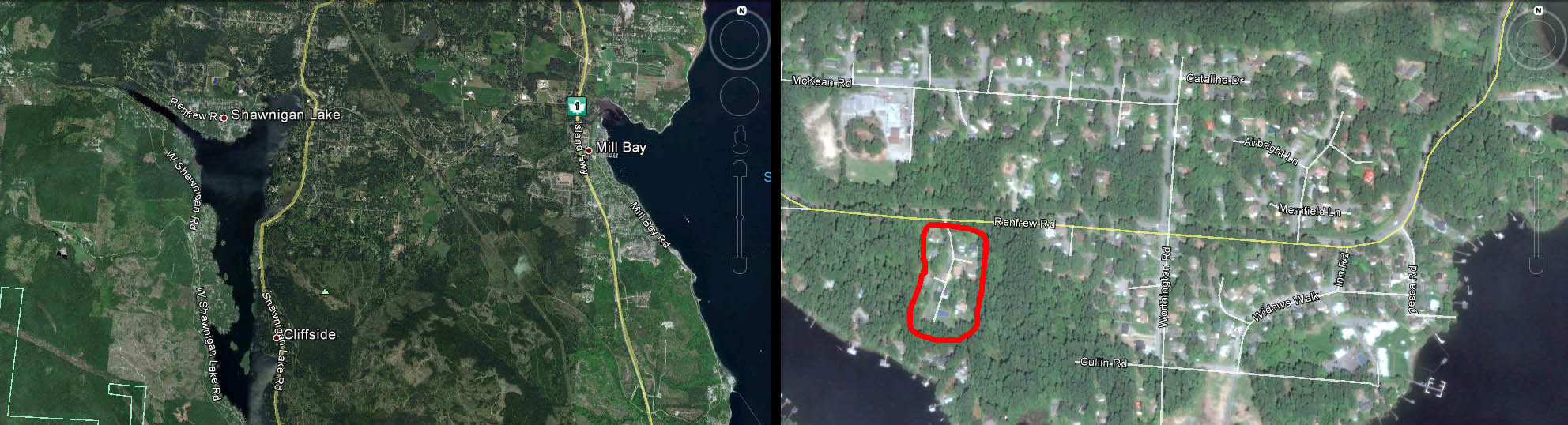
My favourite invasive species is still on the move. It is August 24th, 2015, and I just received photos confirming the presence of European Wall Lizards at Shawnigan Lake, British Columbia. A few weeks back, I was chatting with Rod Park, a cameraman with CHEK news. He said he had lizards at his house in Shawnigan Lake. The behaviour he described sounded suspiciously like that of the Wall Lizard. Alligator Lizards (our native lizard on Vancouver Island) are far more secretive and not so likely to be found in numbers in urban environments.
Today I received photos taken by Rod’s son Sterling, and even though the lizards did not cooperate – the photos are certainly good enough to show that they do indeed have European Wall Lizards. The lizards appeared about a year ago, and now sightings are a regular occurence. This is typical – one or two lizards reproduce, and the population explodes to 50 or more lizards in a few years. Some gardens on the Saanich Peninsula have hundreds of Wall Lizards – this species is highly invasive.
 Photo by Sterling Park, August 2015
Photo by Sterling Park, August 2015
Who knows how they made the jump to Shawnigan Lake – as eggs in a plant pot? Adult stow-aways? Intentional release? We may never know. This record certainly is a surprise – there are a few records from Mill Bay, but none as far west as Shawnigan Lake. Please be careful when moving hay, horse trailers, plants and soil – you never know what else you are carrying. Wall Lizards don’t need any more help – they have gone far enough.
 Photo by Sterling Park, August 2015
Photo by Sterling Park, August 2015
Anyone reading this in the Shawnigan Lake area — please email any sightings to me here at the RBCM so we can keep tabs on the rate of spread in your region. This summer we also had our first record in North Vancouver, and so I wonder where they are next to appear? This species could spread far down the Pacific coast and disturb native wildlife as it spreads. Wall Lizards are an unwelcome addition to our fauna and there seems to be no way to eliminate them from Vancouver Island now.
Photo by Sterling Park, August 2015
Since they are cannibalistic, I could imagine they’d have no problem eating (or at least killing) hatchling Sharp-tailed Snakes and Sharp-tail Snake eggs, or newborn garter snakes. They probably can eat newborn Northern Alligator Lizards, and every sunny day, Wall Lizards in BC must eat hundreds of thousands of insects and spiders. The unique place we call British Columbia is being hit hard by exotic species. You can make a difference. Help prevent the spread of these invasive lizards.
My contact info:
Gavin Hanke Curator, Vertebrate Zoology | Archives, Collections & Knowledge
675 Belleville Street, Victoria, BC Canada V8W 9W2
T 250 952-0479 | F 250 387-0534 (who sends faxes anymore?)
We rarely get a chance to sample freshwater fishes in BC, especially in the extreme north and rely on voucher specimens from other researchers to build our collection. One of the most rarely collected fishes is the Broad Whitefish (Coregonus nasus). Usually I talk about southern fishes invading northward, or exotic fishes released by people, but the Broad Whitefish is an example of an Arctic fish that just barely ranges into British Columbia, and we had six listed for the RBCM collection:
RBCM 000-00155-003; a single fish from off Canoe Creek mouth, Shuswap Lake, collected in 1979.
RBCM 979-11202-001; a single fish from Nisutlin Bay, Teslin Lake, Yukon, collected in 1979.
RBCM 979-11208-001; a single fish from just north of Teslin on east side of Teslin Lake, Yukon, collected in 1979.
RBCM 986-00295-001; two fish with no locality data, although they were collected the same day by the same collector as RBCM 990-00170-001.
RBCM 990-00170-001; a single fish from Kugmallit Bay, Beaufort Sea, collected in 1980.
Only one of our specimens was caught within British Columbia, and this single fish is from Shuswap Lake off the mouth of Canoe Creek. If you check Don McPhail’s magnum opus on Freshwater Fishes of British Columbia, there is no record of Broad Whitefish stocked in Shuswap Lake. Something so obvious as a Broad Whitefish in Shuswap Lake should have thrown up warning flags, but the specimen in the RBCM collection has sat since November 2000 and had not been re-examined. One of the jobs of a museum curator is to periodically look for probable mistakes in specimen identification, and correct identifications if possible.
Here is my Key to help identify whitefishes, and is a combination of identification keys by McPhail (2007), Wydoski and Whitney (2003), Nelson and Paetz (1992), Scott and Crossman (1973), McPhail and Lindsey (1970). The numbers in the key in brackets allow you to backtrack if you made a mistake.
Choice 1a – single flap of skin between front and rear openings of nostrils, snout narrow and somewhat pointed, gill rakers stout and short – go to 2
Choice 1b – two flaps of skin between front and rear openings of nostrils, snout width tapers gradually or is blunt, gill rakers elongate but may be shorter – go to 4
Choice 2a (1a) – small spots (parr marks) retained in adults, snout blunt and rounded, base of adipose fin less than eye diameter, 18 to 20 scales around the base of the tail, lateral line scales same size as adjacent body scales:
PYGMY WHITEFISH (Prosopium coulterii) – found in the Fraser, Columbia, Mackenzie, Yukon, North Coast, and Central Coast drainages. Because parr marks retained as adults, Pygmy Whitefish commonly mistaken for juveniles of Mountain Whitefish.
Choice 2b (1a) –lateral line scales are noticeably smaller than adjacent body scales, snout pointed when viewed from above – go to 3
Choice 3a (2b) – base of adipose fin less then the diameter of eye, snout pinched and slightly pointed, gill rakers short and stout, scales on flank with darker margin:
ROUND WHITEFISH (Prosopium cylindraceum) – found in the Mackenzie, Yukon, and North Coast drainages.
Choice 3b (2b) – adipose fin about 1.5 times the eye diameter, scales on flank evenly pigmented without darker margin, snout variable from ones with evenly tapered convex snouts to those with longer and thinner snouts offset from the general profile of the head:
MOUNTAIN WHITEFISH (Prosopium williamsoni) – found in the Fraser, Columbia, Mackenzie, North Coast, and Central Coast drainages.
Choice 4a (1b) – mouth extremely large and almost as wide as head, jaws with fine teeth, and the jaws appear squared when viewed from top, snout flattened and superficially pike-like, lower jaw extends beyond upper jaw, body cylindrical and elongate:
SHEEFISH (Stenodus leucichthys) – found in the Mackenzie and Yukon drainages.
Choice 4b (1b) – snout small and narrow and not as wide as head, lower jaw may project ahead of upper jaw, body deep and compressed – go to 5
Choice 5a (4b) – tip of snout flat to overhung – go to 6
Choice 5b (4b) – tip of snout pointed, lower jaw may protrude beyond upper jaw – go to 7
Choice 6a (5a) – tail fin shallowly forked, adipose fin nearly the same size as eye, upper jaw far longer than deep, head tapers back towards dorsal fin (deepest part of body at dorsal fin origin):
LAKE WHITEFISH (Coregonus clupeaformis) – found in the Fraser, Columbia, Mackenzie, Yukon, North Coast, and Central Coast drainages.
Choice 6b (5a) – tail fin deeply forked, adipose fin significantly larger than eye, upper jaw is as deep as it is long, body profile raises abruptly from head giving a hump-backed appearance (deepest part of body well-ahead of dorsal fin):
BROAD WHITEFISH (Coregonus nasus) – found in Teslin Lake, Yukon.
Choice 7a (5b) – upper jaw does not reach level with the leading edge of eye or only barely reaches eye level, pelvic fins positioned nearly level with the dorsal fin origin (i.e., the equivalent distance from the snout to the pelvic origin, measures back from the pelvic origin to the narrow part of the caudal peduncle ahead of the origin of the fulcral rays of the tail):
LEAST CISCO (Coregonus sardinella) – known only from Atlin, Teslin and Swan lakes in the Yukon drainage in British Columbia.
Choice 7b (5b) – upper jaw longer, reaching at least the level of the pupil, pelvic fin origin positioned noticeably behind the dorsal fin origin (i.e., the equivalent distance from the snout to the pelvic origin, measures back from the pelvic origin to a point behind the origin of the fulcral rays of the tail) – go to 8
Choice 8a (7b) – deepest part of body ahead of the dorsal fin origin (slightly hump-backed appearance), upper and lower jaws usually equal such that neither protrude, dorsal margin of upper jaw fairly straight, pelvic fin origin positioned mid-way along the length of the dorsal fin base:
ARCTIC CISCO (Coregonus autumnalis) – found in the Mackenzie (in British Columbia known only from a spawning run in the Liard River).
Choice 8b (7b) – deepest part of body at the dorsal fin origin (not hump-backed), upper jaw reaches back level with the pupil of eye, dorsal margin of upper jaw curved, pelvic fins positioned well-behind the dorsal fin origin:
LAKE CISCO (Coregonus artedi) – found in the Mackenzie River system in British Columbia.
NOTE: Lake Ciscoes are highly variable within and between river/lake systems. Within lakes, there may be bottom dwelling and open water populations that evolve distinctive body features and this complexity makes species identification difficult. Molecular data suggest that populations of Coregonus artedi evolved a normal, long-jawed form and a short-jawed form within many lakes. This short jawed form usually gets called Coregonus zenithicus because fisheries biologists considered the short jawed form a distinct species that dispersed from the Laurentian Great Lakes. Molecular evidence suggests that multiple independent origins of “zenithicus-like” short-jawed forms evolved across Canada from separate populations of typical Coregonus artedi. The lumping of all short-jawed forms from all lakes across Canada obscures true evolutionary relationships among whitefishes.
 RBCM 000-00155-003, from off the mouth of Canoe Creek, Shuswap Lake, collected June 12, 1979 (I was in grade 6). It was originally labeled as Salmo sp., and re-identified as Coregonus nasus in November, 2000. What do you think? Does it match the description or the drawing from Scott and Crossman (1973) (below)?? Even Step 1 in the key eliminates this fish as a Coregonus species – RBCM 000-00155-003 has one flap of skin separating the anterior and posterior nasal openings. Lateral line scales are a bit smaller than adjacent body scales, the snout is pointed when viewed from above, and the base of the adipose fin is significantly longer than the eye diameter. Oh well, problem solved. – it is a Mountain Whitefish (Prosopium williamsoni), from well-within the known range for that species.
RBCM 000-00155-003, from off the mouth of Canoe Creek, Shuswap Lake, collected June 12, 1979 (I was in grade 6). It was originally labeled as Salmo sp., and re-identified as Coregonus nasus in November, 2000. What do you think? Does it match the description or the drawing from Scott and Crossman (1973) (below)?? Even Step 1 in the key eliminates this fish as a Coregonus species – RBCM 000-00155-003 has one flap of skin separating the anterior and posterior nasal openings. Lateral line scales are a bit smaller than adjacent body scales, the snout is pointed when viewed from above, and the base of the adipose fin is significantly longer than the eye diameter. Oh well, problem solved. – it is a Mountain Whitefish (Prosopium williamsoni), from well-within the known range for that species.
 Drawing of a Broad Whitefish (C. nasus) from Scott and Crossman (1973).
Drawing of a Broad Whitefish (C. nasus) from Scott and Crossman (1973).
Broad Whitefish have a deep compressed body, and the body profile raises abruptly behind head giving a hump-backed shape (weight forward – in flyfishing terms). This species also is noted for its short, deep upper jaw bone, and short, blunt snout which overhangs the mouth. The head is small relative to the body. The lateral line is straight and the body is covered with large cycloid scales. Broad Whitefish are dusky to nearly black on the back, which blends to silvery sides and a pale yellow-white belly. The head and gill covers may have fine dusky freckles. Fins are dusky to black in adults, the pectoral and anal fins may have blue to purple iridescence, and fins of young individuals are pale and translucent. Young Broad Whitefish also lack spots (parr marks), unlike young Mountain Whitefish which have spotted bodies like juvenile salmon. Broad Whitefish grow to 86 cm.
The diet of Broad Whitefish in Teslin Lake shows they are bottom-feeders. They eat caddisfly larvae, midge larvae and pupae, waterboatmen, snails and fingernail clams, amphiopds, and zooplankton. Larval fish and fish eggs may also be eaten. Young Broad Whitefish take copepods, cladocera and midge larvae, and graduate to larger items later in the first summer.
Globally, Broad Whitefish are found from the Baltic Sea, and Norwegian Sea, east across Russian Arctic to the Kuskowim River, the Yukon River system in Alaska, and other coastal streams and lakes draining into the Bering Sea, Chukchi Sea, and Beaufort Sea. They are also known from the Mackenzie River and Perry River drainages in Nunavut. In BC, Broad Whitefish are restricted to Teslin Lake and its tributaries in the Yukon River drainage. This represents the southern-most limit of the species’ natural range in North America.
 Green dots indicate localities listed by McPhail 2007; the red dot indicates the location where RBCM 000-00155-003 was collected in Shuswap Lake.
Green dots indicate localities listed by McPhail 2007; the red dot indicates the location where RBCM 000-00155-003 was collected in Shuswap Lake.
Broad Whitefish are said to be common in rivers rather than lakes, but also are found in estuaries, rarely venturing far along Arctic coastal areas. Broad Whitefish are tolerant of murky water, but also will be found in clear lakes or streams, and as their hunched-over body shape suggests, the species is oriented towards the river or lake bottom rather than the surface or open water. Broad Whitefish are known to make extensive migrations, but the population in BC is lacustrine and presumably, movements are restricted to spawning runs, movements for feeding, and perhaps retreat to deeper water to overwinter. Fish in Teslin Lake are caught in less than 10 meters depth in summer, and this probably reflects feeding habitat during the productive summer season. Yearling Broad Whitefish also occupy shallow water, as do young-of-the-year, and move to the shelter of any structure or headlands when waves pound the shoreline, and to deeper water in winter.
Broad Whitefish move from lakes to tributary streams to spawn in October to November, but this behavior may begin as early as July to August in the far north. They probably spawn under ice cover when water is near 0°C. Broad Whitefish may spawn in streams which drain from a lake, or they may head upstream in tributaries which feed into a lake to find suitable gravel substrate for egg deposition. We do not know the exact nature of Broad Whitefish spawning in Teslin Lake. Females can carry up to 51,000 eggs, and in the Arctic Red River, females can outnumber males by 15 to one, and they spawn at night. Like other whitefish, Broad Whitefish scatter their eggs over the substrate and abandon their offspring. Because of energetic constraints in the north, Broad Whitefish may spawn every other year, although a good proportion will spawn in consecutive years. Shortly after spawning, adults move back to their resident lake (or downstream to another lake) for winter refuge. Some Broad Whitefish use the same over-wintering areas in consecutive years. Eggs have been incubated for 59 days at 4°C, and hatched as temperatures were elevated to 7°C; this temperature increase probably simulated spring’s slightly warmer runoff to the developing embryos. In the wild, eggs probably hatch around ice breakup in spring. Broad Whitefish are known to live at least 35 years.
 For comparison, here’s the head of a real Broad Whitefish from the Beaufort Sea (RBCM 990-00170-001). The snout profile and the shape of the upper jaw (maxilla) are significantly different – and yes – just to be picky, I checked and RBCM 990-00170-001 has two flaps separating the anterior and posterior nasal opening.
For comparison, here’s the head of a real Broad Whitefish from the Beaufort Sea (RBCM 990-00170-001). The snout profile and the shape of the upper jaw (maxilla) are significantly different – and yes – just to be picky, I checked and RBCM 990-00170-001 has two flaps separating the anterior and posterior nasal opening.
While whitefishes in Canada have been studied intensively, there is still much to learn. There probably are new species to discover (keep your eye on Coregonus zenithicus (Shortjaw Cisco) – it may include several cryptic species). Perhaps with concerns over climate change and with increased attention to northern ecosystems, Canadian whitefish will get the attention they deserve. The single fish identified as a Broad Whitefish from Shuswap Lake, far south of the native range of Broad Whitefish, represents a misidentified Mountain Whitefish (Prosopium williamsoni). In BC, Broad Whitefishes are restricted to Teslin Lake and its tributaries. I think I should bug my friend Kim Howland, she likes whitefish, and suggest she spend some time at Teslin Lake to get more specimens for the museum collection. The RBCM officially has no Broad Whitefish from BC.
Sources:
Howland et al. 2009, McPhail 2007, Wydoski and Whitney 2003, Mecklenburg et al. 2002, Maitland 2000, Nelson and Paetz 1992, Wheeler 1978, Scott and Crossman 1973, McPhail & Lindsey 1970, Carl et al. 1967.
