Natural History Curators
Lizard Lovin post
‘Tis the season: northern alligator lizards are busy making little lizards.
Have you seen alligator lizards mating? If so, please take note how long they spend together. Males bite the head and neck of females and hang on, sometimes for over 24 hours. Mating itself doesn’t take 24 hours—so perhaps males hang on to make sure other males are excluded.

In BC, the alligator lizards tend to vanish when new urban developments spread across the landscape—maybe (probably) the habitat is unsuitable, roadkill happens, a certain invasive lizard certainly doesn’t help, and well-fed domestic cats also take their toll, as do lawn mowers and weedwackers.

But if you are lucky enough to have northern alligator lizards in your garden, please take detailed notes on their activity, prey, and how they use your garden structures. It’s also helpful to record mating activity and how long a pair stays together. Community science (sometimes called citizen science) is a way to get very detailed views of species distributions to see how species are responding to our increasing sprawl.

Greg Pauly, curator of herpetology at the Natural History Museum of Los Angeles County, studies natural history, evolution and conservation of reptiles and amphibians, and the impact of urbanization on biodiversity.
You can share photos and information with Greg at gpauly@nhm.org.
Here are a few of his blog posts:
Love in the Time of Coronavirus: The Alligator Lizard Version
Look Out for Amorous Alligator Lizards
Studying Lizard Love Through Citizen Science
Alligator Lizards: Mating season has begun – YouTube

A small portion of the RBCM Native Plant Garden.
The native plant garden at the Royal BC Museum, with more than 300 species of plants, is a haven for wildlife in an otherwise heavily urbanized setting. The garden was initiated in 1968 in part to promote landscaping with our diverse native flora. The diversity of bird species that are known to visit the garden is truly impressive considering the downtown setting, and the garden also harbours a wealth of invertebrate animals: butterflies, bees, dragonflies, and damselflies, as well as many others.
Some of the animals are just passing through on their migration – for instance the eye-catching Green Darner dragonflies that have been seen there in the fall on their way south (yes! -some dragonflies migrate!) and the gorgeous Wilson’s Warblers that pass through on their way north each spring. Others are year-round residents in the garden: Song Sparrows, Spotted Towhees, while others come to the garden in the winter months and then head off to points north as the rest of the province warms up: Golden-crowned Sparrow, Ruby-crowned Kinglet.
It is not just that the garden offers a patch of greenspace though. What makes the garden a preferred location for our native wildlife are the native plants that inhabit it. The plant species that are native to this region co-evolved with the animals that also live here, seasonally or permanently – they need each other. There is no better example of this then examining the butterfly species reported from the garden. They come to this small area because the plant that they lay their eggs on and that their caterpillar eats can be found in the garden. Some examples include willows for two of our local Swallowtail butterfly species, Ocean Spray for the Lorquin’s Admiral, and Sedum for Moss’s Elfin. In the fall elegant Cedar Waxwings are regularly seen gobbling up the Black Hawthorn fruits, as well as those of some of the other shrubs in the landscape. These fruits are a critical food source for birds that they cannot typically find in urban settings.
The winter garden has value as well: native trees and shrubs offer a year-round source of food to the birds of the region because many invertebrates live on the plants; providing a source of protein to insectivores like Chestnut-backed Chickadees, Red-breasted Nuthatches, and Bushtits. This past winter a flock of as many as fifty Yellow-rumped Warblers spent many months foraging in the garden – always able to find food among the regionally-adapted plants because of the insects feeding on them.
Delta Surprise post
I don’t expect to get new lizard range records in mid-November, especially since the weather has been so grey and cool. This month it seems we have had wind storm after wind storm. Our northern alligator lizard (Elgaria coerulea) should be down for winter—maybe an occasional one will appear on really warm days. Common wall lizards (Podarcis muralis) will emerge, but only when it is sunny. I have seen wall lizards active in sun-exposed locations when the air temperatures are only 5–7°C. But since I have been indoors, I have resorted to correcting United States lizard misidentifications on iNaturalist.
This Monday morning (November 16th, 2020) is grey and cold, and another wind storm is due tonight. But to my surprise, I received an email from Nicole Greenbaum at Paridon Horticultural Ltd. in Delta, BC. She found a lizard in the company’s tropical greenhouse and had correctly identified it as a western fence lizard (Sceloporus occidentalis). Nicole’s discovery is western fence lizard #2 for British Columbia (and Canada).
Canada’s second confirmed western fence lizard; photo by Nicole Greenbaum.
The greenhouse is heated throughout the winter, and so if they are not able to catch this little lizard, it will have quite the winter. Of course I have asked them to catch it to represent BC’s first specimen for the museum collection. Photos are useful, but a specimen is better.
Who wouldn’t want to live in a tropical greenhouse? Photo by Nicole Greenbaum.
This lizard is a juvenile, with a body just over 3 cm. From the photos, I can’t tell whether it is male or female. But the record is now in iNaturalist.
It is possible that a female laid eggs and even more are in the greenhouse, but most likely, this is a single stowaway that came in with a shipment of plants. We have no idea where it originated, nor when it arrived. The greenhouse has not received plants from the United States in some time (it is 2020 after all), so the lizard has likely lived there all summer. I suppose that is effective, pesticide-free insect control.
I wonder when the next one will make its way north of the United States border?
This is the month that the garden explodes with food—and lizards. Everywhere you look, there is food to harvest. We try to weigh everything to get an idea of the cash value in our garden, but sometimes you just have to eat things right off the vine.
Our tomatoes are ripening daily, with the Pineapple Cherry tomatoes at their delicious best now that we are nearing the end of August. Several of our tomatoes are not your typical variety: the Snow White ripens a pale cream-white colour, Black Prince are red-brown when ripe, and then we have the Green Zebra, which (as the name suggests) is striped green when ripe. They are all fantastic.
We have already started saving seeds for next year. The seeds are left in water for a few days to simulate the rotting fruit, then we drain and dry to store seeds over winter.
Zucchini, cucumbers, squash and melons are all producing fruit, though I don’t think the melons will amount to anything. Our pumpkin went nowhere. C’est la vie dans la grande ville. We will buy pumpkins to carve for Halloween and support a local grower in the process.
Cabbages are being converted to sauerkraut. Some cucumbers were grown specifically for pickles, others to eat straight up.
We have collected over 6 kg of blackberries, and our strawberries are still producing. The yellow raspberries produced just enough fruit for all three of us to enjoy a quick snack.
Apples are nearly ready. And if this wasn’t enough fruit, our neighbours have offered some pears—we just have to pop over and pick them.
A grey squirrel has figured out that our garden has loads of food and destroyed our mini-sunflowers. It also has been eating plums, leaving sticky scraps on our compost bins. That is part of gardening—sometimes you have to share. Wasps and house finches have nibbled on their fair share of the blackberries.
Even without our greenhouse—it got crushed by the snow last winter—we still grew some nice peppers, and the Italian parsley and New Zealand spinach are growing well. Carrots are our biggest failure; only a few grew.
Lizards have hatched—everywhere you look, a lizard the size of my pinkie finger darts off to hide in our rock walls and garden beds. The adults are still here too—but they seem to be less interested in basking in the midsummer’s sun. Perhaps the females don’t need to bask so much now since they are not carrying developing eggs.
Much of the work now shifts indoors. Berries and tomatoes are being frozen for winter use. Beans are being shelled and left to dry for winter chili, stews and soup. Onions are pulled and drying on the deck so they will store well. Only the Scarlet Runner Beans are still growing and will be picked later in the season.
The bean plants that are finished are now pulled and left on the ground to decay and add substance to the soil. We still need to harvest the potatoes in our front garden, and then in the next few weeks we will plant a cover crop. Once the cover crop has grown a few centimetres, we will cover it with a tarp to let worms and other recyclers enrich the soil.
And even while the harvest is in full swing, we are planting lettuce and kale starts for autumn and winter. It is amazing to be able to walk outside in winter and get fresh produce when you need it.
This has been a good summer for food production. We have hundreds of dollars in produce, with over $50 in blackberries alone at current market rates. We added oak-leaf mulch and some horse manure early in the season, watered when we needed to, and that is about it. Even a modest city garden can produce plenty of good clean food.
Hatchlings Happen post
I have been watching a female lizard in my garden slowly increase in girth as her eggs developed in May to early June. I recorded each day whether she was gravid or not, and noted when she lost her tail. Simple observational science is something I could do to better understand common wall lizard biology in BC while working from home.
My notes around the time I think eggs were laid went as follows:
June 5 – basking on rocks, 80% tail lost in the last few days, still looks gravid.
June 6 – not sighted
June 7 – not sighted
June 8 – not sighted
June 9 – rainy, cold, not sighted
June 10 – basking on rocks, has folds of skin along the flank suggesting eggs laid.
I have watched our resident female wall lizard as her tail regenerated, and I saw her again yesterday (July 30) sporting a tail several centimeters shorter than the original.
I patrol the garden daily to see what the lizards are doing, and when and where they are active. My wife will confirm this obsessive behaviour. But this morning (July 31) I dropped my daughter off at a day camp, and on return home, as I walked up my driveway, I spotted our first hatchling.
I gasped—I am not ashamed to admit it.
Lizards invaded our garden in 2019, and one year later we have a home-grown baby lizard. It looked pretty comfortable in its new habitat, so perhaps it hatched yesterday (July 30) or the day before. And there is no way to tell, without perhaps a series of DNA samples, whether this hatchling came from the female I have been studying, or whether some other female dug a nest in our garden. Young lizards leave their parents’ territory to avoid cannibalism, so this one may head for the hills. But it does give us an estimate of timing between egg deposition (somewhere between June 6–8, assuming eggs were not laid June 9 when it was cool and rainy) and the first appearance of hatchlings July 31.
Females mature in their second year and can have one to three clutches of eggs each summer, depending on latitude and local conditions, with clutches ranging from 2 to 10 eggs. In nature, incubation ranges from 6–11 weeks. Embryo development is about halfway at oviposition, and females bury eggs in sandy or crumbly substrates at the end of 10–20 cm tunnels (Van Damme et al. 1992). In a laboratory experiment, temperature dramatically changed incubation times, with wall lizards incubated at 32–35°C hatching 10 days earlier than lizards incubated at 28°C, and over five weeks earlier than those incubated at 24°C (Van Damme et al. 1992). We had a cool spring, and I am not surprised then that 50–52 days passed between the first evidence that the female had laid eggs and the appearance of a hatchling.