Think Skinks post
Everyone loves a mystery. Some chase the impossible – like Cadborosaurus – the mythical mega-sea-serpent described by Bousfield and LeBlond. I prefer to look for long-shots that are possible, like Pygmy Short-horned Lizards and Western Fence Lizards in the Okanagan, or the Western Skink on Vancouver Island and some of our adjacent islands. You may think it is nuts, and a waste of time to look for things that probably are not here. But anecdotes from this last year have got me thinking… or is it dreaming… of lizards with bright blue tails here in my back yard.
A colleague of mine, Purnima Govindarajulu (Ministry of Environment) received a report of a blue-tailed lizard from Denman Island. I heard of a blue-tailed lizard near the junction of Wallace Road and West Saanich Road here on Vancouver Island. Another interesting email from the Brentwood Bay area came in this last week of June which stated,
“The wall lizards appeared years ago. I use to find blue tail skinks around – I know they are still on Saltspring island – I spent years there as a kid and use to see them often. They were occasionally on my parents property, but maybe have been pushed out by the wall lizards.”
Yes these are anecdotes with no accompanying photographs, but they are worth investigating. Why? Martin McNicholl, who was at the University of Alberta at the time, published a sight record from 1975 of a skink with a blue tail from near Courtenay. This 1975 record pre-dates the explosion of European Wall Lizards on Vancouver Island. However, the Courtenay record is considered questionable in the 2002 COSEWIC report by Kristiina Ovaska and Christian Engelstoft, as well as by Allan St. John in his book on reptiles of the northwest. In other words, conventional wisdom says that in BC, Western Skinks exist in the dry interior from the Princeton area east to the area around Creston, and nowhere near the coast. But people are reporting lizards with bright blue tails here on Vancouver Island.
While wall lizards are becoming super abundant on Vancouver Island and spreading to some of the other nearby islands, Western Skinks in BC receive comparatively little attention. McNicholl’s report of a skink near Courtenay all but faded into academic obscurity – and I have to thank Pat Gregory (the herpetology professor at UVIC) for reminding me of McNicholl’s article.
If skinks are not here, then what are these blue-tailed lizards people are talking about? Super colourful wall lizards? Lacertids from some areas of Europe are extremely colourful – some are bright blue. Do we have more than one species of lacertid loose here in southwestern BC?
Here is where I have to ask for help – citizen science is the best way to search for the notorious “needle in a haystack”. I have two eyes, collectively we have thousands. If you are out on a walk – especially on Saltspring Island, sitting in a park, or in your back yard and you see a lizard with a blue tail, please take photos. Even smart phone photos like this one I took at Borden mercantile are sufficient to identify what species you have found.
My first evidence of Wall Lizards at Borden Mercantile – an iphone photo taken moments before I stepped on a rusty nail. No one ever said science was easy.
This earlier blog article has a key to the species of lizards found here in BC – you can select features in the key that can guide you to a correct identification. If you find a Western Skink here on the Vancouver Island or one of the Gulf Islands, or any other unusual lizard, then please get as detailed a set of photos as you can, and forward them to me at the Royal BC Museum for verification.
Additional reading:
Matsuda B.M., D.M. Green, and P.T. Gregory. 2006. Amphibians and Reptiles of British Columbia. Royal BC Museum Handbook Series, Victoria, British Columbia. 266 p.
McNicholl, M.K. 1975. Sight record of a Western Skink on Vancouver Island. The Canadian Field-naturalist, 89: 79-80.
Ovaska, K.E., and C. Engelstoft. 2002. COSEWIC status report on the western skink Eumeces skiltonianus in Canada. Committee on the Endangered Status of Wildlife in Canada. 19 p.
St. John, A. 2002. Reptiles of the Northwest, British Columbia to California. Lone Pine Press, Edmonton, Alberta. 272 p.
This summer one of our business records projects is the processing, arrangement and description of the Okanagan Helicopters fonds. These interesting records are being worked on by contract archivist Taryn Jones and UBC student and archivist-in-training Eric Walerko.
Taryn and Eric conferring on Okanagan Helicopter files
Okanagan Helicopters was founded in 1947 in Kelowna as Okanagan Air Services. It provided fixed-wing crop spraying and charter services but soon switched from fixed-wing aircraft to helicopters. As the helicopter fleet grew, so did the services offered by Okanagan Helicopters. They ran a helicopter training school in Penticton and became heavily involved in resource extraction and industrial work including timber cruising, forest seeding, geological survey flights, power line construction and on and off-shore mineral exploration support. In the 1960’s and 70’s the company expanded its services worldwide, especially in the areas of resource extraction support. In 1987 Okanagan Helicopters was purchased by Canadian Holding Company and became CHC Helicopter Corporation.
One of the photographs from the fonds showing an Okanagan helicopter assisting in mountain side timber work
Taryn comparing photographs
In 1986 the BC Archives acquired some film and video records directly from Okanagan Helicopters. The bulk of the records were acquired by the Archives through the Royal British Columbia Museum in 1991.
Taryn sorting through a box of film
Eric protects the photographs by placing them in acid free envelopes
Once the archivists have finished their work, we will be able to provide access to these records in our reference room. The fonds description number is PR-1842.
Okanagan Helicopter’s logo was the hummingbird. The logo continues today in a more stylized form.
Eagle cam post
One of the many interesting ways to spend time on the internet is to view wildlife webcams. You can probably guess how old I am by that statement. Eagle webcams are popular and our fascination with looking at eagles is nothing new.
In Atlin the Muirhead brothers went to great lengths to take various Eagle photographs. These photos were taken between 1901 and 1903.
Photographing an eagles nest under difficulties
This photo shows one of the brothers up on the cliff face standing over a nest.
Baby Eagles
Golden Eagles raised from the nest
Taking babies from the nest is not recommended.
All photos from PR-0507 (198205-002) container 000736-0001 Muirhead Brothers Photographers fonds.
Miners in Atlin post
The lure of gold brought many miners to Atlin. At the BC Archives we have several metres of government records relating to mining activity. There are mining claims, land records and even claim disputes in the Bennett-Atlin Commission records. But there’s something special about photographs. When we look at a photograph of a miner in action, we may not know any technical details about mining but we can put ourselves in the picture. We can look at the clothes, the tools, the tents and imagine the hard work and the difficult conditions.
We can look at a photo of a loaded sled and wonder about the geography and the weather and wonder what it was like to have to haul everything needed in, and how we would get our products out.
“Black terrier bench claim on Spruce Creek, Cornelius Bell, owner”, M. Bro’s. no 117 1901
Loaded sled
All photos from PR-0507 (198205-002) container 000736-0001 Muirhead Brothers Photographers fonds.
When I moved to the BC coast it was a big enough thrill to see whales in the distance, with a spray of air and water and a glimpse of their backs as they surfaced. I have seen several Humpbacks, Killer Whales, and a few Grey Whales while out on research trips, and had a close shave with a Humpback while sailing, but the week of April 20th takes the cake.
 On Monday April the 20th, a 10 m yearling female Grey Whale washed up on the beach, near the Kwisitis Visitor Center. A crew was hurriedly assembled to examine the animal and remove it before the tide came in and before predators scavenged the carcass. The question was – Dump it all at sea, or save the skeleton? The decision was made to clean the skeleton and prepare it for the RBCM’s collection – so that changed things a bit. The disarticulation of the skeleton had to be done carefully – but with the same time constraints – we wanted the whale off the beach by end of Thursday April 23rd.
On Monday April the 20th, a 10 m yearling female Grey Whale washed up on the beach, near the Kwisitis Visitor Center. A crew was hurriedly assembled to examine the animal and remove it before the tide came in and before predators scavenged the carcass. The question was – Dump it all at sea, or save the skeleton? The decision was made to clean the skeleton and prepare it for the RBCM’s collection – so that changed things a bit. The disarticulation of the skeleton had to be done carefully – but with the same time constraints – we wanted the whale off the beach by end of Thursday April 23rd.
 We had people from Parks Canada, Fisheries and Oceans Canada, Cetacea Contracting, Strawberry Isle Marine Research Society, the Ucluelet Aquarium, the Vancouver Aquarium, and numerous whale experts and dedicated volunteers to help dissect the whale. We assembled by 08:30, and after a short set of introductions and an orientation, we discussed safety, and then suited-up and got to work.
We had people from Parks Canada, Fisheries and Oceans Canada, Cetacea Contracting, Strawberry Isle Marine Research Society, the Ucluelet Aquarium, the Vancouver Aquarium, and numerous whale experts and dedicated volunteers to help dissect the whale. We assembled by 08:30, and after a short set of introductions and an orientation, we discussed safety, and then suited-up and got to work.
 Between 9:00AM and 6:30PM on April 23rd, we reduced the whale to its skeleton (mostly) and Lisa Spaven, of the Cetacean Research Program, Fisheries and Oceans Canada, did a necropsy to try determine the cause of death. The short answer? The whale looked pretty normal with no obvious signs of trauma.
Between 9:00AM and 6:30PM on April 23rd, we reduced the whale to its skeleton (mostly) and Lisa Spaven, of the Cetacean Research Program, Fisheries and Oceans Canada, did a necropsy to try determine the cause of death. The short answer? The whale looked pretty normal with no obvious signs of trauma.
 It is quite the sight to reach the top of the beach ridge, and see the whale only a few meters away. The first step – remove the blubber from the left side of the animal (that was the side facing us). This was when everyone was most nervous. No one wanted to be the first to cut too deep and cause an explosive release of gas from the animal’s body cavity. This animal had only been dead a few days, so there was no risk of it going into low-orbit, but there was a fair bit of gas which had to be released. Eventually a high pitched squeal, followed by scattering volunteers, meant that someone had cut a bit deep. Those downwind raised their arms to their noses in a vain attempt to exclude the effluvia. I was amazed how many onlookers stayed downwind (north of the whale) – it was far fresher on any other compass heading.
It is quite the sight to reach the top of the beach ridge, and see the whale only a few meters away. The first step – remove the blubber from the left side of the animal (that was the side facing us). This was when everyone was most nervous. No one wanted to be the first to cut too deep and cause an explosive release of gas from the animal’s body cavity. This animal had only been dead a few days, so there was no risk of it going into low-orbit, but there was a fair bit of gas which had to be released. Eventually a high pitched squeal, followed by scattering volunteers, meant that someone had cut a bit deep. Those downwind raised their arms to their noses in a vain attempt to exclude the effluvia. I was amazed how many onlookers stayed downwind (north of the whale) – it was far fresher on any other compass heading.
 With the majority of the gas released, Lisa continued with her necropsy, which meant she examined the organs for any unusual colours, textures, or any sign of impacts, hemorrhages, etc. Tissue samples taken during the necropsy have been sent away for microscopic examination – we’ll see whether they show any other evidence for the cause of death. As Lisa said in a recent email, “The case is not closed on this animal just yet.”
With the majority of the gas released, Lisa continued with her necropsy, which meant she examined the organs for any unusual colours, textures, or any sign of impacts, hemorrhages, etc. Tissue samples taken during the necropsy have been sent away for microscopic examination – we’ll see whether they show any other evidence for the cause of death. As Lisa said in a recent email, “The case is not closed on this animal just yet.”
 While she did her work, we set about removing as much muscle and fat as we could, and disarticulated the skeleton. I got the job of separating the left pectoral flipper at the shoulder socket. This whale is a quantum leap from the cats and mink used in undergraduate anatomy labs. Luckily, vertebrates have a fairly conservative body plan and the anatomy makes sense.
While she did her work, we set about removing as much muscle and fat as we could, and disarticulated the skeleton. I got the job of separating the left pectoral flipper at the shoulder socket. This whale is a quantum leap from the cats and mink used in undergraduate anatomy labs. Luckily, vertebrates have a fairly conservative body plan and the anatomy makes sense.
Once we got the pectoral flipper and shoulder blade off, I was tasked with the location and removal of the pelvic girdle. Funny thing was: this was my first whale dissection, and someone (laughably) thought I was “the brains” of the operation. Yes I am still laughing at that. The pelvic girdle is reduced to a splint of bone – roughly banana shaped and not far-off the size of a banana on this small whale. You have to remove all the fat from around the animal’s anus, and then press on the muscles in that region. The bones are buried, but you can feel them relative to the softer muscle tissue on the flank. I am guessing the search for pelvic bones is more difficult in larger whales. Once you find the pelvic bones, a sharp knife makes short work of their removal. Ribs also pop off easily once you get the hang of it.
 It is amazing how fast knives lost their edge, and I have to thank Ken Parkinson – my step-Dad for his skills with a whetstone. He lost count of how many knives he sharpened and re-sharpened that day. When Ken volunteered to come along that day, I wasn’t sure what we’d get him to do (he took the photos I have used in this post). But everyone was glad he found his niche in the work party.
It is amazing how fast knives lost their edge, and I have to thank Ken Parkinson – my step-Dad for his skills with a whetstone. He lost count of how many knives he sharpened and re-sharpened that day. When Ken volunteered to come along that day, I wasn’t sure what we’d get him to do (he took the photos I have used in this post). But everyone was glad he found his niche in the work party.
 Large chunks of meat and fat were easy to drop into the tractor’s shovel for disposal, but the intestines were another matter. I think five of us wrestled with the intestines to get them into the tractor – and then we hoped that they stayed in place en-route to the dump truck. The tractor driver shared our sentiment. The discarded fat, organs and muscle were loaded into a boat and then dumped at sea and recycled nature’s way. I am sure there were happy hagfish and sharks that day – and a few days after.
Large chunks of meat and fat were easy to drop into the tractor’s shovel for disposal, but the intestines were another matter. I think five of us wrestled with the intestines to get them into the tractor – and then we hoped that they stayed in place en-route to the dump truck. The tractor driver shared our sentiment. The discarded fat, organs and muscle were loaded into a boat and then dumped at sea and recycled nature’s way. I am sure there were happy hagfish and sharks that day – and a few days after.
 Five people worked on the head – there was a lot of material to remove – including the baleen – which we saved. To be honest, I was so busy, entire organ systems were removed and I never saw them. I never did see the tongue get removed. It was gone by the time we separated the head from the vertebrae.
Five people worked on the head – there was a lot of material to remove – including the baleen – which we saved. To be honest, I was so busy, entire organ systems were removed and I never saw them. I never did see the tongue get removed. It was gone by the time we separated the head from the vertebrae.
It was a non-stop process through waves of rain and strong wind, and long periods of warm sunshine. I am not sure which was worse. The rain and wind was uncomfortable, but we were wrapped up anyway to avoid getting any mess on our clothes. Rain wasn’t an issue – although we did ask for help from people with clean hands to lift our raincoat hoods. The sun also made things uncomfortable – we were wrapped in rain gear, boots, and gloves – all duct-taped together. In the sunshine, our best friends were the volunteers with clean hands that opened juice boxes and handed us cups of water. Thank you very much. All I ate during the dissection was one granola bar – Ken carefully unwrapped it and fed it to me. Don’t get me wrong, there was plenty of food provided by Parks Canada, but I didn’t want to stop. I can go three days without eating – so it was no problem.
 By the end of the day we had the animal into moveable pieces and loaded into a pickup truck. The sand made it easier to grip the slippery remains. The bones now are buried and will decay in a mixture of sand and soil for about a year. Mike DeRoos of Cetacea, suggested we let this skeleton sit for a year and let it decay slower and at a cooler temperature, just in case the bones were not solidly ossified. If the bones are heated too much during the decay process, they will become brittle.
By the end of the day we had the animal into moveable pieces and loaded into a pickup truck. The sand made it easier to grip the slippery remains. The bones now are buried and will decay in a mixture of sand and soil for about a year. Mike DeRoos of Cetacea, suggested we let this skeleton sit for a year and let it decay slower and at a cooler temperature, just in case the bones were not solidly ossified. If the bones are heated too much during the decay process, they will become brittle.
I am hoping to have this whale on display at the Royal BC Museum in the future – it will depend on how nicely the bones turn out at the end of the process. As far as research is concerned, it will be fine as a representative of the species, but for display, we want it to be perfect. Stay tuned.
Once we were done, our raingear was power-washed. Then I ate a few cookies and had some coffee, and Ken and I drove to Parksville. My Mom had dinner ready for us (you guessed it, stew). Then after dinner I drove back to Victoria – arriving home at 11:30 PM. That was a long day, but at least the Crosstrek I rented was fun to drive, and that kept me awake.
Someone on the team mentioned that volunteers commonly help with one whale – then never return. So I’ll end this with a quote from a popular movie personality.
The Muirhead brothers, Charles H. and Lewis P., were born in Scotland in the 1870’s. By 1893 they had arrived in North America. They took up photography and established their partnership, called Muirhead Bros., in 1896 in Sidney BC.
By this time the Klondike gold rush had started. It spread to Atlin in 1898 and shortly thereafter, the Muirheads followed the money. Between 1901 and 1903 they took a lot of photos of mining activities in the area and also documented the non-mining people and activities.
At the BC Archives, we have two Muirhead Bros. photo albums with the title “Views of Atlin, BC”. They were probably created as a commercial souvenir and many of the mounted photographs within have dates and captions. These albums have been described as PR-0507 and can be viewed in the Archives reference room. Some loose copies of prints came in with the albums and I have chosen some to highlight.
Today the theme is gold nuggets!
This photo is called “All from Willow Creek” and is dated 1901.
PR-0507 (198205-002) container 000736-0001 Muirhead Brothers Photographers fonds.
Recent range records for British Columbia’s marine fishes were based on deep-water surveys, from fisheries observers and the commercial fishery, but it is unlikely that these fishes represent northward movement due to warming climate. Deep-water fishes were captured in samples ranging deeper than 1000 m where temperatures remain cold.
In contrast, the RBCM just received two warm-water fishes – a Finescale Triggerfish (Balistes polylepis) and Louvar (Luvarus imperialis). These two species are known to range north in the eastern North Pacific Ocean during El Nino events and other warm periods, with the Triggerfish straying as far north as Metlakatla, Alaska. My predecessor, Alex Peden found Louvar in Washington, just short of the Canadian Exclusive Economic Zone and suggested that Louvar should stray into Canadian territory. Neither species has been found in BC until now, even though Brian Coad of the Canadian Museum of Nature suggested Louvar are known from British Columbia.
 Finescale Triggerfish, Balistes polylepis, 26.3 cm Standard length (SL), scale bar = 10 cm, with 1 cm increments.
Finescale Triggerfish, Balistes polylepis, 26.3 cm Standard length (SL), scale bar = 10 cm, with 1 cm increments.