Hatching success is high in the 24–28°C incubation range, and Van Damme et al. (1992) suggested 28°C is the best trade-off between hatching success, incubation rate and hatchling health. I wonder how many more hatchlings will appear in the garden?
For more information:
Van Damme, R., D. Bauwens, F. Braña and R.F. Verhyen. “Incubation Temperature Differentially Affects Hatching Time, Egg Survival and Hatchling Performance in the Lizard Podarcis muralis.” Herpetologica 48, no. 2 (1992): 220–228.
Ooooohhh Barracuda post
It is 2020, and people are asking whether this year will get any stranger.
How about barracuda in BC waters? Does that qualify?
I received several emails and other messages today (July 10) noting that a 5.4 kg barracuda had been caught off Vancouver Island this week. This is a really cool record, and I hope it’s added to iNaturalist.

Pacific barracuda (Sphyraena argentea) from San Diego, California. Photo by Darren Baker, uploaded to Fishbase IMG-20120830-00071.jpg.
Pacific barracuda (Sphyraena argentea) are known to range all along our coast, and as Alaska-based biologist Scott Meyer notes, they range north to Alaska during El Niño years. The first record from Alaska (off Kodiak Island) dates back to 1937, when a school of barracuda was sighted, though only one was caught. The surface waters of the Eastern Pacific Ocean must have been warm that year, because a barracuda also was caught off Sooke, British Columbia. Another barracuda was found in Prince William Sound, Alaska, in 1958. In BC they are known also from Queen Charlotte Sound and the Prince Rupert area (see Hart 1973). Pietsch and Orr (2019) detail several barracuda records from the Salish Sea in their magnum opus, Fishes of the Salish Sea.
The first record along the BC coast was a specimen cataloged at the Royal BC Museum (RBCM 33), caught at Otter Point in Sooke, July 27, 1904. It is the only Pacific barracuda in the RBCM collection. According to Peitsch and Orr (2019) the earliest record of Pacific barracuda in the area comes from Gig Harbor, Puget Sound dating back to 1878.
I wouldn’t mind another specimen for the museum collection to accompany the 1904 specimen and our other warm-water strays: the louvar and finescale triggerfish found here in 2014, the North Pacific argentine from 2010 and the spotted porcupinefish from 2019.
I wonder what fish is next? Maybe we will get more hammerhead sharks? They were seen off Ucluelet in 1952 and 1953. Sure would be neat to have them here again.
References:
Carl, Clifford C. “The Hammerhead Shark in British Columbia.” Victoria Naturalist 11, no. 4 (1954): 37.
Cowan, Ian McTaggart. “Some Fish Records From the Coast of British Columbia.” Copeia 1938, no. 2: 97.
Hart, John Lawson. Pacific Fishes of Canada. Fisheries Research Board of Canada Bulletin 180. 740 p.
Quast, Jay C. 1964. “Occurrence of the Pacific Bonito in Coastal Alaska Waters.” Copeia 1964, no. 2: 448.
Pietsch, Theodore, and James. W. Orr. Fishes of the Salish Sea, Puget Sound and the Straits of Georgia and Juan de Fuca. Victoria: Heritage House, 2019. 1032 pages.
Van Cleve, Richard, and W.F. Thompson. “A Record of the Pomfret and Barracuda from Alaska.” Copeia 1938, no. 1: 45-46.
A Growing Tale post
Working at home has allowed me to pay plenty of attention to the individual lizards in our garden. I can watch where they go, locate preferred basking spots in the garden, watch what they eat, and try to figure out when eggs have been deposited and, later, when hatchlings will emerge.
Individuals are easy to identify based on size, sex and scarring. Most lizards have tails that have regrown, and the relative length of the original tail helps identify each animal.
Our gravid female had a perfect tail until recently, but on June 5, I noticed that she had been attacked. Her tail was now less than a quarter of its original length. The stump was still fresh and had not grown over with new skin.
We have several domestic cats vying for our garden (they also like using our garden beds as a litter box). The complexity of our garden attracts lots of birds, and it’s prime hunting territory for pudgy suburban felids to roam and kill at will. One cat (we know him as Meow, because that is what he said when we asked him his name), is a frequent visitor to our yard. He is likely the local lizard lacerator.
It appears that the growth is slow at first as the tail heals and the tissues organize themselves, but between June 19 and July 4, the tail grew an estimated 26 mm. The regrown tail will never be as long as the original, but it can be shed again if the lizard is attacked.
Nature is amazing. Field bindweed, an invader from Eurasia, grows at an alarming rate in our garden. Beans can go from a mere sprout to a massive flowering plant in a few weeks. And we can add lizard tails to our list of fast-growing crops.
When we harvest leafy veggies like lettuce, chard or New Zealand spinach, we take a few leaves for our meal and leave the rest of the plant to grow. The would-be predator attacking the wall lizards in our yard is also harvesting tails and letting the lizards regrow a new crop. I don’t think domestic cats have the mental capacity to intentionally farm lizard tails, but that is effectively what is happening.
I recently spent a glorious sunny day on Willows Beach in Victoria. Staghorn sculpins (Leptocottus armatus) and many small soles raced to deeper water as we walked along in ankle-deep water. The tide was out. People were everywhere, but no one was lifting rocks to see everything hiding in plain sight.
Further up the beach was a line of marine macroalgae (especially Ulva, sea lettuce) stranded by the receding tide. Under each rock you can expect a handful of isopods, as well as shore crabs and small Dungeness crabs that scuttle away, and even the tiniest puddles under a rock sheltered up to 8 fish—sculpins and gunnels. The sun heated the beach, but under the algae-draped rock, water stayed shaded and cool and kept everything alive until the tide returned.
Most pools had tidepool sculpins (Oligocottus maculosus), and most were the typical grey-brown mottled form. But there also were many of these green sculpins—the same species as the typical grey-brown tidepool sculpin. These green sculpins are perfectly camouflaged in patches of sea lettuce.
A bright-green tidepool sculpin sure stands out from its typical grey-brown relatives.
Without the usual coloration to rely on, you have to look more carefully to see whether this is a tidepool or fluffy sculpin (O. snyderi). Fluffy sculpins have cirri (little wispy flaps of skin) along the lateral line in clusters of three to six, and the clusters of cirri follow the lateral line along the flank of the body. This photo—even though it was taken with my old iPhone 6. which performs poorly when taking macro shots—shows that the cirri along the lateral line are only found near the head, and they are single. This clearly is not a fluffy sculpin.
Next time you are on the beach and the tide is out, take some time to explore tidepools and rocky ledges along the shore, and carefully lift some rocks. You probably will find a lime-green sculpin or two. You may also find lime-green penpoint gunnels (Apodichthys flavidus) or rosylip sculpin (Ascelichthys rhodorus). If you are really lucky, you will find sculpins with bright-pink colouration to match coraline algae, or a blue-and-red-banded longfin sculpin (Jordania zonope). Even in this city, there is plenty to see along shore.
Don’t Fence Me In post
On June 16, I received notification that my blog post on introduced lizards in Hawaii was live on the internet. The last paragraph in that article is about western fence lizards (Sceloporus occidentalis), where I ask people who see them in BC, to report the observation (with a picture if possible), or tag the occurrence in iNaturalist. Western fence lizards were on Matsuda et al.’s (2006) radar as a potential immigrant since the species exists so close to the Canadian border.
Between submitting that blog post and it going live, the plea for lizard sightings was answered. A report came my way of a western fence lizard loose in BC. I had always assumed the first record would surface in the Okanagan region, since several anecdotes from there suggest fence lizards are already present as far north as Oliver. Furthermore, field guides show western fence lizards just south of the Okanagan region (St. John 2002; Stebbins and McGinnis 2018) and Storm et al. (1995) presented a range map for western fence lizards having a straight line at the international border. Lizards don’t recognize political boundaries, so there is no way Storm et al.’s map is accurate. Fence lizards would do really well in the orchards, fence lines and piles of fruit crates of the southern Okanagan.
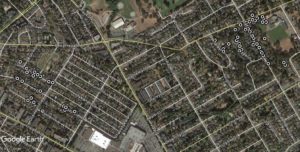
Instead of the Okanagan, the first record of a western fence lizard in British Columbia came from the Cloverdale area of Surrey, on June 6, 2020.
Our first confirmed western fence lizard was a juvenile, and it had lost its tail to some would-be predator. We have no idea how this lizard arrived in BC; most likely it is a stow-away from south of the international border. It could have been an escaped pet, or maybe there is a population now in the area that has gone unreported. The lizard is still loose, but we are hoping to catch it and add it to the museum collection.
The lizard is strangely coloured for a western fence lizard, but it does have the yellow-orange tint to the rear surfaces of the fore and hind limbs. The other possible look-alike in the region, though not in Canada, is the sagebrush lizard (Sceloporus graciosus), which has white on the undersides of its limbs. Sagebrush lizards are in Washington, but nowhere near as close to BC as western fence lizards (Storm et al. 1995; St. John 2002; Stebbins and McGinnis 2018). The nearest population of western fence lizards in Puget Sound is at Cherry Point, about 27 km south of where the Cloverdale specimen surfaced, thanks to a researcher in 1990 who released a handful of fence lizards to see if a population would get established.
In addition to this Cloverdale record in Surrey, a western fence lizard was reported on iNaturalist, April 2019, at MacNeill Secondary School, Richmond, British Columbia. However, this 2019 report cannot be verified since neither specimen nor photograph are available. Perhaps a second western fence lizard is loose in BC. Maybe it’s a sagebrush lizard. It would be great to get a picture of that Richmond reptile to verify the species.
And I am still not giving up on the Okanagan—if you live anywhere between Summerland and the international border, keep your eyes peeled for fence lizards!
Some background information:
Matsuda, B. M., D. M. Green, and P. T. Gregory. 2006. Amphibians and Reptiles of British Columbia. Royal BC Museum, Victoria, British Columbia, Canada.
Stebbins, R. C., and S. M. McGinnis. 2018. Peterson Field Guide to Western Reptiles and Amphibians. Houghton Mifflin Harcourt, Boston, Massachusetts.
St. John, A. 2002. Reptiles of the Northwest, British Columbia to California. Lone Pine Press, Renton, Washington.