In 2013, Eastern North Pacific surface water started warming and since then, conditions remain significantly warmer during seasonal cycles than during the last few decades according to Richard Dewey, Oceans Network Canada. By autumn 2014 this warm surface water shifted eastward, and into 2015, the entire west coast of North America remained unusually warm. While surface waters of the eastern North Pacific warm when coastal upwelling is weak or delayed, storm-mixing of surface waters is reduced, and the California Current weakens during El Niño events, Richard Dewey suggests this 2013-2015 thermal anomaly along the North American coast represents a new pattern, not related to El Niño/La Niña cycles. Temperature increases like those observed since 2013 along the North American Pacific coast should allow southern species to move north either through simple dispersal or changed larval survival and recruitment.
 Louvar, Louvaris imperialis, 72.0 cm SL, scale bar = 15 cm, with 1 cm increments.
Louvar, Louvaris imperialis, 72.0 cm SL, scale bar = 15 cm, with 1 cm increments.
As a result of this warming water, we now have specimens of Finescale Triggerfish and Louvar in British Columbia, with both found in the autumn of 2014. They now leave me wondering what’s next? I am hoping for Angel Sharks and Leopard Sharks.
Additional Reading:
Coad BW, H. Waszczuk H, Labignan I. 1995. Encyclopedia of Canadian fishes. Canadian Museum of Nature, Canadian Sportfishing Productions Inc., Waterdown, Ontario. 928 p.
Dewey R. 2015. Warm North East Pacific Ocean Conditions Continue into 2015. Ocean Networks Canada e-Newsletter. <http://www.oceannetworks.ca/warm-north-east-pacific-ocean-conditions-continue-2015>
Hanke GF, Roias SM. 2012. First specimens of the marine eels Venefica ocella and V. tentaculata (Nettastomatidae) from British Columbia. Canadian Field-Naturalist 126:210–216.
Hanke GF, Gillespie G, Fong K, Boutillier J, Peden AE, Bedard JM. 2014. New records of spiny eels (Albuliformes), true eels (Anguilliformes), and bobtail eels (Saccopharyngiformes) in British Columbia, Canada. Northwestern Naturalist 95:67–76.
Hart JL. 1973. Pacific fishes of Canada. Fisheries Research Board of Canada Bulletin No. 80. 740 p.
Lamb A, Edgell P. 2010. Coastal Fishes of the Pacific Northwest. Harbour Publishing Co. Ltd., Madeira Park, B.C. 335 p.
Love MS. 2011. Certainly more than you want to know about the fishes of the Pacific coast: a postmodern experience. Really Big Press, Santa Barbara. 645 p.
Love MS, Mecklenburg CW, Mecklenburg TA, Thorsteinson LK. 2005. Resource Inventory of Marine and Estuarine Fishes of the West Coast and Alaska: A Checklist of North Pacific and Arctic Ocean Species from Baja California to the Alaska–Yukon Border. Seattle, WA: US Department of the Interior, US Geological Survey, Biological Resources Division, OCS Study MMS 2005-030 and USGS/NBII 2005-001. 288 p.
Mecklenburg CW, Mecklenburg TA, Thorsteinson LK. 2002. Fishes of Alaska. American Fisheries Society Publication, Bethesda, MD. 1116 p.
Peden AE, Jamieson GS. 1988. New distributional records of marine fishes off Washington, British Columbia and Alaska. Canadian Field-Naturalist 102:491–494.
…and the infamous post
Most British Columbians have heard of Bill Miner the train robber, but how about his accomplice Shorty Dunn?
Dunn’s real name was John William Grell and he was known as Billy Grell, Billy Dunn and Shorty Dunn.
George Winkler was friends with Shorty when they both lived in the Princeton area and while Shorty was in the BC Penitentiary, from 1906 to 1918, he and Winkler wrote to each other regularly. Winkler made several efforts over the years to have Shorty released on parole. He wrote to Ottawa, to the Warden at the penitentiary and to mutual friends to try to raise interest in Shorty’s release. When Shorty was released on parole in 1918, he and Winkler continued to correspond and in 1927 Winkler was notified of Shorty’s death by drowning.
Winkler kept Shorty’s letters in several parcels, along with cartoons and drawings that Shorty sent him. He also kept clippings in his “book of crimes” scrapbook on Bill Miner and Shorty. In 1923 he was interviewed by the Victoria Colonist and he provided the letters and drawings to present Shorty’s story.
I found it very moving to open the parcel of letters and to think that I may be the first person to read them since 1923. Shorty was a good writer and it’s clear that he suffered terribly during his 12 years in prison.
Page from “Book of Crimes” container 891390-0001, folder 16
The following letters all from container 891390-0001, folder 15
Draft of letter from GEW to Shorty Dunn, December, 1908
Letter from Warden Brown to GEW, re chance of appeal, 1909
Letter from Ottawa to GEW, re Shorty’s health, 1912
Letter from Shorty to GEW, with accompanying cartoon, July 1914
If you want to read the whole story about Bill Miner, Shorty and the famous train robbery, try Peter Grauer’s book “Interred with their bones”.
George Winkler lived in Victoria on and off throughout his life and often maintained an office here to run his various mining concerns.
“Sayward Bldg. room 412, Neil McGillivray on left” Winkler on right. In container 000491-0002, folder 4
He had many interests beyond mining. He was a socialist and ran in Comox for a provincial seat in the 1916 election. He was a published poet who often had his poems printed in the local newspaper. Here is a 1941 poem dedicated to John Dean on his birthday. Winkler kept a copy in his scrapbook and noted the printing errors.
Poem to John Dean 1941. In container 891390-0001, folder 15
He was also on friendly terms with Nellie McClung. Winkler’s people lived in Treherne in Manitoba and knew the McClung family. Here is a letter from 1935 when Nellie and Winkler were both living in Victoria. It sounds as though she had recently been in contact with Winkler’s sister Grace.
Letter from Nellie McClung, June 1935. In container 891390-0003, folder 22
He was also interested in bringing together art and scientific knowledge. In 1939 Winkler wrote to Walt Disney Productions to complement them on the release of “Snow White” and to suggest a new idea for a movie. This reply from Jane Clark at Walt Disney Productions shows the beautiful stationary the company was using at the time.
Letter from Disney, February 1939. In container 891390-0003, folder 22
Winkler the jailbird post
There is an interesting print of George Winkler among his photographs in accession 197908-016. This particular photograph was taken by Miss M. [Mary] Spencer of Kamloops in 1906 and shows Winkler (back left) and three other men. What were they commemorating?
There are two copies of this print. Winkler wrote “Jail Birds 1907” [sic] on one, and “taken at Kamloops on release from gaol” on the other. It is usual to have a formal portrait taken after release from jail? What were they in for?
Taken at Kamloops on release from gaol. In container 000491-0001, folder 16
The explanation for the photograph showed up in Winkler’s files in accession 89-1390. Winkler kept a scrapbook which he called his “book of crimes”. Here he pasted in newspaper clippings covering his activities in March and April 1906. While living in Penticton, Winkler and a group of local men took it upon themselves to expel 10 Chinese workers from town. It was a labour issue; W.T. Shatford, local M.P.P. and managing director of the Southern Okanagan Land Company had brought in the Chinese workers. Winkler, a socialist, took exception to importing foreign workers to work locally at a lower rate. Along with a group of Penticton residents, he escorted the Chinese men to the lake steamer and paid their boat tickets out of town. A police constable was apparently there but was unable to convince the residents to desist. The ringleaders were arrested and a magistrate led trial was held. They were offered the choice of a $25 fine or 30 days in the Kamloops jail.
Two of the men charged paid the fine and in fact the residents had a whip round to raise the funds to cover all the fines; however Winkler and three of his cohorts choice to go to jail to make their point.
When they got out of jail in Kamloops, they had their portrait taken to commemorate the event.
Book of Crimes. In container 891390-0001 folder 16
Incidentally, a month after she took the photograph of George Winkler and his friends, Mary Spencer photographed the more famous train robbers Bill Miner, Shorty Dunn and Louis Colquhoun.
The Kamloops Museum and Archives are currently showing an exhibit of Mary Spencer’s photographs from now until June, see http://www.kamloops.ca/museum/index.shtml
Slim Jim Unmasked post
Last week I was treated to a day of paintings conservation. I’m actually trained as an objects conservator, which means that I am qualified to work on almost anything EXCEPT paintings. In truth, there are many things that I have little to no expertise conserving, including architecture, paper and photographs, digital media, and so on. I have, however, worked on a variety of strange and wonderful objects ranging from shipwreck nails to stone sculpture to broken natural history specimens. Most objects conservators thrive on variety.
There is no paintings conservator on staff at the Royal BC Museum, mainly owing to the fact that we have a comparatively small collection of paintings. When there is paintings conservation work to be done, a highly respected and dependable local contract conservator does the work for us. The recent work was in preparation for our upcoming Gold Rush exhibition, due to open on May 13th. We plan to display a very large painting, affectionately nicknamed “Slim Jim”, but actually titled “The Parson Takes the Pot”, created by Rowland Lee, which measures almost 2 x 3 meters (much larger if you include its very ornate frame!). The logistics of moving, hanging, and protecting such a large picture is another story. At this point in time, it was felt that the varnish on the painting had yellowed and turned a bit milky over the years, obscuring the vibrant colours of the paint beneath. Investigation by the paintings conservator concluded that the varnish had not discoloured, but was actually a modern varnish applied in 1978 by the Canadian Conservation Institute in Ottawa. The problem was actually a layer of dust and nicotine adhering to the surface. Sometime between 1978 and now, the painting was hung in the office of a smoker. That will do it!
Our contract conservator had little time to spend on the painting, and the exhibition deadline is looming, so I offered to work alongside the paintings conservator, using the hand skills I learned while working on objects, and taking direction from the expert. This way we could finish the cleaning in half the time. It worked out wonderfully. The layer of dust and nicotine was removed with a gentle solvent applied by cotton swab, and rinsed with distilled water. The difference was immediately obvious, as the colours burst forth under our swabs and the actual extent of masking became evident. This is something every conservator appreciates; strikingly different before and after treatment photographs. After only about 6 hours of tandem cleaning, the painting now literally shines and the depth of colour is gratifying. My first foray into paintings conservation was a success!
If you visit Cranbrook BC you might come across a statue of an elephant. In 1926 an elephant called Charlie Ed escaped from the circus and went on the lam for a few weeks. When he was eventually recaptured, he was renamed Cranbrook Ed to commemorate his time there.
In George Winkler’s photo album from 1925 to 1939, there are two pictures of Cranbrook Ed being loaded onto a train. Winkler identified them simply as “Cranbrook Ed re-captured”. This album is part of accession 197908-016 and is stored in container 000491-0002.
He also kept copies of these prints amongst some of his loose photographs identified as: “Elephant that escaped from circus at Cranbrook – 1926 – recaptured” (BC Archives photo I-30605) and “Last view Cranbrook had of recaptured elephant – 1926” (BC Archives photo I-30606). These prints are in container 000491-0001, folder 13.
Did Winkler take these photographs or were they popular prints available for sale? They match the other prints in his album in shape and size so it’s likely that he did take them. Lucky for us that he was in Cranbrook at the time of the elephant capture, and that he had a camera with him.
I-30605
I-30606
You can visit the Cranbrook city website at http://cranbrook.ca/our-city/history/interesting-stories/ to see a picture of the statue and read the story of Cranbrook Ed the elephant.
On Family Day (February 9th, 2015) I received two hatchling Red-eared Sliders. These turtles are just like any other hatchling red-ears except for one thing. They hatched in British Columbia, and came from the first nest known to have survived our cooler coastal climate. These turtles are native to the south-eastern United States, and range south into Central and South America, and have been spread far and wide because of the food and pet trade, including Washington. They are scattered all over south-western BC and the Okanagan.
 An adult Red-eared Slider from Beacon Hill Park, Victoria (my photo)
An adult Red-eared Slider from Beacon Hill Park, Victoria (my photo)
Several nests have been known to survive to near hatching, but our winters prevented survival of hatchlings – yes even the mild winters of south-western British Columbia are too cool – or so we hoped – for Red-eared Slider reproduction. Back in January 2014, researchers with the Coastal Painted Turtle Project discovered nests of the non-native red-eared slider and suggested that this species should be able to breed here in BC. Their excavations of known red-ear nests showed that red-ears are fully developed, but they are hibernating and could emerge in spring if they have enough energy stores for a prolonged hibernation period.
 This composite image from a failed nest is from the Coastal Painted Turtle Project crew’s facebook page (photos by Deanna MacTavish).
This composite image from a failed nest is from the Coastal Painted Turtle Project crew’s facebook page (photos by Deanna MacTavish).
One year later – a different nest – and six live hatchlings were found near Painted Turtle nests that were being regularly monitored by staff at Riefel Island Bird Sanctuary, Delta. This is the first record of fully hatched, viable red-eared Sliders in British Columbia. Until now, populations were maintained by a continual stream of abandoned pets. This nest had been opened by Sandhill Cranes. The cranes eat eggs, but left these hatchlings.
 Here are all six hatchlings showing their unique shell patterns – 1 still shows the remnants of its belly button (photos by Aimee Mitchell).
Here are all six hatchlings showing their unique shell patterns – 1 still shows the remnants of its belly button (photos by Aimee Mitchell).
I am keeping only two to show to the public, namely, 4 and 6 – they will be named accordingly – just like Humanoid Cylons. I’ll post updates to their progress over the years. Three and Five showed strange vertebral scutes and this sort of malformed shell is not uncommon in the pet trade animals. I asked for 4 and 6 because they were decent looking well-proportioned animals. I’ll be curious to see if they are the same sex – sex is determined by nest temperature. Eggs incubated at low temperatures (22-27°C) produce males, warmer nests produce females – to a point – over heat a nest and it produces no turtles. One, Two, Three, and Five will be humanely euthanized and added to the Royal BC Museum research collection.
 Red-eared Turtles from a failed nest, found when land owners were building a boathouse on Beaver Lake, Vancouver Island (RBCM 1994) (my photo).
Red-eared Turtles from a failed nest, found when land owners were building a boathouse on Beaver Lake, Vancouver Island (RBCM 1994) (my photo).
Nest temperature may be our only saving grace now that these turtles are reproducing here. If British Columbia nests produce all male hatchlings, then eventually the population will fail as sex ratios become too biased. I see very few males in wild populations. I have always assumed this is due to the large size of females, and that people prefer turtles that stay small. Large turtles are far more work (water changes, filter maintenance), and so they get abandoned in ponds, streams and lakes far more often than smaller turtles. But if males are produced by cooler nests, then we may have a population boom until all the abandoned females die of old age (or are removed).
In the meantime, we have to feel sorry for all the Western Painted Turtles in British Columbia – they are our only native freshwater turtle, and will have to coexist and compete with Red-eared Sliders for aquatic habitat and nest sites. With all the development in south-western BC, the last thing a Painted Turtle needs is more competition for their place in the sun.
 Red-eared Turtles on log in Matheson Lake, Vancouver Island – not a Painted Turtle among them (my photo).
Red-eared Turtles on log in Matheson Lake, Vancouver Island – not a Painted Turtle among them (my photo).
In the fall of 2014 I had a lot of fun processing the records of George E. Winkler, the poet prospector of British Columbia.
The fonds are described as PR-0045, and consist of records from two separate accessions. One accession came to us in 1979 from George Winkler himself through his will. The other accession came a few years later from Winkler’s great friend Hartley Sargent, Chief Mining Engineer for the province.
Winkler died in 1978 at the age of 103 and he spent his long life involved in the mining industry in a variety of ways. He was a prospector who also managed several mining concerns on Vancouver Island and in Northern BC; he published poetry under the pseudonyms “The Prospector” and “Ernest Altrew”; he was a Socialist who unsuccessfully ran for office in 1916, and he knew some of BC’s famous and infamous people.
Born in Ontario in 1875, he grew up in Manitoba but came to BC to find his fortune around 1900 and never left.
Here’s a portrait of Winkler taken around 1908 in Victoria by Gibson Studio. He probably had it taken to send back to his mother in Manitoba. They wrote to each other regularly until she died. This photo is in container 000491-001, folder 16.
Hit the Road post
I regularly get asked if we go out and actively kill animals for the Royal BC Museum collection. The answer is yes – but only specific groups are sampled regularly. Generally I only catch fishes, anaesthetize them, and fix/preserve them for the research collection. Birds, mammals, reptiles and amphibians generally are found as incidental items – window kills, road kills, natural mortality (like marine mammals and sea turtles), or bycatch from commercial harvests.
We also get bycatch from other sampling techniques – pitfall traps for insect sampling commonly also provide the vertebrate collection with a few salamanders a year.
 This Valentine’s Day we had an event at the museum. Animal Sex and people could chat with experts on the topic… and the questions as you’d expect were hilarious – but in addition to more racy intercourse, many people wanted to know whether I go out and kill animals for research. That prompted this short note.
This Valentine’s Day we had an event at the museum. Animal Sex and people could chat with experts on the topic… and the questions as you’d expect were hilarious – but in addition to more racy intercourse, many people wanted to know whether I go out and kill animals for research. That prompted this short note.
The answer is yes – but it is very rare that I go out to catch and kill wildlife. Instead, I rely on other sources for the majority of mammals, birds, amphibians and reptiles. For example, the day after Valentines (a Sunday) I picked up a road-killed Peregrine Falcon from near Duncan.
 The bird had been found by a high-school teacher back in December, and he saved for bird for the museum. The bird was in great shape. We don’t get Peregrine Falcons every day (thankfully), and I certainly could not justify shooting one just to stuff it and put it away for future study. This bird was found by the roadside and adds to the falcons in our collection. This bird will be prepared as a partial skeleton, a spread wing (detached from the bird) and a study skin, and will be available hundreds of years from now for scientific study.
The bird had been found by a high-school teacher back in December, and he saved for bird for the museum. The bird was in great shape. We don’t get Peregrine Falcons every day (thankfully), and I certainly could not justify shooting one just to stuff it and put it away for future study. This bird was found by the roadside and adds to the falcons in our collection. This bird will be prepared as a partial skeleton, a spread wing (detached from the bird) and a study skin, and will be available hundreds of years from now for scientific study.
 Researchers use study skins in a variety of ways, from genetic research to assessment of colour variation in a species. These photos were taken during a workshop where researchers verified sub-species variation and plumage patterns as you’d find in field books.
Researchers use study skins in a variety of ways, from genetic research to assessment of colour variation in a species. These photos were taken during a workshop where researchers verified sub-species variation and plumage patterns as you’d find in field books.