Storm, R. M., W. P. Leonard, H. A. Brown, R. B. Bury, D. M. Darda, L. V. Diller, and C. R. Peterson. 1995. Reptiles of Washington and Oregon. Seattle Audubon Society, Seattle, Washington.
Rain this June has reduced our watering bill. Rain helps the plants we want, and also helps the weeds, so we spend a few hours each weekend weeding here and there. We are now well into the growing season, and we got a few new plants. From left to right below: the wasabi plant; behind it, an acacia (which will be potted—can’t wait to see it flower); a raspberry that creeps along the ground (YAY—ground cover in the food forest that produces food and limits the regrowth of grass); and a gooseberry. The ferns (below) we have had for a while in the shade along the side of the house and they need some TLC. Both sides of the house need some thought and effort once the main food-producing areas are established. The wasabi is growing well, and that acacia was so crazy looking that I couldn’t resist.
In the front food forest we have apples, cherries and blueberries well on the way.
Beans, onions and potatoes are growing fast.
The honey berries ripened early and we had them in pancakes. The front third of the garden is covered in wood chip and has three new sea buckthorns (far right below), a rose (middle) has set itself along the driveway, and I buried a few salmon berries to see if we can grow a few of those—that’ll bring in the hummingbirds. It is an area the deer can access. They seem to leave it all alone, and now that the grass is bashed back, only minor weeding is needed.
The back yard is looking lush. Along the eastern fence (on the right in this panorama) is our thornless blackberry—last year we had about 3 kg of berries—this year it is COVERED with flowers. What a bounty—and we save them frozen for winter crisps and smoothies (or for a small human who likes eating frozen berries like candy). The area around the sunken patio needs some effort—there are herbs in there—but the grass and other agressive plants have run wild.
The peppers, squash, cucumbers and pumpkin are growing nicely out back, although they are along the west side of the garden and our neighbour’s cedar hedge does drain the life from that soil. Plants grow poorly the closer they are to the cedars. Volunteer kale holds on and will seed again—yay kale.
Strawberries are ripening daily. You can’t beat the flavour of home grown strawberries warmed in the sun.
Ladybird beetles are everywhere. We have at least seven lizards in the front garden, and maybe three or four in the back. The female wall lizard which was obviously loaded with eggs now looks lighter, so we can expect hatchlings in a few weeks. The photo below is the male that may be the father of this year’s hatchlings, and as far as I can tell, our only garden lizard in the front yard with an intact tail.
The poppies are popping up all over the place. Tomatoes and cabbage are rocketing up, and the New Zealand spinach is ready for selective harvest. Chives have gone to seed, a leek has a seed head taller than Anna, and the lettuce is still growing well. There’s so much to eat in such a small space.
The usual avian suspects are flitting about—Bewick’s wrens; crows, which are now dive-bombing pedestrians; robins, which have nested and are much quieter; and Anna’s hummingbirds, chipping sparrows and house finches, which pop by regularly. We were buzzed by an osprey the other day—that was neat to see.
And I finally got around to protecting our grape with netting (above). The local mule deer seem to notice it once it grows about six leaves, and then they strip the plant. Not this year.
The garden is never finished, and it is always a learning experience. We are starting a chop-and-drop program to leave cut debris in place rather than taking it over to a separate compost pile. The chopped and dropped debris acts like leaves in a forest to reduce rain compaction of soil, and eventually it rots and adds organic matter to the surface – to be re-worked by nature. Probably the greatest thing to see is our daughter learning about the seasons in the garden, where food really comes from, what is ready to eat and what is not.
Cork It post
What does a cork have to do with my research? Cork was produced in Europe and North Africa and shipped to North America in bulk until the Atlantic was blockaded during WWII. Stow-away pests were inevitable.
In the 1940s, the western green lizard (Lacerta bilineata) was introduced repeatedly to Gloucester, New Jersey, as a stowaway in bales of cork bark (Kraus 2009; Burke and Deichsel 2008; Lever 2003; Conant 1945). In 1944, a single specimen of the ocellated lizard (Timon lepidus) also was caught on the Gloucester piers (Conant 1945). Common wall lizards (Podarcis muralis) also appeared over several years in New Jersey in the 1940s from shipments of cork bark (Kraus 2009; Conant 1945). The diversity of stowaway lizards underlines the ease and risk of accidental transport of lizards. Maybe the switch to synthetic stoppers has plugged this international leak in border security.
All corks aside, British Columbia’s wine region has been invaded twice by common wall lizards (that we know of). In 1983, a handful of these invasive lizards were intentionally released in two private gardens in Summerland. Fortunately for the ecology of the Okanagan valley, the Summerland introductions failed. The second invasion, in 2015, consisted of a lone wall lizard found at a vineyard in Osoyoos, transported as a stow-away in a shipment of grapes from Vancouver Island. Fortunately for fans of BC wines, the lizard was removed before grapes were crushed, and died in captivity.
You can see the record in iNaturalist, though the exact location in Osoyoos area has never been pined down.
While writing this short post I tripped across a website for the LacertA Winery in Romania. Their label showcases the eastern green lizard (Lacerta viridis), which grows to 45 cm total length. Imagine if eastern green lizards had been released here instead of the much smaller common wall lizard!
Photos courtesy of Walter Friedl, managing partner, LacertA Winery.
According to Speybroeck et al. (2016), Romania has us beaten for lizard diversity with the snake-eyed skink (Ablepharus kitaibelii), eastern slow worm (Anguis colchica), slow worm (A. fragilis), the steppe runner (Eremias arguta), sand lizard (Lacerta agilis), Balkan green lizard (L. trilineata), meadow lizard (Darevskia praticola), viviparous lizard (Zootoca vivipara), common wall lizard (Podarcis muralis) and Balkan wall lizard (P. tauricus). And now I can’t look at, or open, a bottle of wine without thinking of lizards.
References:
Burke RL, Deichsel G. 2008. Lacertid Lizards introduced into North America: History and Future. 347-353. In: Urban Herpetology. Mitchell JC, Jung Brown RE, Bartholomew B. (eds). Society for the Study of Amphibians and Reptiles. Salt Lake City, Utah.
Conant B. 1945. More reptiles in cork shipments. Copeia 1945(4):233.
Engelstoft, C., J. Robinson, D. Fraser and G. Hanke. 2020. Recent rapid expansion of European Wall Lizards (Podarcis muralis) in British Columbia, Canada. Northwestern Naturalist 101(1): 50-55.
Kraus F. 2009. Alien Reptiles and Amphibians: A Scientific Compendium and Analysis. Invading Nature: Springer Series in Invasion Ecology 4. Drake JA (ed). Springer, New York.
Lever C. 2003. Naturalized Reptiles and Amphibians of the World. Oxford University Press, Oxford.
Speybroeck, J., W. Beukema, B. Bok, J. Van Der Voort, and I. Velikov. 2016. Field Guide to the Amphibians and Reptiles of Britain and Europe. Helm Field Guide Series, Bloomsbury, London, United Kingdom.
I learn new things just by sitting in my garden and watching the behaviours of common wall lizards (Podarcis muralis). Flies may be fast, but lizards nab them. I put cutworm grubs on a pile of rocks and lizards happily sneak up and grab them too. Lizards eat ants.
People tell me wall lizards eat earthworms, even dried earthworms. I have received photos of wall lizards eating wasps. Wall lizards eat each other. They eat their own eggs.
But one thing that is abundant in my garden that lizards leave alone is the ladybug. We have several species of ladybug. The lizards have specific rock piles they seem to hold as resident territory, especially this pair of adults (above)—these two are always close to a small rock pile in my garden.
Right now, those same rocks are covered with larvae, pupae and adult ladybugs. Ladybugs smell bad because they emit:
2,5-dimethyl-3-methoxypyrazine (DMMP)
2-isopropyl-3-methoxypyrazine (IPMP)
2-sec-butyl-3-methoxypyrazine and
2-isobutyl-3-methoxypyrazine
The smell is described as a mixture of nutty, green bell pepper, potatoes and mould. I assume ladybugs taste like they smell. Even the yearling lizards in my garden are leaving the pupae and larvae of ladybugs alone, so they must learn early to leave ladybugs alone.
Even a small number of ladybugs can taint a batch of wine, and since lizards taste their way through the garden, constantly flicking their tongues, it is no wonder they leave ladybugs alone. Lizards likely don’t even have to bite a ladybug to know it is unpalatable.
Ladybugs and wall lizards do however eat the same prey—aphids. Gardeners may be happy to have both of these colourful predators in their gardens.
The chemicals in ladybug emissions are from:
Rovner, S.L. 2007. Why Ladybugs Smell Bad. Chemical & Engineering News. https://cen.acs.org/articles/85/web/2007/03/Ladybugs-Smell-Bad.html
Only one species post
I do prattle on about the invasive common wall lizard (Podarcis muralis) on Vancouver and Denman Islands, and the recent appearance of one Italian wall lizard (P. siculus) in Vancouver, but to put things into perspective, only two lizard species have appeared here, and only one is established. Others from the pet trade, or arriving as contaminants in tropical plant shipments (e.g., bearded Dragon, brown anole, green iguana), represent individuals that escape and never get established. Tropical species will not survive our cool wet winters.
Hawaii, by contrast, has more exotic lizard species than BC has reptiles. The common wall lizard pales by comparison to the Green Iguana.
The books I have for Hawaii are out of date, but they will serve to express the magnitude of the problem posed by accidental import, accidental release from the pet trade, and intentional release in areas where exotic species will survive.
Combined with records on iNaturalist, the lizards introduced to Hawaii include the green iguana, green anole, brown anole, knight anole, Jackson’s chamaeleon, mourning gecko, stump-toed gecko, Indo-Pacific slender gecko, common house gecko, Madagascar giant day gecko, orange-spotted day gecko, gold-dust day gecko, Tokay gecko, delicate garden skink, mottled snake-eyed skink, moth skink, copper-tailed skink and azure-tailed skink. Two species of horned lizard (Phrynosoma cornutum and P. coronatum) were released on Oahu, but failed to establish populations, and the azure-tailed skink probably is extirpated—an invasive ant may have caused its demise.
The green anole, a common pet lizard. This one was in captivity.
If we include species that died out, there are 20 species of lizard introduced to the Hawaiian Islands, with 7 species accidentally transported by Polynesians, and the rest more recently from the pet trade or as stow-aways in packing material. Only the yellow-bellied sea snake and sea turtles are native to the Hawaiian Islands, all other reptiles and amphibians were introduced by humans.