 This is a great example of citizen science – and I thank everyone who remembers to save specimens for the RBCM – we even want specimens that represent common species. I am happy to get new specimens as long as they are in good shape, and you record the location, date, and time the animal was collected.
This is a great example of citizen science – and I thank everyone who remembers to save specimens for the RBCM – we even want specimens that represent common species. I am happy to get new specimens as long as they are in good shape, and you record the location, date, and time the animal was collected.
 Honest, I am listening intently in this photo – and not thinking about other birds.
Honest, I am listening intently in this photo – and not thinking about other birds.
I recently came across a folder of photographs in one of our “miscellaneous” accessions. Although the accession was created in 1991, we had probably had the material for many years without knowing quite how and when we acquired it.
The folder contains photographs of Fanny Buckthought Morse and her husband Arthur Cromar Bruce as well as a photograph of the Women’s Volunteer Reserve that Fanny served in.
Using the online Vital Event registration indexes, I’ve been able to track down their marriage registration as well as Fanny’s death registration and it’s interesting how much information you can glean from these records:
They were married in Vancouver in November 1916. Arthur is listed as being born in Aberdeen, Scotland, was a 28 year old bachelor living in Vancouver and his profession was journalist. Fanny was born in Bournemouth, England, was listed as a 30 year old spinster and her profession was also journalist. Her death registration from December 29th, 1961 has her living in the Nanaimo area but gives her birthdate as February 8, 1884 which means she was actually 32 at the time of her marriage.
From 199105-003 (miscellaneous accession), container 001239-0001, file 13:
“Mother, Fanny Cromar Bruce (nee Morse) camping Horseshoe Bay”
“1914-18 Women’s Volunteer Reserve (FCB not present owing to duty on News-Advertiser) steps of Aberdeen School, Vancouver”
According to the stamp on the back, this photograph was reprinted by Vancouver Drug Co. Ltd., May 9, 1939. Fanny’s son or daughter wrote the notes on the back of the photographs.
There’s quite a variation in the women’s uniforms, I wonder if they had to supply their own?
Yesterday (Monday January 19), I was pulling out specimens of Weather Loaches for a presentation I will give in Richmond later in the week in invasive species. In the adjacent tub of unsorted fishes was a jar with a sizable fish (152 mm Standard Length, 177 mm Total Length), and its label said, “Unknown Fish”. Once again I heard Lando Calrissian: “Hello, what have we here?”
 It didn’t take long to figure out what species we had. The fish had an adipose fin, a smelt-like deep and compressed body, and a sharp nose with a small mouth. It also was well-preserved – that makes a huge difference. This new fish is a Pacific Argentine – Argentina sialis. According to the Love et al. (2005), and Eschmeyer et al. (1983), this species ranges in the eastern North Pacific from Baja to offshore of the mouth of the Columbia River, and south of the equator to northern Peru. Our new location is roughly 350 km north of the mouth of the Columbia River.
It didn’t take long to figure out what species we had. The fish had an adipose fin, a smelt-like deep and compressed body, and a sharp nose with a small mouth. It also was well-preserved – that makes a huge difference. This new fish is a Pacific Argentine – Argentina sialis. According to the Love et al. (2005), and Eschmeyer et al. (1983), this species ranges in the eastern North Pacific from Baja to offshore of the mouth of the Columbia River, and south of the equator to northern Peru. Our new location is roughly 350 km north of the mouth of the Columbia River.
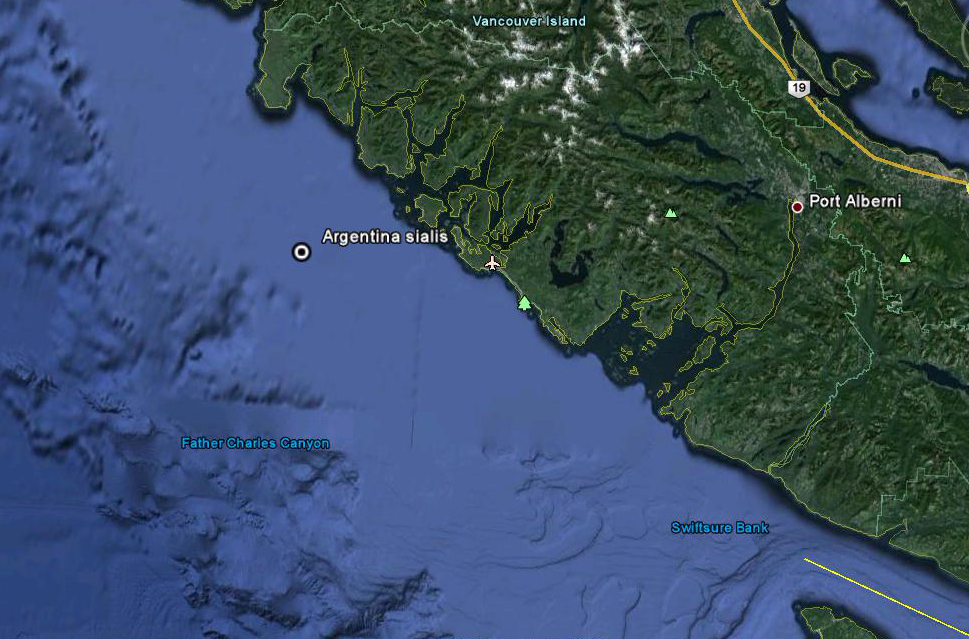 Peden (2002) is the most recent checklist specific to British Columbia, and his list does not include this species (nor it’s family, Argentinidae). Chalk up another new marine fish for our fauna. Is it a case of northward expansion due to climate change? Time will tell – right now we only have one fish from southern BC from 2011. One fish does not make a trend. However, if more appear – then we’ll have a better argument for warming water and northward dispersal of marine fishes. Maybe the Leopard Shark will be next (fingers crossed).
Peden (2002) is the most recent checklist specific to British Columbia, and his list does not include this species (nor it’s family, Argentinidae). Chalk up another new marine fish for our fauna. Is it a case of northward expansion due to climate change? Time will tell – right now we only have one fish from southern BC from 2011. One fish does not make a trend. However, if more appear – then we’ll have a better argument for warming water and northward dispersal of marine fishes. Maybe the Leopard Shark will be next (fingers crossed).
 Argentina has it root in argentus (silver), and the species name sialis may refer to the snout of this fish and its resemblance to a birds beak. Although perhaps the type specimen had a blue tint due to refraction off its skin and scales and reminded Gilbert (1890) of the Eastern Bluebird (Sialia sialis).
Argentina has it root in argentus (silver), and the species name sialis may refer to the snout of this fish and its resemblance to a birds beak. Although perhaps the type specimen had a blue tint due to refraction off its skin and scales and reminded Gilbert (1890) of the Eastern Bluebird (Sialia sialis).
At its very simplest, a biological specimen in a museum collection is one representative of a species from a specific time and place. Some may say, “Who cares, you’ve seen one Leopard Frog, you’ve seen ’em all”.
Did you see a male? Female? Young? Old? Abnormal? What did it eat? Was it gravid? What diseases or parasites were present? Was it from within its normal range or was it in a new location? Every specimen is different. Every specimen is worth keeping – not just for present-day research – but because each specimen represents a time capsule of biological information. We always save specimens in museum collections precisely because biological specimens are unique. In some cases, species represented in museum collections have vanished from BC. There are very few breeding Leopard Frogs in BC – perhaps like the Sage Grouse, British Columbia’s Leopard Frogs will become extirpated. That’s beyond sad.
But in this era – the Econocene – where we seem to frame decisions on the almighty dollar – what is the value of a specimen? Can you put a dollar value on a dead animal? To break it down to the basics, you have to consider what it would cost to go out and collect a new animal for research. You may also reflect on whether you should kill another animal given present rates of habitat destruction and population declines.
Consider just vehicle rental, fuel, ferry to the mainland (since this institution is on an island), permits, field gear, food for the field crew, and salaries – and if you work it out – each museum specimen at the time of collection is worth hundreds to thousands of dollars depending on where you sampled. You’d think entomology may be the exception because they can dilute the cost per specimen by taking thousands of specimens per field trip – but this is not the case. To paraphrase Claudia Copley:
The true cost to send 3 people into the field and do the work is like that of any other collecting trip (we just happen to be 2/3 volunteers). Although thousands of specimens may be brought back, the real cost emerges during identification. A researcher may charge an hourly rate for identifications, and it sometimes takes a few hours to identify a specimen. In the end, a specimen identified to species in an entomology collection probably ends up having a similar value as a vertebrate specimen.
Entomologists collect thousands of specimens because many cannot be identified in the field, and a large sample is needed to encompass the diversity that is present.
Furthermore, costs always rise (although the price of oil has experienced a hiccup lately). As Lando Calrissian said, “This deal is getting worse all the time”.
Imagine the escalation of costs if you needed to build a time machine – yea – obviously I am doing my usual trick of “one step too far” with this argument. But this is to demonstrate that you can’t slap a price tag on historical specimens like you can with a recently manufactured wrench, or a pair of socks at a department store. There is no amount of money that can send you back in time to sample Leopard Frogs from the 1800s. If we don’t save biological specimens – and properly care for them in museum collections – historical biology will go extinct.
 Where else but at a museum can you study a Cape May Warbler from 1927.
Where else but at a museum can you study a Cape May Warbler from 1927.
This thought hit-home yesterday (January 15th, 2015) when a researcher from the University of Victoria was here looking at mammals in our collection. She wanted to see what was available in the collection just in case she had the chance to expand her work beyond Alberta’s borders. Fortunately, we have loads of Fishers in the collection (the mustelid, not people with fishing rods – although we do have historic fishing rods). If the museum had not saved specimens (skins and skeletons, and in some cases, tissue samples), she’d have to request tens-of-thousands of dollars more in future research grants, and spend many long hours in the field (actually this part sounds nice), to get enough samples from BC for her work.
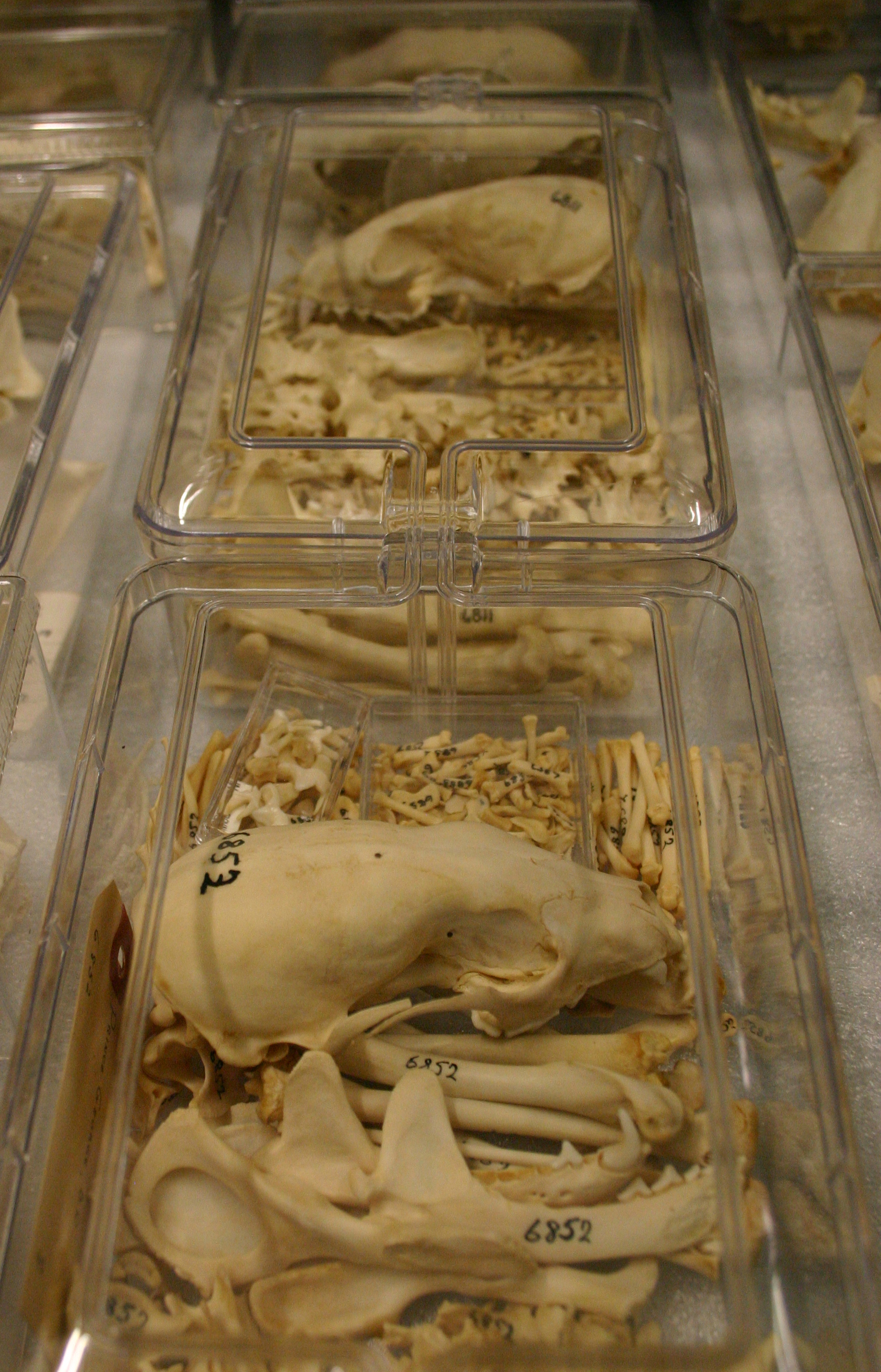 Another researcher came to us for tissue samples of Wolverines – and especially samples from Vancouver Island. Without museum specimens, his work would not be possible. Wolverines are extirpated from Vancouver Island.
Another researcher came to us for tissue samples of Wolverines – and especially samples from Vancouver Island. Without museum specimens, his work would not be possible. Wolverines are extirpated from Vancouver Island.
These are two examples where the foresight of museum experts from over 100 years ago, have ensured that research today is cost effective (and possible). Yes museums spend money maintaining their collections, but can you really put a dollar value on knowledge and scientific advancement? Can you put a dollar value on the snoot-value (prestige) of being the one place in the world where you can study Dawson’s Caribou? How about the value of a bee? Its value as a museum specimen notwithstanding, as a pollinator, bees more precious than gold. You don’t want a world without bees.
 Regardless of your interests, you have to agree that museums are unique and worthy of support – financial support. It doesn’t matter whether you want to study Eocene leaves, a Mosquito (the airplane or the insect), or the diet of Cutthroat Trout from the 1930s, collection and maintenance of specimens is costly. Only at a museum can you touch these objects from the past – and in this respect, museums are the cheapest time machine money can buy.
Regardless of your interests, you have to agree that museums are unique and worthy of support – financial support. It doesn’t matter whether you want to study Eocene leaves, a Mosquito (the airplane or the insect), or the diet of Cutthroat Trout from the 1930s, collection and maintenance of specimens is costly. Only at a museum can you touch these objects from the past – and in this respect, museums are the cheapest time machine money can buy.
On January 23rd, I will be presenting at a workshop on invasive species – and the fish I am showcasing is the Weather Loach (Misgurnus anguillicaudatus). Weather Loaches are related to minnows and suckers, but most will agree that they look more eel-like with their elongated bodies. There are other types of loaches, the most commonly seen in the pet trade are the Botia species and of course the Kuhli Loach. They all have a fringe of barbels around their mouths – not unlike that of like catfishes.
Most loach species in the pet trade are tropical and pose little threat to our waterways, however, European and North Asian loach species can tolerate cool water and will survive here if released.
 Weather Loaches are popular aquarium pets and also are cultured for food by some people. They are known to have survived in garden ponds in Canada, and escapees have established populations in streams in Washington. Others have escaped from commercial fish farms and have established breeding populations in many countries. Some countries now have banned Weather Loaches because of the risk to aquatic ecosystems – permits are needed to keep a related species (Misgurnus fossilis) in England. If only we could be so proactive regarding high-risk exotic species in the pet trade.
Weather Loaches are popular aquarium pets and also are cultured for food by some people. They are known to have survived in garden ponds in Canada, and escapees have established populations in streams in Washington. Others have escaped from commercial fish farms and have established breeding populations in many countries. Some countries now have banned Weather Loaches because of the risk to aquatic ecosystems – permits are needed to keep a related species (Misgurnus fossilis) in England. If only we could be so proactive regarding high-risk exotic species in the pet trade.
 Weather Loaches are unique among BC fishes since they are superficially eel-like in shape, but have barbels like a catfish. The body, which can reach 24 cm in length, is oval to almost square in cross section, and is golden-yellow with various amounts of darker brown mottling. Sometimes the mottled pattern coalesces into weakly defined stripes. Body scales are minute and embedded in thick skin. The head terminates in a small, almost tubular mouth that is surrounded by a fringe of 10 fine barbels. The eyes are small relative to the head, and a blade-like spine (which normally is retracted) is present just below the eye. This spine is sharp and may be extended for defense when loaches are captured by predators (or fish enthusiasts).
Weather Loaches are unique among BC fishes since they are superficially eel-like in shape, but have barbels like a catfish. The body, which can reach 24 cm in length, is oval to almost square in cross section, and is golden-yellow with various amounts of darker brown mottling. Sometimes the mottled pattern coalesces into weakly defined stripes. Body scales are minute and embedded in thick skin. The head terminates in a small, almost tubular mouth that is surrounded by a fringe of 10 fine barbels. The eyes are small relative to the head, and a blade-like spine (which normally is retracted) is present just below the eye. This spine is sharp and may be extended for defense when loaches are captured by predators (or fish enthusiasts).
Weather Loaches eat aquatic insect (chironomid) larvae, worms, molluscs, amphipods, and cladocera (nicknamed “water fleas”), and live in shallows of lakes, ponds, rivers, and swamps. They usually are collected over softer substrates like sand, silt, and mud in slow-flowing water. They use leaf litter and aquatic or emergent vegetation as cover, and will lie stationary with only the head protruding from shelter. Weather Loaches “comb” the river or lake bed for edible material and expel silt and mud out of their gills. The barbels taste the food hidden in the muck.
Weather Loaches are active at night, tolerate cold water, and survive in low oxygen conditions by swallowing air from the surface. The ingested air is forced through the gut. Oxygen is absorbed through the intestine lining, and bubbles of remaining air and carbon dioxide are passed out the anus. The excess gas is forced out when the fish flexes its body in a tight curve. They also can breathe through their skin.