A female common wall lizard in Saanich, Vancouver Island.
I am sitting in my dining right now and looking out at the grey sky, knowing that it is 8 degrees Celsius outside. Tropical lizards have no chance on Vancouver Island, and so I am not alarmed at the annual wave of brown anoles that arrive as eggs in plant shipments from the USA. The lizard species that could survive here need to be able to withstand freezing temperatures. European lacertids, like the green lizard, western green lizard, Ibiza wall lizard, Dalmatian wall lizard and Italian wall lizard that at one time or another were established in the United States, and the viviparous lizard which appeared in Japan, are the only lizards that fit the profile. Perhaps the entire family should be prohibited from the pet trade in North America to limit the risk of introduction.
The western fence lizard also is a species that could survive in BC—it may be in the Okanagan region already, as far north as Oliver—so keep your eyes peeled for these guys. They have also been introduced to the Puget Sound area in Washington and would do fine on southern Vancouver Island. These prickly lizards are common around human habitation. They have bright blue patches on their bellies and do push-ups as a territorial display—if you see one, let me know or tag it in iNaturalist.
After picking up a range of starters at the local nursery, we are finally ready to plant the rest of the beds in the garden. A pumpkin starter arrived from the garden of Steve and Amy Lewis—they started a few too many mega-pumpkins—we were happy to get a seedling. Wasabi is the experimental plant for this year.
Soil was turned over, manure mixed down a bit, and weeds were pulled in preparation for planting. One of the most invasive plants in our front garden is Calendula—ours has a gorgeous orange flower, but this flowering plant had overgrown a third of the front vegetable bed.
Seeds arrived by mail, and we bought a range of peppers, some hotter than others—the scorpion pepper will be interesting.
All our tomatoes went in, as did cabbages, and we have a few hundred New Zealand spinach plants poking up. Ladybird beetles are pupating and there are loads of adults, so let’s hope they keep busy dealing with aphids.
New Zealand spinach (above) has an odd textured leaf and is not to everyone’s liking, but as it’s an edible weed, I am OK with it. Between New Zealand spinach, alexanders, kale, chives, oregano and leeks growing wild in the garden, we will always have some garden fresh food. The lettuce we bought as starters has overgrown its pot!
Lizards are active and emerge by 08:30 as the sun hits the front garden. Two larger lizards seem to have replaced the three yearlings that were on the back fence, and our gravid female still has not laid eggs. Two lizards seem to have been attacked—tails were shortened—but that seems to be it for lizard drama in the last few weeks.
Bewick’s wrens are nesting in our back yard, bushtits are stocking up on spider webs (and the cotton thread we use to tie up plants), crows are building nests, robins have hatched over a week ago, and Anna’s hummingbirds, chipping sparrows and house finches pop by regularly to liven up the garden.
Now to set up the watering schedule and let nature do its thing.
Light rain all day yesterday and the soil is nice and damp, but now warmed by today’s sun. Everything smells fresh and amazing. Lettuce starters are doing really well.
Last year’s kale is in full bloom, ensuring we have kale year round with zero effort. Other flowers do their part to attract bees.
Fruit trees flowering everywhere you turn. Blueberries soon will flower in the front yard.
Strawberries in bloom, rhubarb growing nicely, the fennel is up, and red sorrel pops up where it wants.
Rosemary attracts bees to the garden, while our raspberries and thorn-less blackberries send off many new leaves. Their flowers will soon appear. Oregano is everywhere in the raspberry bed.
Forgotten onions are growing, others have been planted. Potatoes and carrots are planted. Free range chives, kale and chard is as luxuriant as ever.
And our tomatoes—grown from seeds saved from last summer’s successes—are enjoying the sun on our south-facing deck.
And the lizards are increasing in abundance. This one likely hatched out early last summer. I haven’t seen our gravid female in a few days—maybe she is digging a nest.
Bewick’s wrens seem to have staked out territory and may nest here this summer. Chipping sparrows appear daily, as do American robins, dark-eyed juncos, chestnut-backed chickadees, house finches and our resident Anna’s hummingbirds.
Turkey vultures and bald eagles soar in thermals. And Cooper’s hawks keep the smaller birds on edge.
This city garden is alive.
Not much has changed in the garden itself since late March. Some weeding has been done, and I am specifically targeting a few plants that produce seeds early. Purple Deadnettles are everywhere and the battle with them essentially is a Kobayashi Maru scenario. Hairy Bittercress is another I target early since it catapults its seeds early in spring when the seed pods dry. Other than weeding and a bit of cleanup, we are now starting seeds indoors.
Thyme, tomatoes, basil (the seeds were not faulty), cilantro and peas came up nicely.
The arugula and cress are grown as “microgreens” to go directly into salads and sandwiches instead of into the garden. Growing microgreens is a year round thing for us—we have a small light stand with LEDs to start plants from seeds. We also have some flowering plants just for the flowers.
Leftover potatoes and newly bought onions are getting planted this Eostre weekend.
One of my favourite plants in the garden, however, is not something we eat. Mosses—mosses grow like mini forests and while some people power-wash the moss away, I like moss. I wish lawns were entirely made of mosses. Moss is soft on the feet, requires no mowing or maintenance or fertilizer, there is no need for aeration, and it makes plenty of habitat for tardigrades. I may never see a tardigrade in my garden, but I hope they are there and thriving. Since they can survive exposure to outer space, my garden likely is a tardigrade-happy place.
I remember the lifecycle of mosses from my time as a teaching assistant in Intro Biology (course # 71.125) at the University of Manitoba. Moss plants—more specifically, the haploid gameteophyte (gamete producing plant)—we are used to seeing come in a male and female form. The female and male gametophytes produce gametes—egg cells and sperm cells respectively. When water splashes on the male plant, some sperm splashes on top of the female plant, and fertilization of the egg cell occurs. Fertilization of an egg (syngamy) merges each haploid copy of the species genes into one cell (the zygote) which continues to grow with its diploid gene complement.
The sporophyte (or spore producing plant) is the next stage of the plant which grows as a stalk out of the top of the female gametophyte. The stalk elongates and develops a pod (known as a sporangium) at the tip where haploid spores are produced by meiosis. Those haploid spores are released and produce the next generation of haploid male and female gametophytes. This is my memory of the basic moss lifecycle—not bad considering I last taught intro biology labs in the early 1990s.
And I will end this Printemps-post with a gratuitous photo of one of our Common Wall Lizards, one of four (possibly five) which have taken up residence in the front garden. They were basking in the sun at 08:30 this morning.
When a trawl net goes down to the sea floor, you have no idea what will appear when the net is hauled to the surface. Some fish are caught as the net descends, others as the net ascends, but most are caught at the target depth. One thing is for certain, trawl nets sample very specific habitat – open smooth substrates where the net is unlikely to snag.
Since we don’t drag nets over rocky habitat, that habitat is poorly known. You’d think that in the dark depths, substrate wouldn’t matter – but apparently it does. And you’d think we’d already know whether a fish the size of a coffee table inhabited our waters. Not so. Since 2005, we have discovered three new skates in BC, and you guessed it, one was the size of a coffee table. The first Angel Shark for British Columbia was discovered in 2016, and captured on camera, not a net.
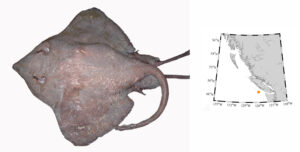
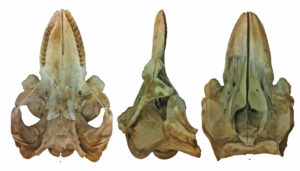
The Pacific White Skate (Bathyraja spinosissima) was caught in a trawl net by accident. From footage from submersibles, we know this species cruises about 1 m above rocky habitat. The single specimen caught in 2005 had strayed over net friendly habitat. When this skate was caught, we were unable to assign it to species. Its DNA suggested it was a species from the Atlantic. After a careful measurements, we determined it was a Pacific White Skate, and its discovery was published by Orr et al. (2019).
Bathyraja spinosissima (Pacific White Skate) – this specimen was the first to be included in DNA barcoding which gave us strange results when the tissues were originally sequenced. This large male is the first adult male in museum collections, a first from BC, and extended the known range north from Oregon.
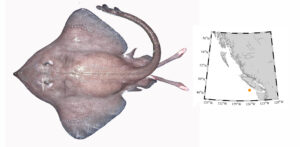
Orr et al. (2019) also listed a second new skate species for BC, the Five-spined Skate (Bathyraja microtrachys). Five males were collected west of Tofino, off Vancouver Island in 2005. These are the only adult males known for this species.
Bathyraja microtrachys (Five-spined Skate) – originally thought to range from Washington south to San Diego, we now known they range into Canadian waters.
In 2009, a third skate species was discovered, the Commander Skate (Bathyraja lindberi), and was published by King et al. (2019). This is a northern skate, and the discovery in British Columbia extends the species’ range south about 600 km.
Bathyraja lindberi (Commander Skate) – appeared in 2009, and now is known from the Sea of Okhotsk, the Bering Sea, Aleutian Islands, south to Queen Charlotte Sound.
Finally, in 2016, a Pacific Angel Shark (Squatina californica) was spotted and photographed by Mark Cantwell while diving off Clover Point, Victoria. The photograph was proof enough of the species’ presence (King and Surry 2016). Since this species was known from a single specimen in Alaska, one from Puget Sound in Washington, and exists south to the Gulf of California, its discovery in British Columbia was only a matter of time. More details on the Angel Shark can be found in Pietsch and Orr’s (2019) magnum opus on Fishes of the Salish Sea.
Squatina californica (Pacific Angel Shark) – was is known from one specimen in Puget Sound, Washington and an old record from Alaska, and finally has been confirmed for British Columbia. Photograph by Mark Cantwell, April 30, 2016 off Clover Point, Victoria, Vancouver Island.
For more details on all cartilaginous fishes in British Columbia, buy the latest RBCM handbook by King and McFarlane due out May 2020.
Non-game fishes receive little attention and of these, the deep sea anglerfishes (Families Oneirodidae, Melanocetidae and Ceratiidae), are among the poorest known. Deep sea anglers live at extreme depths and few people study them.