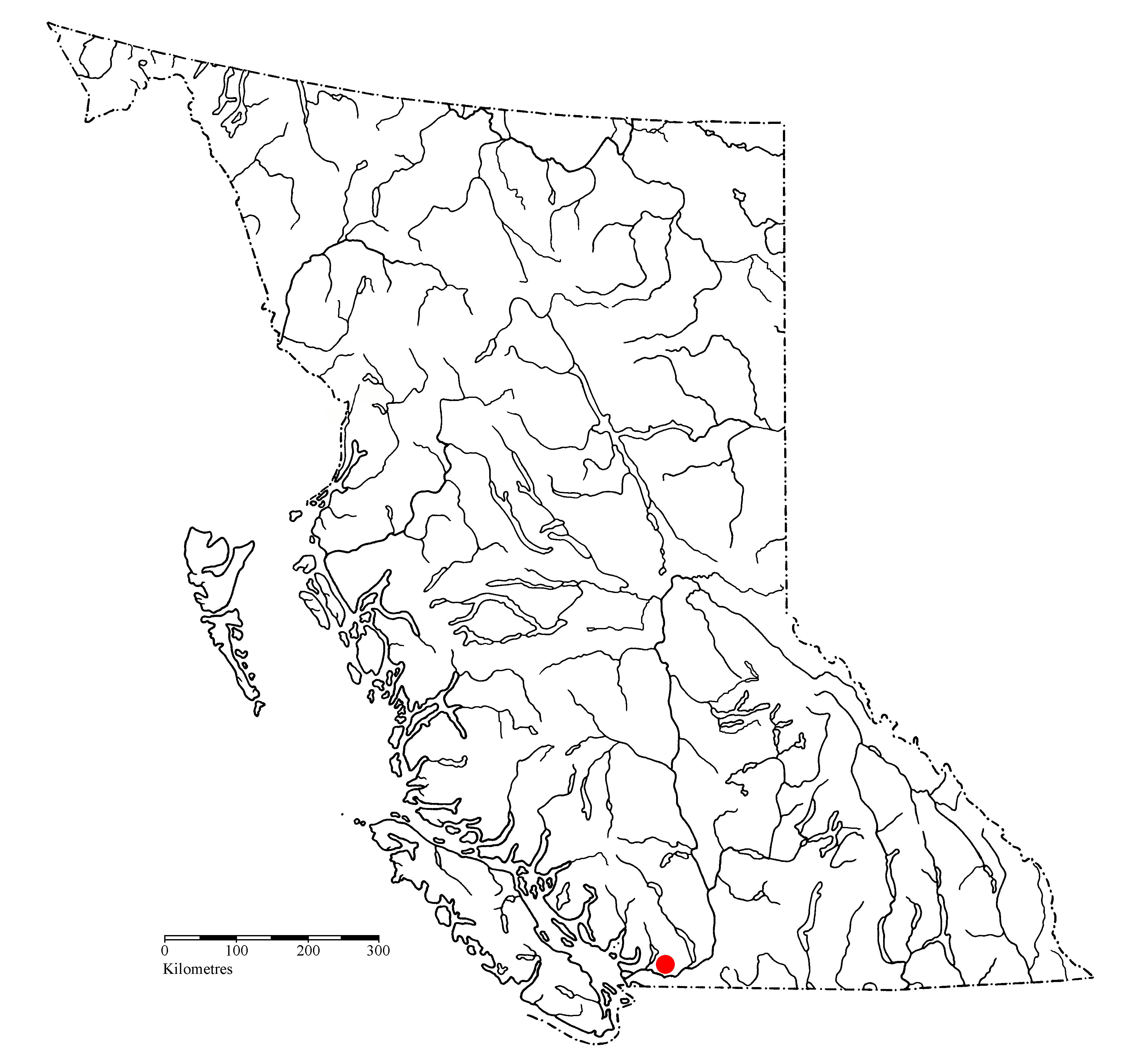 Weather Loaches are found from eastern Asia, from the Tugur, and Amur rivers, Sakhalin Island, and south to Korea, Japan, Taiwan, Vietnam, Thailand, and Burma. They also are known from introduced populations in the Philippines, Mexico, Australia, Palau, and isolated ponds in Germany, Italy, the Aral Sea basin, and England. Populations of Weather Loaches also are established in Tennessee, Florida, Michigan, Illinois, California, Oregon, Idaho, and Hawai’i.
Weather Loaches are found from eastern Asia, from the Tugur, and Amur rivers, Sakhalin Island, and south to Korea, Japan, Taiwan, Vietnam, Thailand, and Burma. They also are known from introduced populations in the Philippines, Mexico, Australia, Palau, and isolated ponds in Germany, Italy, the Aral Sea basin, and England. Populations of Weather Loaches also are established in Tennessee, Florida, Michigan, Illinois, California, Oregon, Idaho, and Hawai’i.
Weather Loaches were found in the summer of 2008 in the Alouette River system on the Lower Mainland, although rumours of their presence near Chilliwack date back to 2003. In 2009, 2 were caught, and in 2010, 22 were caught, with fishes ranging from 123 mm to 205 mm in length. The size range of fishes caught in the Alouette River system suggests that several age classes of Weather Loach are here and they may already be breeding in British Columbia. It is likely that the species will spread in the sluggish streams of the lower mainland in British Columbia. In Washington, most specimens are from independent releases of one or only a few fishes and they fail to establish breeding populations (e.g., Jovita Creek, and Tulalip Creek), but unfortunately, several specimens now are known from Lake Washington and its tributaries and so it is likely that a breeding population exists there.
Weather Loaches spawn in spring on silt to mud substrates and in beds of aquatic vegetation. Males and females swim around the fertilised egg mass until it is covered with a layer of silt, and then abandon the site. Eggs hatch in a few weeks and the young loaches bury in the muddy substrate.
Weather Loaches are able to move across land during wet conditions and so could transfer between headwaters of closely-spaced drainage basins. Because of their cold water tolerance, Weather Loaches commonly are sold as scavengers for goldfish tanks (marketed as Weather Loaches, Dojo Loaches, and Golden Dojo Loaches). Any Weather Loaches in North America should stay in captivity as long as they are alive, and given their ability to move across land, should never be kept in garden ponds. The source of Weather Loaches in British Columbia is unknown. They may have been released here in an attempt to produce a local population for harvest, either for bait or human consumption. While the fish themselves may seem innocuous, they are known to carry Birnavirus (LV1), which is related to pancreatic necrosis in salmonids, and they carry parasites which could survive and transfer to native fishes. Please think before releasing any fish from captivity – and then take your fish back to a pet shop. In doing so, you’ll be protecting BC’s native flora and fauna.
Christmas cracker post
Doing some last minute shopping on Christmas Eve? If you were in Ocean Falls in 1963, you would probably visit the Hudson’s Bay Company store. There you would find delicious Christmassy food, stop by the drugstore to buy a ponytail extension and then visit Toyland for the kiddies. These photographs are from MS-2173, “A History of Ocean Falls in pictures”
Maybe you’ve got some activities planned with the children…you can make a snowman and attend a Christmas party at the school. These photographs are from the Harry Jomini fonds, PR-2291. They show the temporary camp at Nechako which was built to house the Kenney Dam workers and their families between 1951 and 1954.
Speaking of snowmen, here are some snow sculptures from Winterfest in Kimberley, BC. You can see that the Kimberley people also had the choice of shopping at Eaton’s (remember Eaton’s?). These photographs were taken by a BC Government photographer for Beautiful British Columbia magazine in 1976 and are part of GR-3265.
I-08424 and I-08425
And sometimes just getting together for drinks is celebration enough. This photograph is from 1903 and was taken at Deep Lead Camp, Slough Creek in the Cariboo district
H-02842
The staff at the BC Archives has always appreciated the kindness of our regular researchers when they remember us at Christmas. Over the years we have received many good wishes in cards, which we display in our break room over the holiday season.
But I think the most unusual card came to us from G.S. Andrews. Gerald Smedley Andrews was the Surveyor-General of British Columbia and a long time user and supporter of the Provincial Archives. We are lucky enough to have his private records, described as PR-1059, which consists of 6.4 m of textual records and other material.
In 1960, Gerry created the following card.
The front of the card is very restrained and elegant.
The inside contains a drawing of the Parliament Buildings done in his best “surveyor” style, and a message to the staff.
The back of the card contains the footnotes!
This card, and others, are part of accession 92-0334.
It’s one thing to build a bridge, but once it’s on the books, it needs to be maintained, refurbished, repaired and generally kept safe.
Maintenance is one of the most importance functions of any Public Works or Engineering Dept., because a collapse can be disastrous.
The Chilcotin suspension bridge was built ca. 1913 and a mere twelve years later, this paragraph appeared in the Public Works report for 1925, district no. 9, 150 Mile House:
“The most important bridgework was the repairs to the Chilcotin Suspension Bridge. The 2 ½ inch main cables on the 320-foot suspension span was shortened to give camber and rigidity to the deck. A new deck and hand-rail truss were put on, also guy-cables, all without closing the bridge to traffic. This bridge, which has been undergoing a series of extensive repairs during the past four years, is now in first-class condition. E.S. Jones, district engineer.”
Here are some photographs of the Chilcotin suspension bridge from GR-3293, file AAAE2160, stored in container 000705-0013. The file contains photographs taken between 1922 and 1926.
42-2-1.2
File 239 Lillooet dist. Chicotin suspension bridge 17/9/24 main cables have been shortened and lightened…inverted cable slack and counterweight boxes dismantled.
42-2-1.5 Chilcotin suspension bridge re-construction completed 8-12-24.
Compost Creativity post
In my previous blog article on the Humpback Whale from Tofino, I mentioned that the whale’s skeleton was cleaned with a combination of mechanical removal of meat, and then it matured in a mixture of soil and aged manure.
A few weeks ago I found a perfect skull in one of my garden compost bins. It came from one of the Norway Rats that I caught this summer. I actually set a mine-field of rat traps in the compost to get those few rats that try to raid the bin for kitchen scraps. If they set off one trap, they generally jump land on another trap. People say not to throw meat in a compost, but we don’t notice any smell from few rats we toss in the compost each year.
Compost – who knew it was so versatile. It fertilizes your garden, but it can help you prepare skeletons.
There are a few animal skeletons you probably can possess without a wildlife permit – Grey Squirrels and Norway Rats, Rock Pigeons and perhaps deceased pets (Gerbils, Hamsters, etc.). If you want to prepare skeletons it’s a fairly simple process.
Let’s assume you are preparing a Grey Squirrel. It is messy at first. You skin it, and remove most of the meat (and guts). Don’t bother trying to remove the brains – they’ll rot away. Make a long flat bag out of standard window screen (or some finer mesh) – and staple that around the stripped carcass so that the bones will be held in place once the connective tissue is gone. The screen also prevents scavengers from scattering bones.
The red dots around this Wall Lizard show where I’d staple or pin together the mesh to hold the bones in place.
Place the carcass in its screen bag in a shallow tray (the plastic trays you get from Garden centers are perfect) with a mixture of aged manure and soil.
Let it sit for a few months. If that tray with the carcass/manure mix is on top of the contents of your compost bin, it should keep the temperature and humidity just right. Check periodically to make sure the soil is fairly damp.
Ages ago I bragged to a colleague about my wonderful technique for rotting carcasses and reconstruction of nice clean skeletal material. He gave me a sideways glance and said, “where do you think fossils come from”. Yep, I have to admit that the technique is nothing new. Here are naturally prepped fishes from the Early Devonian – only the mineralised bits remain.
After a month or two you should end up with a clean skeleton that smells like soil. The time required is directly dependent on temperature, humidity and the amount of meat you left on the animal. The skeleton won’t be perfectly white – but the screen bag should contain all the small bones roughly in sequence. Now you carefully cut the bag a bit at a time (I’d suggest starting with the extremities and work inwards) and you can carefully sort the bones as you encounter them to make an accurate reconstruction. Set out the bones in sequence on paper towels and let the bones dry.
Some people use peroxide or chlorine bleach to whiten bones, but I don’t. Bones will deteriorate faster if bleached.
I use standard white wood glue to reassemble the skeleton. If you get something wrong, wood glue can be soaked in warm water and removed. Plasticene or silly-putty can be used to hold bones in position while the glue sets. This is a long-term project – not a snap-together kit. Glue a few bones together – wait a day for the glue to set. And repeat until the skeleton is assembled. You only need enough glue to bridge joints. Remove any excess glue so there are no drips.
Once the skull or skeleton is complete, give it a spray with a 5 to 10% solution of white glue in water to seal the bones and prevent drying and cracking. This top-coat of dilute glue was an exaptation from my model railroading hobby (yes, I can tie skeletal preparation to one of my many hobbies). In model railroading, you spray dilute glue onto the scenery to keep bushes etc., in place. Here’s an example of exceptional railroad scenery where a miss-timed sneeze could destroy a forest (if things were not fixed in place).
Some people ask why I don’t use beetles or ants to clean display skeletons. Simply put, insects can scatter the smaller bones. Beetles are fine for museum specimens where the skeleton will be stored as disarticulated bones. But if you want to reconstruct the skeleton, you want to know where each tiny bone originated.
You can also soak a mostly-cleaned carcass in a water-filled aquarium and let the bones rot until clean [don’t put bones in an aquarium containing fish or any other aquatic pets]. To do this, you also put the carcass in a mesh bag, and then weigh it down. Sometimes the gas produced will cause a carcass to float – and then any bones will be displaced and you’ll have to search for them. It also will be very difficult to reassign finger bones (phalanges) to their correct position. The downside to this technique is the smell, and while an aquarium filter can reduce the odour, eventually you will have to pick bones from the stinky pink sludge which accumulates on the bottom of the aquarium. These bones can be rinsed in freshwater and then left to dry – and eventually the smell is not so noticeable.
The skulls below (Cuban Anole, Red-footed Tortoise and Spectacled Caiman) were prepared in an aquarium – any loose bits were glued back together. They did turn out really nice, but you do need to be prepared for the smell. The Anole skull fell completely apart and had to be glued back together: there were 6 bones just for the jaws, and you have to position the bones of the palate relative to the rest of the skull – it is quite a 3D puzzle.
I don’t know of a step-by-step instruction manual to reconstructing skeletons – they are not model kits after all – so you do have to do some research into anatomy and figure out how all the parts fit together. The up-side to assembling a fully disarticulated rat skeleton is that you can position your newly cleaned rat in a dynamic pose – climbing, running, or working a computer’s mouse…
Has it snowed in Vanderhoof yet? Has the snow plough been spotted clearing the roads?
Here are some charming action photographs from GR-3293, Engineering photographs. They are in container 000705-0014, file AAAE3585.
Although this series of photographs tends to focus on the building of bridges and roads in the province, it also shows the routine seasonal chores undertaken by the Public Works Dept.
87-1-3 Vanderhoof snow plough 1933
The ocean is renowned for its spectacular diversity: endless shapes, brilliant colours, extraordinary species and some of the most magnificent habitats on the planet – indeed, the ocean supports a great diversity of life and ecosystems. While much of this richness lies beneath the surface of the sea, sometimes (on rare and special occasions) we get to experience these hidden treasures first-hand.
This past month, the Royal BC Museum and the Victoria Natural History Society co-hosted a public beach seine. More than 100 people, including several members of the Canadian Network for Ocean Education (CaNOE), turned out on a dark November night to participate in the “in-seine” event.
The plan was to seine over a nearby eelgrass bed, catching as many species of squishy (invertebrates) and fishy (fishes) as possible. Unfortunately for our beach seiners, a large amount of sea lettuce was present near shore, resulting in a very large shrimp-fish salad. Not to be deterred by a bit of leafy green, we carefully sifted through the seaweed, discovering a variety of fascinating animals, from tiny amphipods and shrimps to large dungeness crabs, gunnels, and starry flounders. We explored, we learned, we connected. It was in-seine-ly fun!
 Huge thank you to Chris O’Connor and Gavin Hanke for help with organizing the event, Gavin Hanke and David Robichaud for braving cold November water, Nikki Wright for highlighting the importance of eelgrass beds, Yogi Carolsfeld, Phil Lambert, and Marilyn Lambert for sharing their expertise and passion for marine critters, Aerin Jacob for giving a positive shout out, Heather Murray for photocapturing the in-seine diversity and fun, and the avid natural historians who turned out to participate in the event.
Huge thank you to Chris O’Connor and Gavin Hanke for help with organizing the event, Gavin Hanke and David Robichaud for braving cold November water, Nikki Wright for highlighting the importance of eelgrass beds, Yogi Carolsfeld, Phil Lambert, and Marilyn Lambert for sharing their expertise and passion for marine critters, Aerin Jacob for giving a positive shout out, Heather Murray for photocapturing the in-seine diversity and fun, and the avid natural historians who turned out to participate in the event. 
A unique footbridge post
Sometimes an explanation is needed to fully appreciate the ingenuity of the engineers who provide bridges in the province.
This photograph is from GR-3293, file AAAAE2160 from container 000705-0013. The file is merely marked 26-2-2 “Crown Mountain suspension footbridge (Pemberton Meadows).” There is no date but I think it was taken around 1925.
If the file only contained the photograph, we might not pay too much attention, but the accompanying note from O.W. Smith provides some interesting information. This is a temporary footbridge created each winter by using the reaction ferry cable. I personally can’t imagine actually walking over this bridge, especially with a bag of groceries in one hand and a small child in the other, but no doubt the residents got a lot of use out of this unique footbridge.
Over the last few years, Makerspaces have become popular ways for people to explore their creative side. A facility provides a space, a variety of materials and the visitor brings their ideas and time to make something. Locally the Victoria Makerspace in Saanich is cool place to learn and share and there are many museums getting on the makerspace bandwagon including The Exploratorium’s Tinkering Studio http://tinkering.exploratorium.edu/ and Children’s Museum of Pittsburg Makeshop https://pittsburghkids.org/exhibits/makeshop .
On November 15, we experimented with the makerspace concept in a workshop called “Creative Collections”. Working with some community members, Ben Fast and Lauren Chancellor, we devised an event that involved using the collection to find inspiration for our creativity.
The workshop began with a trip behind the scenes to look at our handling collections. Participants looked, took notes, snapped photos and even drew sketches.
Then they went through the exhibit arts studio where our professional “makers” spend their days creating and building museum exhibits and interactives.