Alive they are quite elegant with black-brown skin, flowing fins and globular bodies. But when hauled to the ocean surface and preserved in museum collections they tend to resemble shrivelled hockey pucks – with teeth. Most of what we know about these fishes is based on the females. Males are minute but have enlarged olfactory chambers. Males have one goal – to find and latch onto a female. Once attached, males draw nutrition from the female’s bloodstream. Sometimes more than one male attaches to a female, and they are in place ready to mate whenever a female is ready to release eggs. Some have said this relationship is parasitic, and while males do derive nourishment from the female, they fertilize a female’s eggs. In contrast, a parasitic relationships is one-sided. Perhaps the only time the relationship is parasitic is when a male attaches to a female of a different species – he draws nourishment, but provides no genetic contribution.
Because these fishes rarely reach the surface in good shape (trawl nets are not forgiving to soft-skinned fishes), many deep sea anglers are difficult to identify. Identification often is based on the structure and shape of tip of the lure – a structure called the esca – which sits at the tip of the first ray of the dorsal fin. If that delicate structure is lost, identification can be difficult without DNA barcoding. With DNA barcoding, we can identify species based on unique genetic sequences taken from tissue samples. One of the fishes photographed in this article, Oneirodes thompsoni, had been sampled for DNA barcoding to get a representative sequence for this species. The white tag with a code number is attached to the fish so that the fish and its genetic sequences can be matched later.
Until the paper by Weil et al. 2015, only three species, had officially been recorded from BC waters. Specimens of the large Krøyer’s deep sea angler fish (Ceratias holboelli) had been preserved, but no one had detailed where the species had been found. Several other dreamerfish had been misidentified in museum collections. The review of species known to exist here was prompted by the identification of a specimen of Melanocetus johnsonii in the RBCM collection, and the discovery of Oneirodes acanthias and Cryptopsaras couesii in 2006. The other species deemed new to BC, or representing significant range extensions, were discovered during the preparation of the paper by Weil et al. and his review of the RBCM collection.
Below is the list of anglerfish species known to exist in British Columbia (as of March 2020):
Order: Lophiiformes
Oneirodidae:
Oneirodes thompsoni
Oneirodes bulbosus
Oneirodes eschrichtii
Oneirodes acanthias
Chaenophryne melanorhabdus
Chaenophryne longiceps
Melanocetidae:
Melanocetus johnsonii
Ceratiidae:
Ceratias holboelli
Cryptopsaras couesii
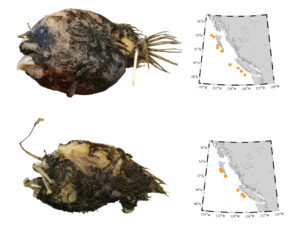
Oneirodes thompsoni, RBCM 010-00196-009, 10.9 cm Standard Length (above); and O. bulbosus, RBCM 004-00005-001, 8.8 cm Standard Length (below), are the most commonly caught dreamers along the entire BC coast. Photo by G. Hanke.
Oneirodes eschrichtii, RBCM 998-00323-002, 8.3 cm Standard Length, represents the first of its species in BC and a northward range extension of 1700 km in the eastern Pacific Ocean. Photo by G. Hanke.
Oneirodes acanthias, RBCM 010-00197-004, 10.5 cm Standard Length, represents the first specimen from BC, and a range extension of 600 km north of the species previously known range off Oregon. Photo by G. Hanke.
Chaenophryne melanorhabdus, RBCM 999-00107-002, 8.9 cm Standard Length, is another commonly caught dreamer, but we have no records north of Vancouver Island. Photo by G. Hanke.
Chaenophryne longiceps, RBCM 998-00344-001, 9.8 cm Standard Length, is one of three specimens now known from BC, roughly 600 km north of the species previously known range off Oregon. Photo by G. Hanke.
Melanocetus johnsonii, RBCM 01400308-001, 9.8 cm Standard Length, is the only specimen known from BC waters and is the first record north of Oregon on this side of the Pacific. Photo by G. Hanke.
Ceratias holboelli, RBCM 999-00081-001, 42.0 cm Standard Length, is our largest deep sea anglerfish, distinctive with its thick elongate fin rays and cover of large thorny denticles. Photo by G. Hanke.
Cryptopsarus couesii, RBCM 014-00309-001, 14.2 cm Standard Length, was caught as bycatch from the commercial fishery and since it was so strange, was kept for identification. It represents the northern most record for the species in the eastern North Pacific Ocean and a first for BC. Photo by G. Hanke.
Spring has sprung, and it is great time to get going on a garden. There are a ton of things you can grow at home to supplement your diet and reduce your dependence on grocery stores. My wife and I have jokingly named our property UF1510 (Urban Farm 1510). Our street address obviously is 1510, and our garden is the only one on this street fully devoted to food production.
Planting seeds for this summer.
When we bought the house over 10 years ago, the front yard had a small patch of St. John’s Wort, a spindly ornamental shrub, a sizable Laurel tree, an isolated Camelia bush and a dodgy coniferous tree. But the majority of the front yard was a flat expanse of lawn. Everything but the Laurel was removed quickly, and over the years, the lawn has been eliminated. We have dedicated a third of the front yard to bare ground for potatoes, beans, carrots and a few other crops. The remaining two-thirds is a “food forest.” We planted blueberries, sour cherries, apple trees, hazelnut bushes, asparagus and a range of flowering plants to attract pollinators. Don’t forget the bees—if you want tomatoes, berries, cucumbers, squash—you need pollinators.
Alexanders (left) and Salad Burnet (right) are available in winter as a supplement to salads.
The back yard also was a monoculture of grass when we moved in. It now is changed to a series of raised beds and we grow many varieties of tomatoes, kale, chard, onions, leeks, chives, cucumbers, squash, blackberries (a thornless variety) and raspberries—to name just a few of our home-grown food items. Many of our plants self-seed and effectively are edible weeds (cilantro, salad burnet, alexanders, arugula, kale, leeks and chives are weeds in our garden). I also eat dandelion leaves.
The back yard with its raised beds just waking up from the winter. Oregano, chives, raspberry canes and leeks are going strong—we will have to wait for the raspberries of course.
This change in gardening practice took us from a flat lawn which needed to be mowed regularly (wasted energy, in my opinion) to a garden we can walk around and pick fresh produce for our daily meals. Even in winter (because we live on southern Vancouver Island), there is food available in the garden. We also leave the deadfall from the previous summer in place all winter. Why leave the deadfall in place?
1) I am lazy.
2) it is habitat, shelter and a source of food for overwintering birds.
Our garden is chemical-free so there also are insects—but thanks to our local birds (and the invading wall lizards), we get free pest control, entertainment and a dash of colour as they flit about.
Rhubarb, kale, chard, rosemary, and winter cress seem to look after themselves.
This year I am going to post a series of blog articles to show the progress in the garden—this early in the spring it looks messy—weeds have grown, and last summer’s deadfall has yet to be cleared away. But the garden is full of life—birds are everywhere. Lizards skitter around on the warmer days (they invaded in 2019 from a small population to the north of us). We welcome spiders, but we do draw the line at rabbits and deer – they are not permitted.
Strawberries, a potted peach tree and some early flowers.
This month, we will clear out the dead stems and weeds. The dead stems go into the compost—they form the dry brown material that alternates with wet kitchen scraps. We don’t eat meat, so everything from the kitchen goes into the compost. And in the kitchen we have light stands to start seeds. Once the risk of frost is gone, the seedlings will be planted.
This summer’s tomatoes will be started indoors.
I hope this series inspires people to change their gardening practices and replace lawns with food plants. Even a deck or apartment balcony can provide a small mountain of food. You can even grow your own sprouts in your kitchen to add some home-grown greenery to a sandwich. Our garden is a green security blanket, a source of exercise, entertainment and also nourishes us with food and a sense of pride.
Tonight, we are having homemade soup which will have diced leaves of home grown kale, chard and alexanders. To kick the series off, here is the recipe for the soup.
2 chopped carrots
1 chopped medium sized yellow onion
2 chopped celery stalks
3 cups of vegetable broth plus 3 ½ cups of water
1 pack of simulated shredded chicken
about 1 teaspoon of poultry seasoning (I never measure exactly, this is cooking not chemistry)
about a 1 cup of pasta or 1/3 of a package of spaghetti or linguini
1 cup or so of frozen peas
We sauté the carrots, celery and onions in a large pot on medium heat for 5 or so minutes. Then we add the broth, the 3 ½ cups of water, fake chicken, and seasoning and bring to a boil, then simmer for 10 minutes – maybe more – until the veggies are cooked. Then toss in the pasta and peas and simmer until the pasta is to your liking, and then we add a handful of chopped kale, alexanders, and chard at the end. Season with salt, pepper to taste. I also add a few drops of a decent hot sauce to my bowl.
Thank you garden.
Not a Killer Whale? post
When is a whale not a whale? When taxonomy conflicts with everyday language.
According to the author of a recent article on the internet, “Although they’re called killer whales, orcas are not actually whales; they belong to the oceanic dolphin family, of which they are the largest members.”
That statement is both correct in that orcas are in the family Delphinidae, and also incorrect at a higher level—and such statements are ubiquitous on the internet. Using this logic, a goldfish is not a fish because it is a member of the carp family. You are not a placental mammal because you are in the family Hominidae. And roses are not plants because they are in the Rosaceae.
Carp are fish. Minnows are fish. Goldfish are fish. Placing species in family groups in manmade taxonomic schemes does not eliminate the larger more inclusive groups defining types of organisms. A goldfish is a cyprinid, cyprinids are a type of fish, and therefore, goldfish are fish.
Orcas presently are in the family Delphinidae. The family Delphinidae is one of many cetacean (whale) families. So by the transitive relations: orcas are whales.
Whales ultimately evolved from a group of terrestrial mammals, derived from synapsid reptiles, which evolved from amphibians, which evolved from lobe-finned fishes. So mammals, including whales, also are a subset of fishes, if we take this to a crazy extreme. I take great delight in being a fish and a Pisces—not that the present alignment of stars is an indicator of anything.
How Handy is That? post
During the yuletide season of 2014, Rhapsody was found dead. Her unborn daughter had died days earlier. Rhapsody was unable to expel her calf and died from the decay and infection. Her calf was examined at the pathology labs in Abbotsford, then returned to Vancouver Island so that we could prepare her skeleton. She sat in a freezer while we debated the best way to prepare so delicate a skeleton.
In the spring of 2019, Mike deRoos of Cetacea Inc. decided we should immerse the calf’s remains in a tank of warm water which was oxygenated with an airstone, and let bacteria do the work for us. Once the tank was ready, I drove to Saltspring Island with a fragrant tub in the back of my car.
Months later, the bones were ready for pickup. My car is nicknamed Tydirium – after the shuttle from the Return of the Jedi – it still smells new inside. Fortunately, the car still smells ok after shuttling a foetal orca back and forth from Saltspring Island.
Now I have the task of sorting out all the disarticulated bones and putting them back in order. Rhapsody and her calf will be displayed alongside each other in our Orcas exhibit opening May 2020.
Like humans, bones of a newborn are still growing – and skull bones are not solidly sutured together. The skull bones from Rhapsody’s calf came apart. Vertebrae are not fully formed either – the neural arches, lateral processes, centra and cookies are all separate. What a puzzle! Ribs, sternal ribs – all of these need to be sorted out. And then there are the finger bones in the flippers – I was not looking forward to sorting these out.
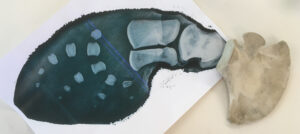
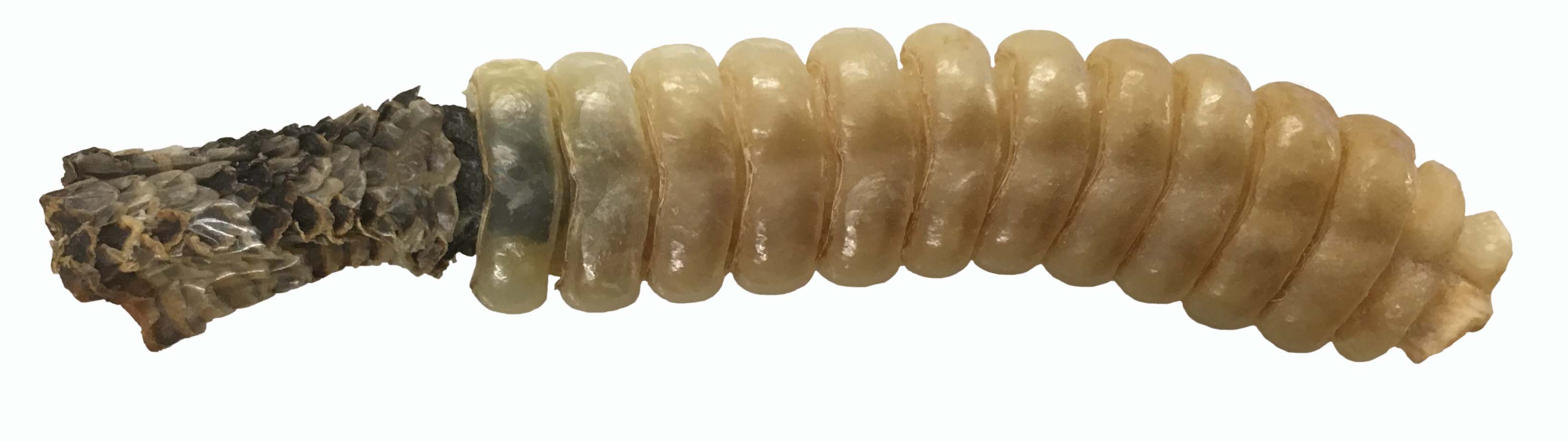
But here is where Mike deRoos saved the day – he arranged to have the calf’s flipper x-rayed, and the image printed at full size is better than an IKEA or LEGO instruction booklet to guide reconstruction of both left and right flippers.
The three carpals and a few of the distal phalanges are not formed – and the metacarpals and phalanges are widely separated by cartilage. But the size and shape of each bone is so distinctive, that I was able to place each bone where it should go in minutes. Two phalanges from the fingertips of the right flipper were lost during preparation – but overall the flippers look great.
Ribs are next.
Nattering post
We rely heavily on technology – integration – connectivity. This was the downfall of the newer generation of Battlestars when the Cylons attacked. Today the government network was down. But it was still possible to connect my phone and a memory stick and write something. And today I am writing about an integrated, connected global network for biological information which at the moment, is inaccessible to me.
For a few years now I have been collecting lizard range records in a spreadsheet to support a research paper on rapid range expansion in the Common Wall Lizard. That paper now is in-press and will be printed this autumn. Now that we don’t need to be so secretive with our data, we have changed strategies and are encouraging people to report lizard sightings directly to iNaturalist.
iNaturalist is a global initiative where anyone can sign up, and then report the species they find. A global network of experts help make sure identifications are accurate, and the combined efforts of thousands of observers creates incredible maps for each species. Obviously the species reports are biased – the maps detail where humans go, and microscopic life and invertebrates are underrepresented because they are really hard to ID from a simple photo. But fungi, larger insects, plants, vertebrates – all are mapped in amazing detail.
These location records now are available to people around the world to track species ranges and how species ranges may change in the future – climate change, habitat loss – you name it – the data is out there to study.
To enter a record, first you need to get a photograph –you can make a report without a photo – but it is impossible for others to verify what species you found. Photos need to be as good as you can get – a fuzzy blur is not much help. You also can report a sound recording (bird calls for example, or maybe sound files from bat detectors).
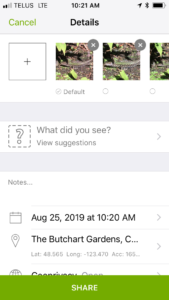
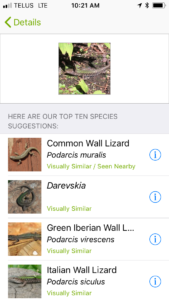
This weekend my wife and I were walking around Butchart Gardens on Vancouver Island, and the organism I found – no surprise – is a Common Wall Lizard. In the case I detail below, I have iNaturalist on my phone, and this series of images depict the process of discovery to creation of iNaturalist record. The process takes seconds.
You take the photos – and this can be tricky – some animals are skittish. I get a basic shot and then slowly close in to get better photos until the subject runs away (botanists and mycologists don’t have this problem). Even a fairly bad photo like the one below is good enough in some obvious cases- but better photos makes identification easier.
Link the photo(s) to the record.
Then you specify what species you think you have found – or you can leave it to genus, family, or blank.
Then you specify the location and date – you can do this manually, or the software draws the location and date from metadata in your photographs.
Then click on the submit record button – and the record now is live for others to view.
If your identification is correct, the global community will also ID the organism as you did, and the record goes to a status known as ‘research grade’.
If you are wrong, people suggest other identifications. You can choose to agree or let the global community of experts come to consensus. Several of my spider and insect identifications have been corrected by experts – and that is the power of this network. We help each other refine the accuracy of these data.
When I take off with my phone to make iNaturalist records, I say I am Nattering. May has well have a fun name for this activity. It is kind of like trophy hunting – but you create a digital record for the world to see rather than a stuffed animal, or pressed plant, or pinned insect. It’s also highly educational – I have no idea of the identity of many of the insects I photograph, but the software and global community really helps teach what is living right in my own neighbourhood.
It costs nothing to load and use the software and for my wife and I, we use it as a fun activity we can share while enjoying hikes on this Pacific island, or while our car charges during road trips.
Why not join the team and photograph what ever you want – starting with Common Wall Lizards and plot their locations in this software. It is fun and you’d be contributing to science.
FTL post
In science fiction, FTL means Faster Than Light. But today I am changing the acronym to suit my needs – Fast Tiny Lizard. On June 19th, 2019, Cathy Judd caught a lizard in an industrial area of Vancouver, and reported to Ashlea Veldhoen (Habitat Acquisition Trust) for identification. I got an email from Ashlea stating that a small green coloured lizard had been caught in Vancouver. I could hear Captain Piccard bellowing Red Alert in my nerdy mind, and I replied with questions and an urgent plea: “PLEASE DO NOT LET IT LOOSE.”
Then I asked for pictures.
I was expecting to see a photo of a Common Wall Lizard in any reply – but nope – to my delight and horror, the lizard in the photograph had two well defined stripes and a pale belly with no dark blotches – it was obviously not a Common Wall Lizard.
This little lacertid was an Italian Wall Lizard (Podarcis siculus) – a species known from scattered locations in the United States including a population on Orcas Island, Washington. They apparently have existed on Orcas island for about 12 years now, but had only been reported in 2018.
Dorsal view of the female Podarcis siculus (RBCM 2187, TL = 143 mm, SVL = 59 mm) from Vancouver, British Columbia.
Ventral view of the female Podarcis siculus (RBCM 2187) from Vancouver, British Columbia.
This lizard, a female, based on its light build and weakly developed femoral pores (males have well developed pores along their thighs) is a first for BC, and possibly also for Canada.
This lizard could have carried a clutch of 5 or more eggs. But no others have been seen in the area, so I assume this Italian immigrant was a lone stow-away. How it got here is a complete mystery. Was it a dumped pet? Did it stow away in camping gear used on Orcas Island? Or was it hiding in shipping materials from Italy or one of the colonized locations in the USA? We may never know. Perhaps some enterprising student will want to examine its DNA and see if there is a match for a source population.
Since Italian Wall Lizards are established on Orcas Island in the same climatic zone as southern Vancouver Island and Vancouver, is successful in cooler climates elsewhere in North America, and known to prey on animals as large as shrews, it represents a high risk invader. At least this one is safely preserved in the RBCM collection, and won’t be the matriarch of a second invading Podarcis species.
Missed the Bus post
Some people get angry when they miss a bus. This morning as I crossed Bowker Creek, and saw my bus heading south. It was a cool morning. It had rained overnight. The air smelled of spring. Birds were everywhere. Missing the bus was a good thing.
Even along suburbian streets, you can do a bit of bird watching when you miss a bus. Regulars along my street include: Northern Flicker, Common Raven, Northwestern Crow, American Robin, Bewick’s Wren, Merlin, Spotted Towhee, Chestnut-backed Chickadee, and Anna’s Hummingbird.
A nesting Anna’s Hummingbird – our neighbour’s Holly bush makes pretty safe nesting habitat.
Overhead you can see a range of gulls, Great Blue Herons, Red-tailed Hawks, Turkey Vultures, and Bald Eagles. Mallards and Canada Geese fly over on their way to and from the local golf course water traps, and a pair of Mallards have taken up residence on the neighbour’s lawn.
And it is no surprise we have exotics like the California Quail, House Sparrow, and European Starling.