Back in the “lab” we provided a bevy of supplies – from clay to copper wire and lots in between. Participants made many wonderful things including a camera made from broken records, note cards personalized with handmade linocuts and even a letter about a talking cockroach.
The workshop was satisfying in many ways and I am interested in exploring how it might work in a more ongoing way. At the workshop we had pretty low-tech equipment and I am not sure if that is a bad thing or not. What are your thoughts? Do makerspaces need 3D printers and drill presses or can they work as a simple, collaborative place to come together and make time for expressing your creative side.
Indoor flood post
Here are two reasons to be careful when storing records in a basement.
These images are from GR-3422, box 1, folder 6. This series is called Hospital programs photographs and consists of over 2000 photographs created by the British Columbia Hospital Programs and its predecessor body the British Columbia Hospital Insurance Service. This file is dated December 1956 and is called BCHIS: flood damage to basement.
We don’t know too much about these pictures; where they were taken, what kinds of health records were affected by the flood, how high the water got or even if any of the records were salvaged…but the photos are a good reminder to think carefully about how we store our valuable records.
A rat in a compost pile is nothing new for Victoria. Sometimes I set a small mine-field of traps in the compost. A rat may jump clear of one trap, but they commonly land on a nearby trap and their fate is sealed. The rats I kill sometimes end up as museum specimens – even common animals should be added to museum collections. If I leave the rat in the compost too long, I find nice clean skeletons. However, on Friday the 7th of November the museum received a specimen which had sat in a mix of well-aged manure and soil, and is far more spectacular than an everyday compost-raiding rat.
Mike deRoos of Cetacea Inc., backed into the RBCM’s loading bay and delivered most of the skeleton of a humpback whale. I wrote a short article about the jaws of this whale months ago, and now we have limbs, ribs, and vertebrae. The skull will arrive in a separate shipment because there is a limit to what you can safely stack in the back of a pickup truck.
Most of the Axial Skeleton lined up.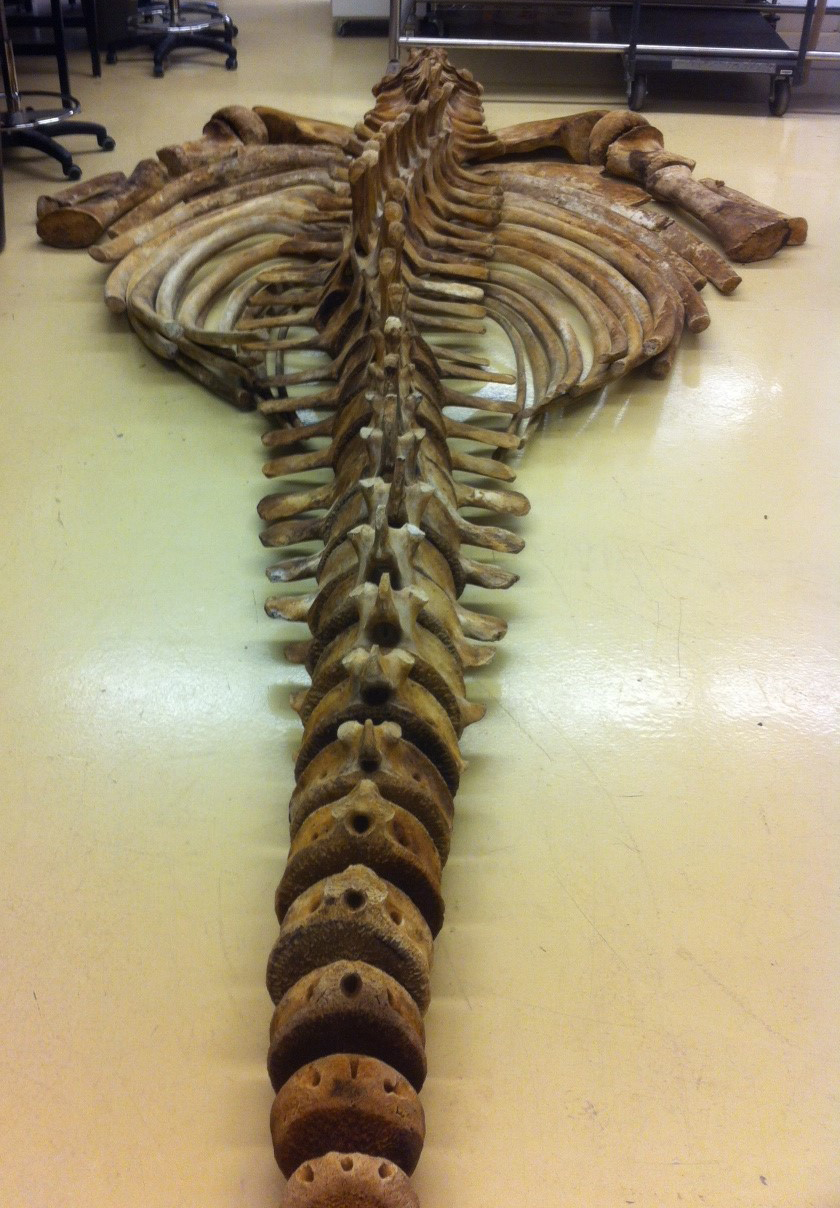
It took 5 trips from the loading bay to haul all the bones to the vertebrate prep-lab. Because these bones had been prepared in compost, they smelled sweetly of soil from a pine forest. Seriously – next time you are out in a pine forest, grab a handful of moist soil and smell it. You’d think the skeleton of a recently prepared whale would smell of rancid oils and rotten flesh – but not this Humpback. We could market the bones as “forest scent” potpourri. Call me strange (many have already), but I like the smell of soil.
A series of photos showing the length of the axial skeleton.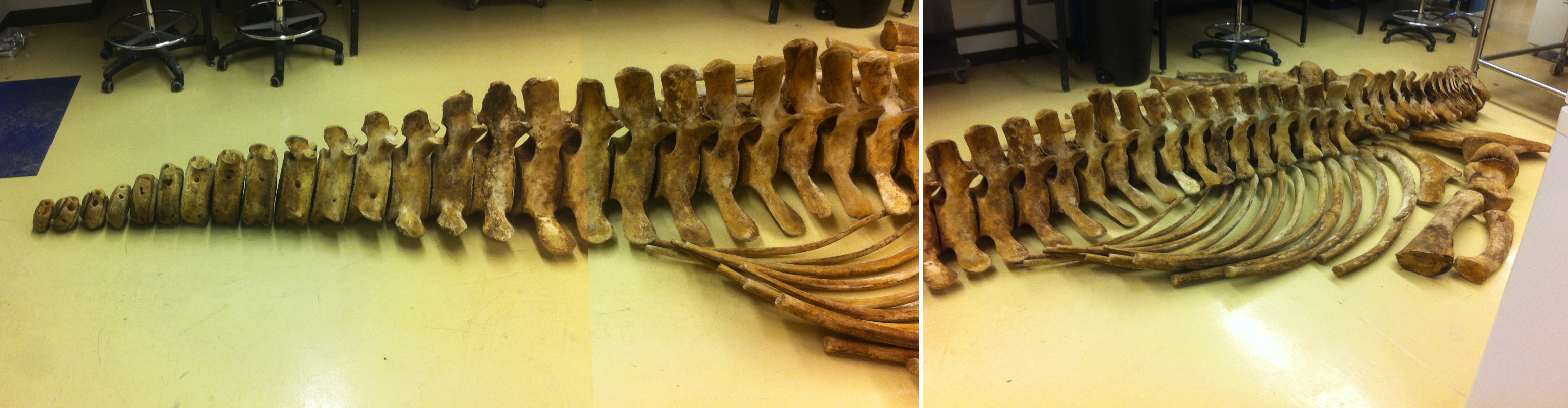
This whale was young – and only 9 meters. The ends of the bones (the epiphyses) have not fused and so the ends of the radius, ulna, and humerus all are separate.
The tips of the humerus, radius and ulna with unfused epiphyses.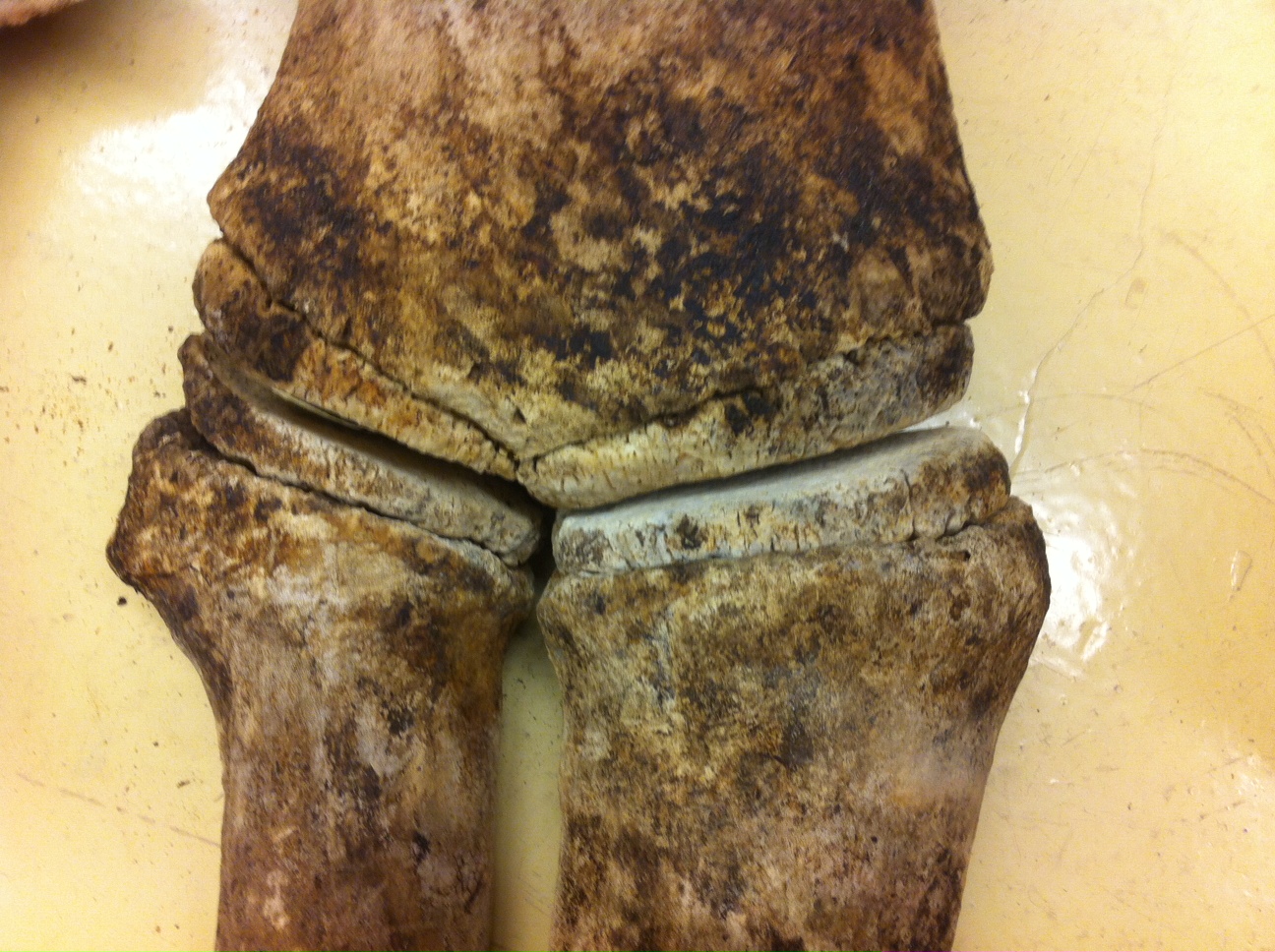
Each vertebra also has a pair of “cookies” (I think Mike deRoos’ daughter called them pancakes because the disks are so large). The cookies have a specific fit to respective vertebral centra (they grow together so you’d expect an intimate connection) – and so one of the first things I did was to carefully test fit the cookies to the vertebrae. The size and shape of each cookie gives you an idea where it originated along the vertebral column, and from there it is a matter of a few minutes of trial and error to get each cookie to lock into place.
Even though we will not re-assemble this whale into an articulated skeleton, we do want to know the order of each bone in the body, and make sure we have cookies and vertebrae properly matched. Ultimately this skeleton will be stored in our mammal research collection and will be available for researchers to examine.
This whale at 9 meters was a few meters short of the adult size – at birth they are 4 – 4.6 meters according to John Ford’s new book (Marine Mammals of British Columbia). Its unfused epiphyses certainly show that this whale was young, and several ribs had abnormal bone growth. We also have a Killer Whale in the RBCM collection with similar abnormal bone growth. It makes me wonder if the odd growth caused pain and whether it was a contributing factor in this animals’ death.
 This sure was a neat way to end a work-week. It is not every day you get to assemble a whale’s skeleton. Now I have to say a HUGE thanks to Kate Kerr and Jana Stefan from our Exhibit Arts Department. When this whale washed up and we were offered the carcass for our collection, Nick Panter had just retired from his position as our mammal preparator. I was on parental leave.
This sure was a neat way to end a work-week. It is not every day you get to assemble a whale’s skeleton. Now I have to say a HUGE thanks to Kate Kerr and Jana Stefan from our Exhibit Arts Department. When this whale washed up and we were offered the carcass for our collection, Nick Panter had just retired from his position as our mammal preparator. I was on parental leave.
In our place, Kate and Jana were sent to help remove flesh from the carcass – you have to admire them for having the gastro-intestinal fortitude to see such work through to completion. I am betting that Shane Lighter, the museum photographer plied his trade from a respectable distance – upwind. It took hours of de-fleshing, and over 6 months buried in compost, followed by months of drying to get the skeleton to its present state. Kate happened to walk by when I was unloading the skeleton, and commented on how much nicer the whale smelled compared to the last time she had seen it.
Now I am back from parental leave, so I won’t have any excuses the next time a whale washes up. I’d better practice my whale flensing work song. The next marine mammal is already waiting in the freezer – a young Dall’s Porpoise. Once veterinarians do a formal necropsy, I will remove most of the muscle and get it ready for our newly minted mammal preparator. He will have the fun of either putting the bones in the dermestid beetle colony, or perhaps he will boil the bones to remove the last bits of meat. After that, we have a Northern Right Whale Dolphin to prepare.
More information on the Humpback and all other cetaceans in BC can be found in John Ford’s new book on Marine mammals of BC
Our contractor Sam has taken on the tedious task of separating negatives and prints in the Goertz Studio fonds, described as PR-1021.
This accession came to us in 1969 with a sad and interesting story. Helmuth Goertz was a well-known Victoria photographer from 1949 to 1956. In 1956 he and his family left for England to explore new prospects. While crossing to Spain on their yacht, they were caught in a storm and the ship was wrecked. Helmuth, his wife and son were drowned. His daughter survived and returned to Canada.
He had left his Victoria photographs behind with Campbell Studios of Victoria and in 1969, Campbell Studios donated them to the Provincial Archives of British Columbia. They were accessioned as 198403-006.
The majority of the photographs are studio shots of individuals and each file may include several negatives and proofs from a sitting. Goertz kept an order book, assigning a chronological number to each new sitting.
Some of the images show evidence of touch up work and hand colouring like this one of Miss K. Thompson.
There’s also an example of the fun things you could do with a photograph, a jigsaw and a wooden stand; we all enjoyed looking at the little hockey player.
At the end of the project, we plan to freeze the negatives to extend their life, while keeping the prints onsite for access.
Quesnel Dam bridge post
In the Public Works annual report of 1924, you can find the following information about work that took place the previous year in district no. 9, 150 Mile House.
On the subject of bridges G.G. Mackay writes “The most important work was the completion of Quesnel Dam Bridge, the total cost of which was as follows: demolishing section of dam, $2,498.28; building bridge $19,598.58; constructing road approaches $6,590.25; total $28,687.11.”
The area once known as Quesnel Dam is now called Likely and it’s been in the news a lot lately because of the Mount Polley tailings pond breach.
Photographs of the work in progress on the bridge are in GR-3293, Public Works engineering photographs, file AAAE2706.
54-1-1.5 Quesnel Dam bridge May 31, 1923
54-1-1.6 Cariboo district Quesnel Dam bridge 5 July 1923
54-1-1.4 Quesnel Dam bridge fill at North approach May 31, 1923
54-1-1.2 Quesnel Dam bridge from South abutment 21 April 1923
54-1-1.7 Cariboo district Quesnel Dam bridge 5 July 1923
This last photograph is a homemade panorama. Three photographs have been cut and taped together to provide a complete side view of the bridge.
Pre-Emption Government Records GR0112
Pre-emption was a method of acquiring provincial Crown land by claiming it for settlement and agricultural purposes. It was also a method of selling Crown land which had not been fully surveyed, designed to quickly provide temporary title or permission to occupy land to incoming settlers, for purposes of building a homestead and commencing agriculture.
The process was formally established under the 1870 Land Ordinance (although the earliest pre-emptions began in 1859) and was terminated by the 1970 Land Act. Under this process, individuals, as well as companies and partnerships, could purchase land, but grants to this land were not issued until specified improvements and residency requirements had been completed, and the land had been fully surveyed.
Pre-emption records are arranged by district and were transferred to BC Archives from the Department of Lands, Forests and Water Resources in 1976.
These record books were at times referred to as “Gum Stub” or “Guard” books. Documents were placed into the book, a gummed strip of brown paper adhered on top, and subsequent layers of documents positioned upon the next until the final document of a Crown Grant was issued.
 Crown land pre-emption record book
Crown land pre-emption record book
Information on many pages in these record books are obscured by a gummed strip, the pages can be brittle and difficult to turn. The risk of damage to the fragile documents is high.
 Gummed brown strips obscuring information
Gummed brown strips obscuring information
To prevent further damage and associated information loss caused by age and client handling, preservation staff have removed the glued brown paper gummed stubs to reveal information previously hidden by these, or covered over when the documents were adhered to one another.
 Records after treatment to remove gummed brown strips
Records after treatment to remove gummed brown strips
These documents are now available for use by genealogists as well as people researching pre-emption records for land use issues such as First Nations land claims, environmental assessments, historical land use policy and legal disputes.
In 1930-1931 work took place on the Kitsault River in Alice Arm. According to the 1931 report of the Chief Engineer “in the Atlin district, breakwaters were completed to protect the town of Stewart from encroachment by the Bear River and the town of Alice Arm from the outflow of the Kitsault River.”
Here are some more photographs from GR-3293. These ones are from file AAAE4527, in container 000705-0010.