Violet-green Swallow – always fun to see them overhead.
In summer we get Violet-green Swallows, Barn Swallows, Downy and Hairy Woodpeckers, White-crowned Sparrow, Purple Finches, and the occasional Western Tanager, Wilson’s Snipe and Common Nighthawk. A few years ago over 30 nighthawks cruised the neighbourhood – that was neat. Steller’s Jay, Varied Thrush, Golden-crowned Sparrow, and Dark-eyed Junco are more common in winter.
Missing from this list is owls – there have to be suburban owls in my neighbourhood – especially Barred Owls – I just haven’t seen any.
Great Horned Owl – not in my garden though (the jesses give it away).
This morning I walked smack into the middle of an irruption of Bushtits, Chestnut-backed Chickadees and Golden-crowned Kinglets. It’s hard to be mad at missing a bus when you are surrounded by those tiny energetic birds, each searching for a morning meal. Then an Anna’s Hummingbird chased one crow away, and while another crow flew by with nesting material in its beak. Seconds later, the bus arrived to ruin the fun – everyone on board staring at their smartphones.
I wonder if any of the passengers were checking a birding app? Were they on iBird? Ebird? Or adding a sighting to iNaturalist? I wonder how many know that a smartphone can be used for more than playing Angry Birds.
The diversity of wildlife apps is increasing at a time when global biodiversity is in peril. Some apps like the Sibley’s bird guide on my phone are more for simple identification and to log your life list. Others feed into bigger networks that help science and feed into wildlife management decisions.
Not sure which app is right for you? Read this review of 5 top birding apps.
If you aren’t interested in slogging through a birding app in a phone or tablet, maybe this would be for you – a project proposed for Google Glass – Glass Birds – as for this YouTube video though – anyone can ID a Western Tanager.
I wonder if Glass Birds is refined to ID birds like this Rock Wren? I need the Google Glasses for gulls, some sparrows, fall warblers, flycatchers, and shorebirds, and best of all, I would be a taxonomic cyborg!
We hear on the news about our armed forces personnel helping people overseas during peacekeeping missions. This year we saw our forces helping flood victims and airlifting communities threatened by forest fires here in Canada. But military presence also can help wildlife – yes, wildlife can flourish even on live fire ranges where tanks range far and wide.
A frame from Sebastian Koerner‘s film footage of a wolf pup and a Marten (Marder) in the Munster-Military training area Lüneburger Heide, Germany.
A recent article in the NewScientist detailed how military ranges are benefitting wolves in Europe. Large carnivores tend to fare poorly when in close contact with our species – we shoot them, our highways and industry fragment and destroy their habitat, and we can thank our automotive industry and its infrastructure for the term Road Kill.
On military ranges, the gun fire is routine and predictable and animals get accustomed to the episodic timing of activity. The range itself and the area nearby generally is devoid of people – except for the short periods during training exercises – and the military presence is a huge deterrent for poachers. I took a Wildlife Management course years ago, and the professor said, “Wildlife management is not about controlling wildlife, it is about controlling people.”
Regulation of human activity in military training areas is so effective, wolves seem to prefer testing ranges in Germany – they are more abundant in military training areas than in nearby wildlife management areas. In addition, roads in testing ranges make convenient thoroughfares for large carnivores and have far less traffic compared to highways near parks.
While reading this NewScientist article I had a flashback to my M.Sc. supervisor, Ken Stewart and his recollection of a misty morning searching for snakes around the Canadian Forces Base in Shilo, Manitoba. The testing range near CFB Shilo is adjacent to the Sprucewoods region and what was once Lake Agassiz beachfront, is now undulating terrain and perfect for training against hidden targets. For tank crews, this sandy, gravelly substrate reduces the risk of ricochets during live-fire exercises and makes it easier to retrieve ordnance. This same sandy habitat also is home to some rarely encountered animals and plants in that province.
The Plains Hognose Snake (Heterodon nasicus); photo by Dean Hester.
Ken described his morning, bushwhacking through a forested area adjacent to the base and how the conifers reminded him of the Ardennes Forest and the Battle of the Bulge. He imagined he morning’s sub-silence shattered by the clanking tracks and diesel engine of a Tiger Tank, and said, “All I needed was Lili Marlène playing on an old turntable to complete the scene.” But when Ken was there, it would have been a Leopard, not a Tiger ensuring that all but the most intrepid herpetologists were kept well-clear.
Leopard 1 Main Battle Tank; photo by Sergeant Dennis Power, Army News-Shilo. ©2008 DND/MDN Canada
Essentially, this pocket of land within the ranges of the Northern Prairie Skink (Plestiodon septentrionalis) and Plains Hognose Snake (Heterodon nasicus) is protected by our armed forces and right next door to Spruce Woods Provincial Park. Ken’s story dates back years before he and I met – and so the concept of military ranges as critical shelter for wildlife is certainly not new. CFB Shilo, recently saluted for its work on wildlife conservation, has a Base Biologist who oversees natural resource management, gives tours of the sand dunes, and facilitates research in this restricted area. Other bases like CFB Suffield provide grassland habitat for a myriad of species including Burrowing Owls, Sprague’s Pipit, Pronghorn and Ord’s Kangaroo Rat. CFB Gagetown is working to protect one of nature’s tanks – the endangered Wood Turtle.
Sandy blow-out areas and mixed woods habitat protected by CFB Shilo, image courtesy of the Manitoba Association of Plant Biologists.
Since the concept of military ranges as shelter for wildlife is not new, the NewScientist story on wolves serves to reinforce the fact that human activity now is the primary factor influencing nature – as also stated in Bill McKibben’s book, The End of Nature. If we restrict human activity, nature thrives. Since wolves in BC capitalize on human activity and use pipelines, roads and seismic lines cut during oil and gas exploration, and well-packed snowmobile trails as easy avenues to track Caribou, you can bet BC’s wolves also could adapt to military ranges as their relatives do in Germany. The list of species protected on Department of National Defense (DND) land is extensive and it is good to see that in areas where soldiers practice and you’d expect disruption and devastation – the opposite is true – human activity is restricted, and life adapts to, and is protected by our armed forces.
Branching Out post
I recently spent an enjoyable day hunting Common Wall Lizards to help Camosun College students with a research project. Lizards were easily caught with nooses, by hand, and using elastic bands. These lizards are to be used in a diet study to see whether there is any pattern between historic and current arthropod diversity in pitfall trap samples, and to determine what lizards are selecting from the available invertebrates.
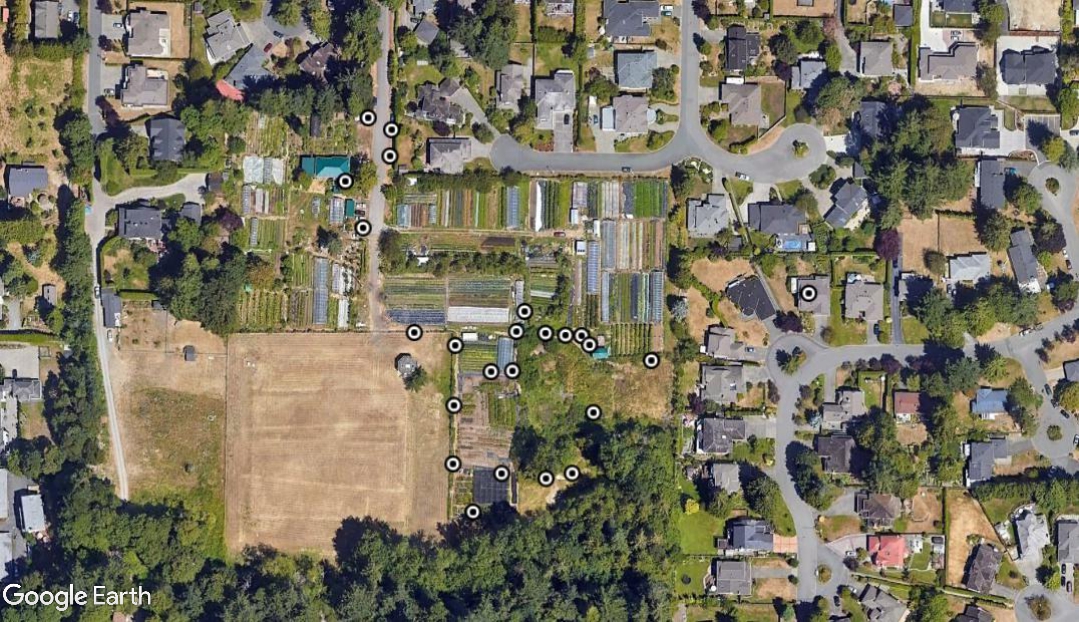
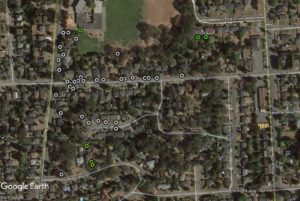
We sampled at Haliburton Farm in Saanich, here on Vancouver Island. Lizards were everywhere – and that is no exaggeration. Every few steps would cause one or more lizards to skitter way into the forest of potted plants and garden veggies growing at the farm.
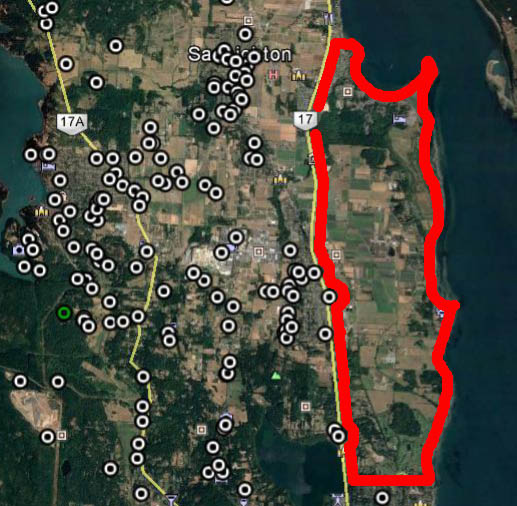
My Google Earth Map totally under estimates the number of lizards because I couldn’t map the location of each one. There were hundreds of lizards in each section of the farm. Adults were predominant in the heavily modified areas, and yearlings seemed to be occupying peripheral areas that were almost semi natural – young ones likely avoided the main farm to avoid cannibalism.
The tree and a closer view of the knot-hole where the Wall Lizard sought refuge.