98-44-2.1(9) to 98-44-2.3(11) Engineering dist.#7, 1930-21 report. Protection work, Kitsault River, Alice Arm
Now Alice Arm and the town of Kitsault are considered to be ghost towns with just a few residents left, but in 1931 it was going concern.
Here’s an image clipped from our website showing Alice Arm sometime in the 1930’s.
I once had a Cuban Anole (Anolis equestris) as a pet when I was in high school – I still have his skull. Yea I know, it is creepy to have your pet’s head on a shelf. But we are approaching Halloween and at this time of year, creepy is cool.
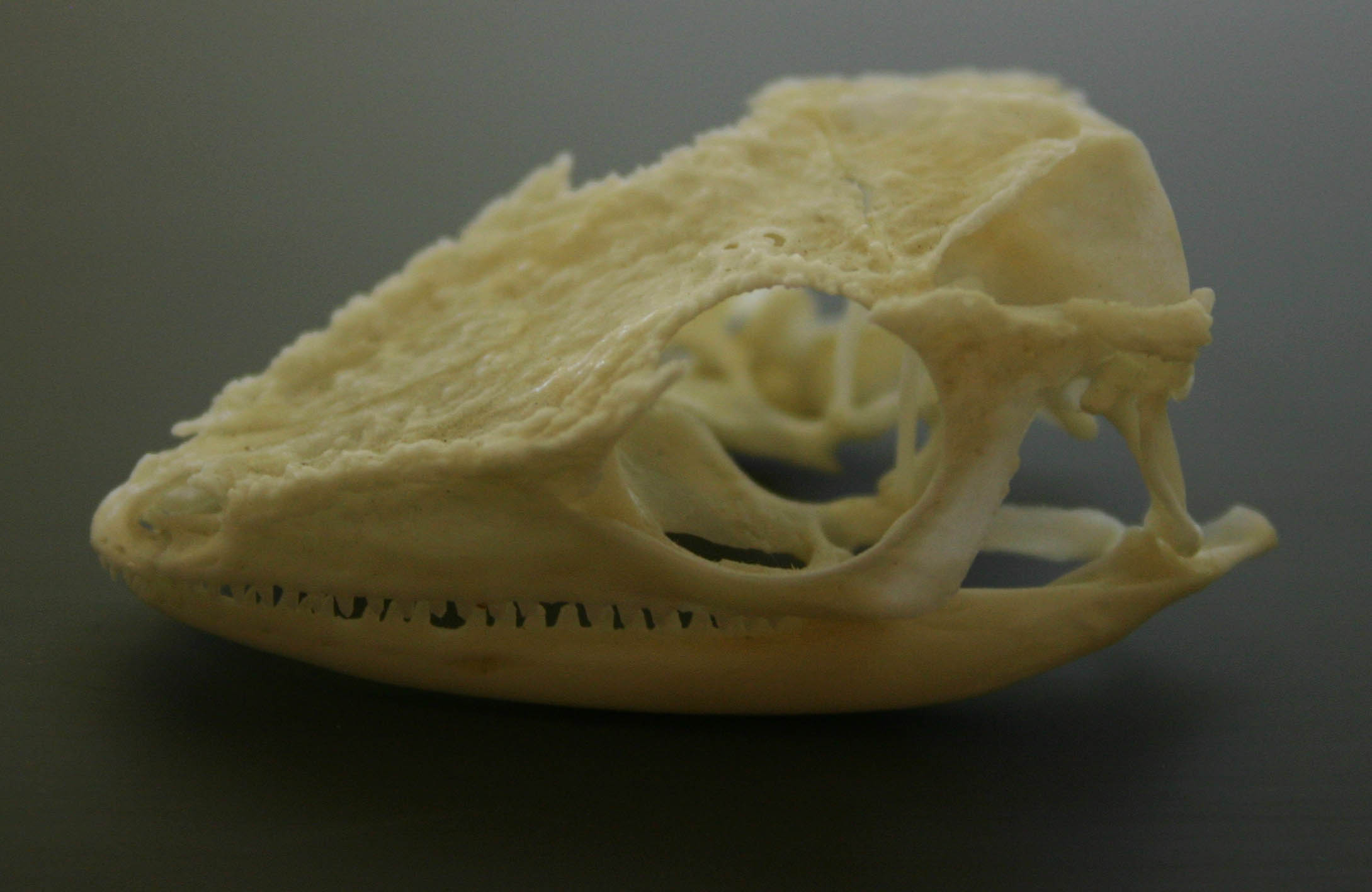 A photograph of the skull of my old Cuban Anole (Anolis equestris).
A photograph of the skull of my old Cuban Anole (Anolis equestris).
I was reminded of my old pet because of an article which recently appeared in the scientific journal Nature (Helmus et al. 2014). In this article, the authors show that island biogeography – where species disperse and diversify based on island size and isolation – has been overturned in the Caribbean by economic processes. In the Caribbean, anoles have dispersed far and wide as contaminants with cargo of one sort or another. Several species have colonised new islands as stow-aways with cargo, or as fugitives from the pet trade. The sharp increase in anole dispersal began around WWII, and increased significantly after the “cold war” thawed.
 Brown Anole (Anolis sagrei) in Florida (photo by Marlene Gifford).
Brown Anole (Anolis sagrei) in Florida (photo by Marlene Gifford).
Cuba stands out (Figure 1) as a large island with diverse habitat that is not geographically isolated, but has not been invaded by hordes of rampaging anoles. The authors of this new paper suggest that the isolation of Cuba is economic, and there are few chances for stow-away lizards to get a toe-hold on that island. However, lizards were able to leave. Cuban Anoles have invaded islands on Great Bahama and Little Bahama banks, several locations on mainland Florida, and as far west as Oahu. The Brown Anole, also from Cuba, has invaded Belize, the Cayman Islands, southern Georgia, Texas, Alabama, Louisiana, California and Hawaii. Obviously these multiple exotic occurrences do not represent repeated independent dispersal events from Cuba, but they almost certainly represent translocation of anoles by commercial activity (the pet trade, gardening industry, or fruit shipments).
 One of our Wall Lizards (Podarcis muralis) in Saanich.
One of our Wall Lizards (Podarcis muralis) in Saanich.
Compared to Cuba, our island is quite isolated as far as reptiles are concerned (and a touch cooler for much of the year). The water surrounding Vancouver Island is cold, tides are strong, and there is little chance that a lizard could raft over from the mainland. But like the Caribbean, the probability that exotic lizards move with human help is 1. Natural island biogeography is circumvented when animals are brought here intentionally, and released. Luckily most pet lizards escape or are released one at a time and never find mates. Within the Saanich area though, European Wall Lizards disperse in shipments of hay, in livestock trailers, and possibly also in plant pots (as eggs) or crates of produce. While our exotic lizard first arrived here intentionally, recent dispersal on Vancouver Island is increasing due to commerce. Perhaps the next bale of hay my wife buys for garden mulch, will explode with lizards when the twine is cut.
Who knows, if climate warms significantly, even anoles could colonise our island in the Pacific.
For more reading:
Lever, C. 2003. Naturalized reptiles and amphibians of the world. Oxford University Press, New York. 318p.
Matthew R. Helmus, M.R., D.L. Mahler & J.B. Losos. 2014. Island biogeography of the Anthropocene. Nature 513: 543–546.
I have now been interning in the Objects Conservation Lab at the Royal British Columbia Museum for a little over a month, where I am completing the final requirements for the Fleming College Cultural Heritage Conservation and Management program.
Every day in the lab is a new adventure. Some adventures are slightly less exciting, like vacuuming the litany of artifacts that don’t require a serious treatment but still need a clean-up. Other adventures turn out to be an exciting mystery to test your wits and exercise your fear of heights!
Last Tuesday started like any other. I had a little condition reporting planned, maybe some rehousing of objects in the history collection, no big plans. Then we got the call: An exhibit tech had noticed insects on the sea lions in the BC Seashore diorama. Kjerstin and I travelled upstairs to the galleries to check it out. We picked several dead beetles off of two of the sea lions, and a number of shed larva skins from a Pigeon Guillemot’s butt. On inspection, these were definitely examples of adult carpet beetles, and the shed larva skin of their young. Horrifying!
 Picking insects off the sea lion
Picking insects off the sea lion
We decided to launch a vacuum attack, with Exhibit Tech Megan and Kay from the Paper Conservation Lab, and after carefully donning nitrile gloves, dust masks, and lab coats (to protect us against dust, dead insect bits, and the potential of poisonous pesticide residues that sometimes accompany taxidermy specimens) we started to scour that diorama.
 Vacuuming the birds, perched on a ladder
Vacuuming the birds, perched on a ladder
We vacuumed every mammal and bird, and a good deal of the rocks and seashore in between. Some were high up on simulated rock cliffs, and I had to perch on a ladder to reach them! Most of the mounts in the exhibit turned out to be fine, and were untouched by insects. We did, however, find a number of adult carpet beetles on the sea lions, all dead. The only larva skins found were those on the Pigeon Guillemot, and although we also found some strange, well desiccated eggs on another bird, on consultation with Claudia the entomologist, it was decided that they were unrelated and probably very old.
Photos taken with Miscope ® Portable Digital Microscope
The mystery here is: Where did the carpet beetles come from? And how did they die? What were those eggs? The eggs are from an insect that would probably not harm the collection, and were most likely laid there a long time ago, perhaps by a fly attracted to the delicious smell of a dead pigeon. Carpet beetles are a more serious problem, and can do a lot of damage if left unchecked. Other dioramas in the same section of the RBCM have had issues with them in the past, and have dealt with them with success. The going hypothesis is that the larva hatched on the pigeon, and had a happy life there until they became adults. They then travelled to the sea lions, where for some reason they died. Because of this, the possibility of pesticide residue on the sea lions is again something to be wary of. Until they can be tested for contaminants, it will be important to wear gloves, a dust mask, and a lab coat when cleaning or examining them.
At the end of the day we could rest assured that the BC Seashore at the RBCM was safe from carpet beetles- at least for now!
Rock quarries post
Local rock quarries were used for more than just crushed rock for road building. The Public Works annual report for 1937 includes the report from Engineering district no. 7 by J.C. Brady. District no. 7 was Prince Rupert and it included Atlin and Stewart. In 1937 work was done on the Bear River at Stewart to reinforce the old groyne. This was protection project #20.
According to the report “the old groyne extends from the west end of Bear River Bridge to a point 3,360 feet in a southerly direction. The present work included the strengthening of the old groyne where necessary, and its extension in a south-westerly direction for 1,755 feet. A total of 8,580 cubic yards of rock was used. The average width of the wall or groyne is 16 feet at the base tapering to 6 feet at the top. Three hundred feet of ditching and excavation was also done.”
I found an online report from 1993, of work done by a contractor to the Ministry of Environment. The report makes interesting reading but I particularly enjoyed looking at the appendix of photographs starting on page 73. Photo 17 on page 81 shows the groyne and photo 18 may well be some of the ditches mentioned in the 1937 report.
What interested me the most though, was the figure of 8,580 cubic yards of rock used in the 1937 strengthening of the old groyne. Here are some photographs from GR-3293, file AAAE4494, in container 000705-0010.
98-35-1.1 No. 15 Bear River protection project #20, rock quarry October 1937 after wall shot down and prior to breaking up large rocks, Stewart BC
98-35-1.2 No. 16 Bear River protection project #20 rock quarry October 1937 showing height of quarry, Stewart BC
Note the size of the man compared to the rock.
I’m not sure what kind of equipment they used to break up the large rocks as the equipment inventory for that year doesn’t list any rock crushers in the Atlin district.
My partner and I recently celebrated our anniversary with a trip to San Francisco. It happened that our anniversary was on the first Thursday of the month, which is also when the Exploratorium hosts “After Dark” – an adult evening program which features speakers, extra activities in addition to the amazingly hands-on experiences they already offer, cash bar and music. Luckily, my partner is a museum fan too, so the idea of spending our anniversary at a museum event appealed to both of us.
The Exploratorium has been an example of best practice in museums for many years so my expectations were high. I was not disappointed. By attending in the evening, we had full access to every activity without having to negotiate the younger visitors who frequently visit. Being in an all adult environment, we also felt like we had “permission” to play, something that I don’t always feel like I am getting when visiting a museum. Please don’t think that I am an advocate for “child-free museums” – as a provocative post did recently – but I did feel more at ease and willing to try everything I could during the evening program.
What I appreciated most were the number of interactives that were designed to play with another person. Visiting museums is often social, but the interactives are not always designed to be used by more than one person at a time. The Exploratorium made it explicit that you needed to try this activity with someone else and often we both had a different role in the activity. This encouraged us to switch sides and try it again and then discuss what we experienced.
There were also a variety of ways to interact with a theme. Instead of one interactive about magnets there were probably a half dozen. The advantage of this was that I could try the one that appealed to me most or try them all, a great technique to appeal to different styles of learning.
With so much to see and do, we spent four hours at the museum – a new record for me . The experience was living proof to me that interactives can increase enjoyment, learning and social interaction making the museum visit more engaging .
The Rock Crusher post
Imagine producing crushed rock for roads and other public works before the machine age…
J.P. Campbell, General Foreman for Islands and Saanich district shows his appreciation for machinery in the 1925 annual report. First he notes that “an air-compressor and rock-crusher were installed at North Saanich and are giving entire satisfaction and producing crushed rock at a low cost.” Near the end of his short report, which covers public works activities in his district, he states “I would like to emphasize the fact that to the modern machinery engaged is due a very large percentage of efficiency obtained.”
And it’s no surprise when you see the size of the rocks they were dealing with.
In GR-3293, file AAAD3058 you can see the following photographs:
1-32-1.1 Quarry East Saanich Road April 1925
1-32-1.2 Rock Quarry East Saanich Road April 1925
I’ve always liked the portrait of Dave Barrett that hangs in the hall of Premiers in the Legislative Assembly buildings in Victoria. When I go over for lunch in the legislative dining room and I pass through the hall, I sometimes stop to admire the portraits of the Premiers of BC. Most of the portraits are quite formal black and white photographs, until you get to Dave Barrett. Suddenly you are confronted with a colour photograph of a smiling Barrett with a halo or sunrise effect behind him. I have wondered about the origins of this photograph and why it is so different from the others.
The other morning I was viewing a CBC video of the opening of the Legislature, 2nd session, 30th parliament in January 1973. Jack Wasserman of the CBC talked about the Barrett portrait and said that the photograph had been taken by staff of Beautiful British Columbia magazine who had rushed over to Barrett’s house after the surprise election of the NDP. Barrett was apparently in his shorts but pulled on a shirt, sports jacket and tie and posed in front of a painting, created by his wife Shirley. In “Barrett, a passionate political life” by Dave Barrett & William Miller published in 1995, Barrett notes that the photograph was taken by Gar Lunney and confirms that it was taken early in the morning after the night of the election, and that he was hung-over and in his underwear.
The photograph on our website, E-03014, is a black and white copy. It was given to the BC Archives around 1974 by a Mr. Protheroe. I don’t know if we have the original colour negative but anyone visiting the Legislative Assembly buildings can see and enjoy the colour portrait hanging in the hall of Premiers.
UPDATE APRIL 2016. I am happy to report that I have located the original colour print, given to the Archives by Mr. E. Protheroe (probably civil servant Ernest Protheroe). Our photographer Shane has made a beautiful copy of it so I have reloaded the colour image in our Atom catalogue, still under photo reference number E-03014.
Office of the Apes post
Sometimes I’m lucky enough to come across bizarre files that provide lots of scope for speculation.
I’ve spent a lot of time with a series of government photos called “Film and photographic branch travel industry and tourism photographs” described as GR-3425. This series, which consists of over 56,000 prints, negatives and slides, was created by Government between 1943 and 1985. Staff photographers were responsible for creating images to meet any kind of government need from travel brochures to annual reports to publicity shots. The photographs were catalogued and used in a variety of ways and eventually ended up as a large photo bank or image library managed by the Government Information Services. When the GIS dissolved in 1988, the photographs were transferred to the BC Archives.
Because we want to preserve the negatives and colour slides, we’ve had to painstakingly remove the negatives from each file, rehouse them into acid free envelopes and record the photograph number on each one. This job has taken four years of staff time as well as using the partial services of interns, co-op students and contract staff. There are multiple “runs” of numbers, each with their own meaning and as always with large groups of records; there are the unclassified miscellaneous photographs that we tend to file at the end of the run.
One of these unclassified miscellaneous files in container 002340-0491 is called only “Mr. Gallardi’s chimp, 3/57”. Mr. Gallardi refers to Dept. of Highways Minister Phil Gaglardi. Inside the envelope are four black and white prints, their accompanying negatives have been carefully rehoused.
All we know is that the setting appears to be Gaglardi’s office in the Legislative buildings and that the photographs were taken in March 1957 by a government photographer. After that we can only speculate….
What was life like in British Columbia during the time of the Great War?
Several collections in the BC Archives are available to tell some of that story.
Let’s start with some reasonably new technology.
You would need to get a license for your car.
 And register the license with the BC Motor Vehicle Branch (GR-0665 Vol. 22).
And register the license with the BC Motor Vehicle Branch (GR-0665 Vol. 22).
The License was issued to the Vehicle and any change of ownership was tracked through these registers.
Of course, you could then join with like-minded folks and go on car rallies.
Or join a parade in Kamloops.
While there, you could take in the May Day festivities.
From Kamloops you could head over to Nakusp and help Hendrick Aalten clear the right of way for the Canadian Pacific Railway.
You could then try another form of transportation as you climb aboard a speeder on the tracks of the Pacific Great Eastern Railway.
That form of transportation just might bring you to the Victoria terminus of the Esquimalt and Nanaimo Railway.
While you were in Victoria you could visit Dr.Helmcken at his residence on what are now the grounds of the Royal BC Museum.
The winter of 1916, would not have been a convenient time to visit though.
One of the shop windows is numbered “82”
Page 7 of the 1903 Victoria Fire Insurance maps shows that “82 Douglas Street” was reassigned as 1304 Douglas Street and is located just north of the Yates Street intersection.
Across the Harbour a Commission was taking place investigating “all matters affecting the value of lands of the former Songhees Reserve”.
If anyone was injured in clearing that property, An Order In Council signed off by Premier Bowser, was required.
Money also needed to be approved for large infra-structure projects such as sewers.
Or in house improvements.
Elsewhere on the home front, the Women’s Institute movement was opening up throughout BC.
The BC Archives holds several collections of minute books from these organizations from throughout the province
Institute members were active in promoting the creation of libraries.
Or the promotion of urban gardening on city owned property.
Elsewhere, the Girl Guides were busy gathering information on how to receive the Laundress badge.
Meanwhile, business proceeded apace.
While the provincial government ensured that the motion picture industry met the standards of the day.
For more information about life during this era (or any other time…) please visit the BC Archives on line or in person.
Needles and Pins, Needles and Pins, When You Get Married, Your Trouble Begins. By Colleen Wilson post
One of the principles of good conservation is that the work should be reversible. Although there are times when a fabric is so shattered that an adhesive patch is considered, that is a treatment of last resort. Glues tend to change over time becoming discoloured, rigid, and very difficult to remove.

soft sculpture, tapestry (#’s 6 and 28), beading, curved needles (for surgery and carpet) and almost 1,000 #12 sharps
So the work of a textile conservator frequently involves sewing. If a heavy weaving is to be displayed, it needs both an overall support so that it won’t sag under its own weight, and some means of holding it up so it won’t droop and distort. To prevent the yarns of the weaving (or hooked rug or blanket) being split, a large blunt needle is used to stitch between the threads. A hole or damaged area in fragile, brittle silk can be reinforced with a patch placed behind, but only the finest, sharpest needle can pull the thread without causing more damage. Replacing missing stitches in a three dimensional object can mean working in the awkward corners of a structure (like a hat or tent); using a curved needle is sometimes the only way to place the stitches through the original holes.
Pins are a temporary hold. They can keep two fabrics in place until stitching is complete, and they can hold wet fabric in place while it is drying. In both cases, they must be fine enough not to make permanent holes in the fabric. For blocking a washed textile, pins with large smooth heads are easier on the fingers – a lace shawl or crocheted bedspread require a lot of pinning to ensure they dry square.
If you are married to Conservation finding the right tools for the job can be troublesome indeed.
When patching a textile artifact to fill a hole, reinforce a weak area or consolidate unravelling yarns, couching is the technique of choice. We want to hold the patch to the artifact securely but with a minimum number of punctures by the needle. And, reversibility being an important principle of conservation, we want to be able to remove the patch without risk to the artifact.
Couching holds the two fabrics together without obscuring the artifact, or putting much strain on the aged threads; darning or “invisible mending” (darning with threads unraveled from the damaged cloth) are far too invasive. With the patching fabric behind, a long thread is laid on the face of the artifact perpendicular to the damage, but aligned with either the warp or weft threads. Then tiny stitches are made over the laid thread to hold it in place. Should the repair be removed in the future (because the patch has faded and no longer matches the artifact or because the patch is obscuring some information on the back of the textile or a hole is now believed to be more interesting than the appearance of completeness) the stitches holding the laid thread can be snipped at the back, the scissors only approaching the patch, not the artifact, and the laid thread lifted off the front.
A dress from 1855-60 is being prepared for inclusion in the Gold Rush exhibit. This was a period in women’s fashions when skirts were extremely full with yards of fabric displayed over cage crinolines. This dress was worn as a costume for many, many years and the brilliantly coloured plaid silk is in very poor condition. There are many miles of couching thread to be laid before the dress will be ready for exhibit – if it wasn’t so exciting, it would be enough to turn a conservator into a vegetable.
We recently had need to move a locked file cabinet to make desk space for volunteers – the keys to the cabinet were missing. A short while later – and some lock-picking – revealed folders of exquisite fish art. The art had been gathered together 6-7 years ago and placed in the file cabinet for safe-keeping. Perhaps we should have disabled the lock when we originally stashed the art in the cabinet – hindsight is 20-20. Now the art is stored in my lab in a filing cabinet (with a deactivated lock).
Below is a sample of some of the work with each artist’s name underneath. The art is protected by plastic – so some photos will show reflections. These photos were shot quickly in my lab for this blog article – and certainly not to the usual standards of Shane Lighter, our Digital Imaging Technician/ photographer.
 1) line drawing of a Coryphaenoides species prior to charcoal dust and highlights; by Norman Eyolfson
1) line drawing of a Coryphaenoides species prior to charcoal dust and highlights; by Norman Eyolfson
 2) Arctic Shanny, Stichaeus punctatus; by Norman Eyolfson
2) Arctic Shanny, Stichaeus punctatus; by Norman Eyolfson
 3) Atka mackerel, Pleurogrammus monopterygius; by Norman Eyolfson
3) Atka mackerel, Pleurogrammus monopterygius; by Norman Eyolfson
 4) Smootheye Poacher, Xeneretmus leiops; by Norman Eyolfson
4) Smootheye Poacher, Xeneretmus leiops; by Norman Eyolfson
 5) Black Skate, Bathyraja trachura; by Norman Eyolfson
5) Black Skate, Bathyraja trachura; by Norman Eyolfson
 6) Slimy Snailfish, Liparis mucosus; by Norman Eyolfson
6) Slimy Snailfish, Liparis mucosus; by Norman Eyolfson
 7) Matcheek Warbonnet, Chirolophis tarsodes; by Patricia Drukker-Brammall
7) Matcheek Warbonnet, Chirolophis tarsodes; by Patricia Drukker-Brammall
 8) Arrowtail, Melanonus zugmeyeri; by Patricia Drukker-Brammall
8) Arrowtail, Melanonus zugmeyeri; by Patricia Drukker-Brammall
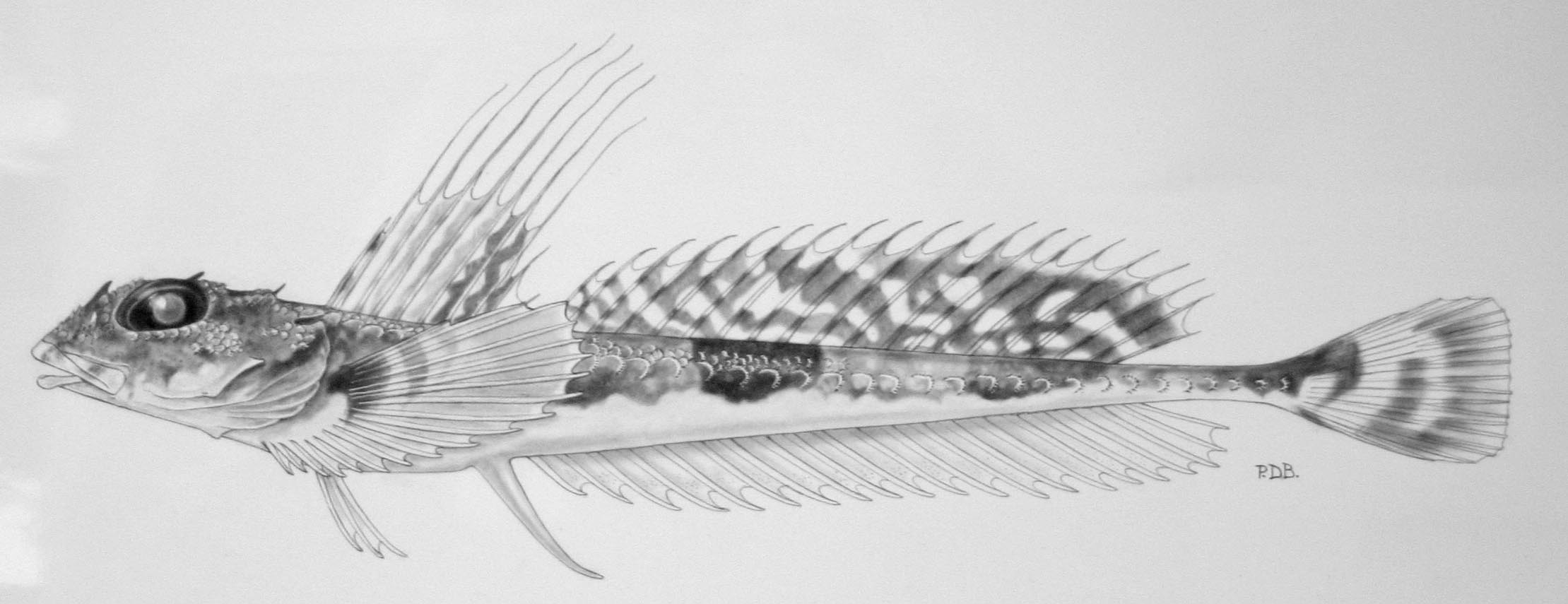 9) Darter Sculpin, Radulinus boleoides; by Patricia Drukker-Brammall
9) Darter Sculpin, Radulinus boleoides; by Patricia Drukker-Brammall
 10) Looseskin Eelpout, Taranetzella lyoderma; by Kathy Schuster
10) Looseskin Eelpout, Taranetzella lyoderma; by Kathy Schuster
 11) Shortmast Sculpin, Nautichthys robustus; by Karen Uldall-Ekman
11) Shortmast Sculpin, Nautichthys robustus; by Karen Uldall-Ekman
 12) Leister Sculpin, Enophrys lucasi; by Karen Uldall-Ekman
12) Leister Sculpin, Enophrys lucasi; by Karen Uldall-Ekman
Here are some dace – used in manuscripts to highlight the presence and range of Rhinichthys in the Columbia River basin.
 13) Leopard Dace, Rhinichthys falcatus; by Karen Uldall-Ekman & Patricia Drukker-Brammall
13) Leopard Dace, Rhinichthys falcatus; by Karen Uldall-Ekman & Patricia Drukker-Brammall
 14) Speckled Dace, Rhinichthys osculus; by Karen Uldall-Ekman & Patricia Drukker-Brammall
14) Speckled Dace, Rhinichthys osculus; by Karen Uldall-Ekman & Patricia Drukker-Brammall
 15) Umatilla Dace, Rhinichthys umatilla (Kettle River); by Karen Uldall-Ekman & Patricia Drukker-Brammall
15) Umatilla Dace, Rhinichthys umatilla (Kettle River); by Karen Uldall-Ekman & Patricia Drukker-Brammall
 16) Umatilla Dace, Rhinichthys umatilla (Otter Creek); by Karen Uldall-Ekman & Patricia Drukker-Brammall
16) Umatilla Dace, Rhinichthys umatilla (Otter Creek); by Karen Uldall-Ekman & Patricia Drukker-Brammall
The work was done using a clay coated surface with carbon dust brushed on. Light coloured highlights appear to have been created by scratching carbon dust away.
Some of this art has been used in publications by Alex Peden (my predecessor here at the RBCM), but Catherine Mecklenburg also used some of the art in her book:
Mecklenburg, C.W., T.A. Mecklenburg, & L.K. Thorsteinson. 2002. Fishes of Alaska. American Fisheries Society, Bethesda, 1037 p.
I can only assume these drawings were stock-piled in preparation for a book on Marine Fishes of BC, but for now, this book is way off in the future. For now we are publishing records of fishes from a few families at a time to get our marine records updated. There are at least 44 species with significant range records or representing first records for BC. Any book on Marine Fishes of BC would be hopelessly premature until we get our series of peer-reviewed papers to print.
I was reading the Dept. of Public Works annual report for 1929 (an enjoyable Friday afternoon occupation) and saw that G.T. Mitchell, General Foreman, Islands District reported the following:
“In December [1929] the district office was removed from Thoburn to Sidney, where a four-roomed modern office building was erected on Government land at the junction of Second Street and Bazan Avenue.”
Where was Thoburn? I couldn’t find an area or a road of that name in Google maps so I searched the BC Geographic place names database and found the entry for the rescinded name “Thoburn”. It referred to a post office site in Esquimalt. Could this be where the Public Works Islands district office was located before December 1929?
http://apps.gov.bc.ca/pub/bcgnws/names/50786.html
I also checked our old Provincial Archives of British Columbia map collection BC place names file and found the following information under the name Thoburn: P.O. on SE corner of Esquimalt & Head Streets, Esquimalt.
I looked at a fire insurance plan for Esquimalt and various city and telephone directories but none of them led me to a Public Works district office in that area.
I was more successful with Sidney. Although current maps don’t list a Bazan Avenue within the town itself, I consulted a blueprint plan of Sidney from the Archives map collection and found that what is now called Bevan Avenue was once called Bazan Avenue and it does cross Second Street. The plan I looked at, CM/C320, is called “Map of Sidney, BC, drawn & compiled from official plans and sources by S.C. Weston, Victoria BC.” It is undated but we have other plans drawn by Weston in the teens and twenties.
At any rate, in July of that same year, 1929, the Public Works Dept. spent $313.81 on the paving and maintenance of Beacon Avenue. Here are some photographs from GR-3293, file AAAD2352:
Photo 1-13-4.4 showing wharf and road to ferry at right
Photo 1-13-4.3 showing widening of paving and new concrete curbing
I like to think of the Public Works staff eating doughnuts at the Sidney Bakery or enjoying a bicycle ride on the newly paved street.
Last May it was time to calibrate the Museum’s electronic environmental data loggers, little boxes attached to walls of collections storage and exhibit areas that record temperature and relative humidity. In the pictures below, you can see the myriad of cables, electrical cords and equipment assembled for the operation. It looks like mayhem, but in fact, it was all under control. In the background, you see a plastic humidity tent, which was used to create an environment separate from that in the conservation lab. Humidity was created by placing hot water in a shallow pan. This is all that’s needed in a closed environment to create humid conditions (remember that the next time you leave standing water around your house). The data logger probes were placed into the humidity tent and the readings recorded by the loggers, which were plugged into the laptop computer in the foreground. Each logger requires a power source as well as a network cable, hence the mess of interwoven cords. I’m sure our textile conservators would have preferred that we neatly plait them or otherwise create a pleasing pattern.
In the end, the loggers were all adjusted to read the same temperature and RH as the one created in the humidity tent, thereby calibrating them to the same standard. This is so very important, as we use these loggers to monitor the environments in our storage and exhibit areas, to promote preservation of the collections. They even send us email alarms if things go awry. If the temperature in a storage area gets too high, chemical reactions speed up that can cause damage, such as hardening of rubber and leather artifacts. If the relative humidity in an exhibit area gets too high, mold may disfigure collections or cause metals to corrode. Low RH causes glues to fail and mounted animal specimens to split. It’s the job of the conservators to ensure the best preservation environments for our world class collections.
A Glove Story post
Have you lost that glovin’ feelin’? Or more to the point, do you lose some tactile sensitivity or manual dexterity when you use gloves? Many archivists and archival researchers report this problem when they use gloves. That’s why we don’t always require that gloves are worn when handling historic records in the Archives Reference Room. It has been shown that more damage can be done when researchers try to turn pages with gloved fingers than if they simply use clean, bare hands.
Of course there are some exceptions, such as when handling photographs, which can be irreparably damaged by bare hands. Some objects in the Museum’s collections must also be handled with gloves all of the time. This includes anything made of metal, which can corrode from the acidic oils in our hands and natural history specimens that may once have been treated with nasty pesticides and so now present a health risk. Even in the Royal BC Museum, though, there are some objects that should not be handled with gloves. This includes very slippery surfaces, such as glass and glazed ceramics and fragile basketry, with small brittle fibres that can catch on gloves. In these cases cotton gloves are just too risky.
Of course, nitrile gloves are acceptable, but some people find them to be uncomfortable. Occasionally, museum conservators have been known to use mittens, in this case to prepare wheat starch paste for mending documents.
Did you know that museum workers now tend to use nitrile gloves instead of latex when they need to keep a good barrier between their hands and an object? This is because of the very common latex allergies people have. Even if you don’t currently have a latex allergy, you can develop one from continual exposure to latex.
So if you’re working in a museum or archives and you’re wondering “when will I be gloved?” just assume that gloves are worn whenever museum or archival collections are handled, unless you are advised otherwise.
Flood damage post
After several months of hot sunny weather, we finally had some rain in Victoria this week. I started thinking about floods and the kinds of situations that would precipitate one. While it’s pretty unusual for Victoria to have such long hot spells, sudden heavy rain on dry ground can cause flooding. It’s more usual for us to have the kind of flooding that comes from long term steady rain and melting snow, the kind that raises the levels of rivers and other watercourses. When combined with poor or blocked drains and frozen ground, this kind of flooding can seriously damage roads and bridges.
According to the 1935 annual report of the Dept. of Public Works, the winter of 1934-1935 was an unusually cold one on Southern Vancouver Island, with heavy snow throughout December and January. In mid-January, the temperature soared and the snow changed to heavy rain. The result was a flood at Jordan River which resulted in the loss of the bridge. This photo is from a series of engineering photographs created by the Dept. of Public Works; GR-3293, file AAAD3731, number 2-1-2.
Another rain storm near the end of February caused serious road flooding near Duncan. These photos are from GR-3293, file AAAD3288.
The top one is numbered 1-40-1.3 and on the back of the print it says “Storm Feb 26-27, look south (towards Victoria) from point ½ m north of tennis courts”
The bottom one is numbered 1-40-1.4 and on the back it says “At tennis court corner looking north, max depth of water on road was 3’! All other roads giving access to Duncan from S. were under water to some extent”

Week #16- Ann ten Cate- Archivist
Text to come…

Week #17- Claire Gilbert- Archivist
Text to come…

Week #22- Claudia Copley, Collection Manager of Entomology
Text to come…

Week #23- Erica Wheeler, Collection Manager of Botany

Week #28- Professor Jack Lohman, CEO
Text to come…

Week #29- Marji Johns, Collection Manager of Paleontology
Text to come…
The Golden-Yoho Road post
Here are some more images from GR-3295, folder 2. The portion of the Trans-Canada Highway that runs from Golden to Yoho National Park was worked on by the Dept. of Public Works (on contract to the Government of Canada) between 1924 and 1927. This album contains photographs embossed with the name W.J. Oliver, a professional photographer based in Alberta.
The quality of these prints is surprisingly poor. I don’t know if it’s just age or if they weren’t properly processed in 1927. Maybe it’s the acidic paper in the album? I’ve interleaved the pages with acid free paper to give a little protection but we can’t bring them back.
Views on Golden Yoho Road
A good spot to pull over.













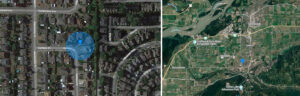




























































![Whale-011[1]](http://staff.royalbcmuseum.bc.ca/wp-content/uploads/2015/09/Whale-0111.jpg)
![p1090693[1]](http://staff.royalbcmuseum.bc.ca/wp-content/uploads/2015/09/p10906931.jpg)