One lizard stuck out in my memory – because it was trying to shed the “wall lizard” stereotype by living in a tree. I spotted an adult male well up a tree – and as I approached, it bolted into a knot-hole. The knot-hole led to a significant cavity inside – I used a long dry grass stem to get an idea how large the cavity was. It was at least 20 cm long, plenty of room for an adult Wall Lizard. Years ago Richard Hebda noted that Common Wall Lizards had started to occupy grassy habitat as well as the typical more solid habitat. This lizard seemed more interested in becoming a Tree Lizard – sorry Podarcis, you can change habitat, but not your taxonomy. Luckily Urosaurus ornatus does not live here and won’t have to deal with this arboreally inclined invader.
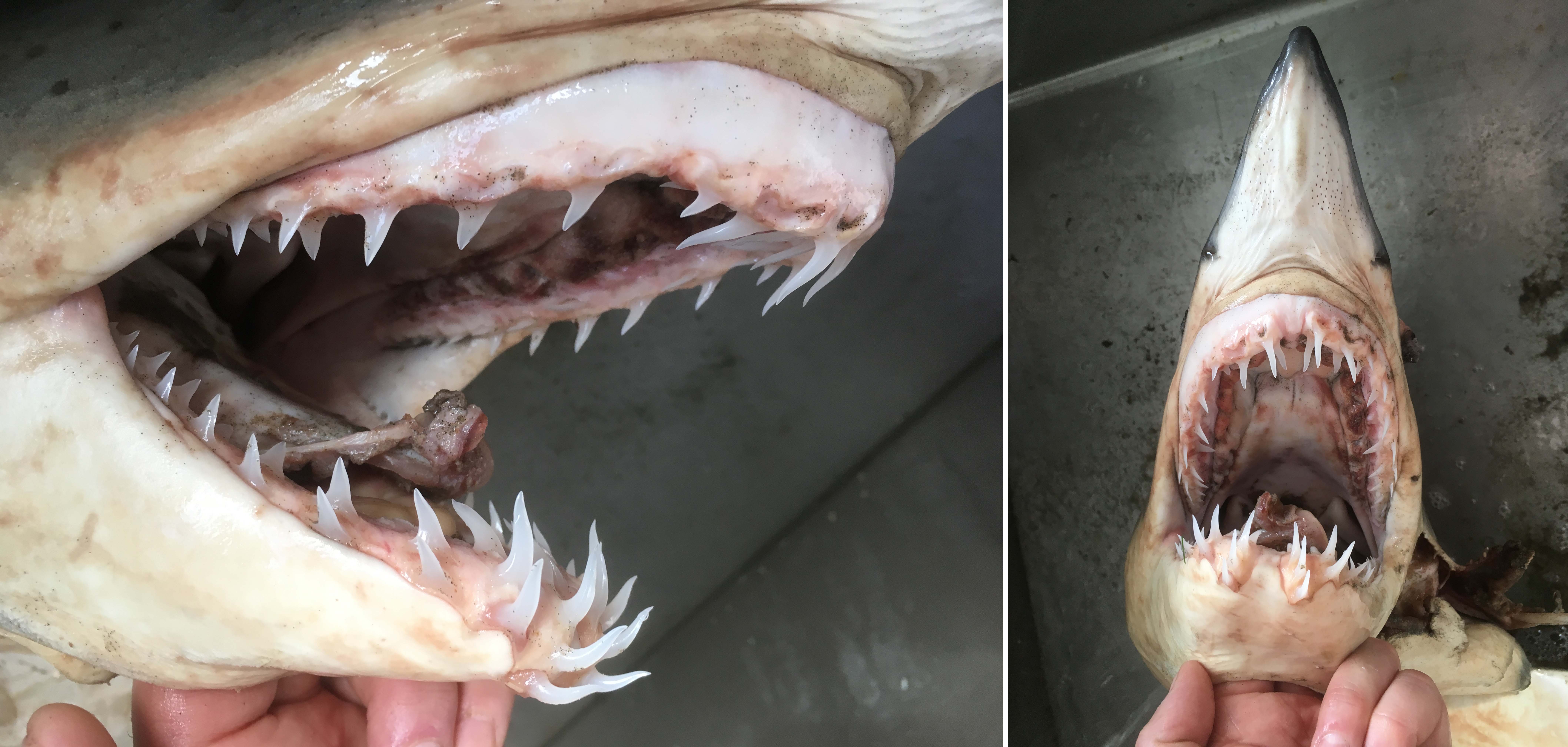
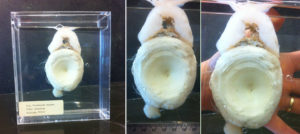
Last summer my wife and I bought a new car – it is less than a year old and has already transported quite an assemblage of BC species (Southern Resident Killer Whale foetus, Mule Deer, River Otter, Red Fox, Northern Alligator Lizard, Common Wall Lizard, Commander Skate, and 49 species of birds – including the museum’s first Brown Booby). The most recent passenger was a 1.2 meter Shortfin Mako Shark (Isurus oxyrhinchus) which easily fit into the back of a 2018 Nissan Leaf. Chalk up another reason why electric cars are awesome.
The Mako Shark (wrapped in plastic) arrives at the RBCM loading bay.
As far as I know, this is the second Shortfin Mako Shark specimen from BC waters. The first specimen, from 185 nautical miles west of Cape St. James (Haida Gwaii), was made into a taxidermy mount and only a few of its teeth were deposited in the Royal BC Museum collection (993-00039-001). You have to wonder how often they range this far north?
The mako shark thawed and ready for a long soak in formaldehyde.
This new mako, found September 27, 2016 on shore in Florencia Bay, Pacific Rim National Park Reserve is almost perfect. It had been studied by Jackie King (Fisheries and Oceans Canada), tissue samples were taken, and then was shuttled to the Institute of Ocean Sciences (IOS) in Sidney. I picked up the fish at IOS and kept it frozen until I had the time to prepare the shark for the Royal BC Museum collection. Mako sharks are most streamlined representatives of the Family Lamnidae, the same family containing the Great White Shark. It was a thrill to see this amazing fish up close. Its only damage came from scavengers – the left eye is missing, and something – a wolf(?) – had ripped at the gills on the left side.
Tooth rows are easy to see in the jaws of this Shortfin Mako.
The teeth are amazing – and let’s face it – this is what most people want to see on a shark. But have a look at the tail! Without an efficient tail – the teeth would have nothing to bite. Mako sharks are amazingly fast and almost appear nervous when they are swimming – they are certainly the Formula-e cars or jet fighters of the shark world.
The base of the tail on our new mako shark.
Makos have a lateral keel at the base of the tail which allows the fish to efficiently oscillate its tail fin from side to side. In lateral view the base of the tail is narrow – in dorsal / ventral view – the tail base is broad. Salmon Sharks (Lamna ditropis), Porbeagles (Lamna nasus) and Great White Sharks (Carcharodon carcharias) have this same feature – it is all about efficient locomotion – hydrodynamics which submarine designers envy. Even the Ninespine Stickleback (Pungitius2) has this basic tail structure – but on a far smaller fish. Evolution is awesome.
Enough fish worship – back to the task at hand. Preservation of a large fish. You have to make sure the internal organs and muscles fix – and since formaldehyde takes time to infiltrate tissues – you inject 10% formaldehyde deep into the muscles to make sure the specimen fixes from the outside in, and inside out.
If the specimen does not fix fairly rapidly – then decay of the tissues begins. The specimen degrades and gas is produced. A gas-filled specimen displaces fluid and can result in a bit of a mess in the lab. When I was a student, we put a sizable sample of suckers in a vat of formaldehyde, closed the lid, and then left them to fix. Oily suckers are always a challenge to fix, and these were no exception. They bloated over night and displaced formaldehyde – which spilled out of the vat. The spill was large enough to draw the attention of the University of Manitoba’s Workplace Health And Safety team. Ooops.
Reptiles also can be tricky to fix – their skin slows the uptake of formaldehyde. As a dewy-eyed student I was keen to check out all the specimens in the vertebrates lab – and was particularly happy to find a forgotten jar with dark brown glass – a mystery. I had to know what was inside. When I reached in and grabbed the snake – it simply fell apart – ribs straining through my fingers. The mouth and cloaca allowed formaldehyde to enter and so the snake’s head and tail preserved well. Its body though, had rotted from the inside out and was mush.
The rattle from the rotten Pacific Rattlesnake (Crotalus oreganus).
I now use a needle to perforate reptile legs and tails to make sure formaldehyde infiltrates everywhere. I also inject 10% formaldehyde into the body cavity to make sure the internal organs of reptiles fix rapidly.
This mako shark was no different – I injected about 500 ml formaldehyde into the body cavity to make sure the internal organs fixed well – then left for the weekend. After a day in formaldehyde, the body already was rubbery and well on its way to making a decent specimen (yes I came to work on a Saturday to check on my precious). It also was not floating – that is a really good sign that the specimen is fixing well and not filling with decay gases.
The mako shark after a day in formaldehyde.
Once the mako shark is fixed (maybe three weeks in formaldehyde just to be sure), then it will get a rinse in water for a few days, and will go into a vat of alcohol for permanent storage. Alcohol is far easier on the eyes and nose than formaldehyde. Ethanol or Isopropanol are our preservatives of choice. Call me crazy, but I am guessing this shark will be a popular item during museum collection tours, so it better be stored in a manner that is fairly safe for visitors. I may as well get a few more larger fishes preserved while the formaldehyde vat is fresh – next up is a 1 meter Blue Shark (Prionace glauca) and a similarly sized Pacific Sleeper Shark (Somniosus pacificus).
Mighty Mouth post
Museums contain the commonplace, normal, typical specimens as well as the specimens we call TYPES which serve as the golden standard when doing systematics research. But the real attention goes to the oddities – they seem to naturally draw your eyes away from all other specimens. Leucistic birds, albinos, a marmot with overgrown incisors, an Orca with nasty dental issues, an Orca with spinal deformity – these are the specimens that get the WOW vote on collections tours.
Albino Starlings in the Royal BC Museum Ornithology collection – abnormal specimens certainly do catch your eye.
A year or so ago we were clearing out an area we referred to as Room 17 (our version of Area 51), and found jaw fragments from a Sperm Whale mixed with the bones of other whales. Sperm Whales have teeth along the lower jaw but no teeth along the upper jaw and Sperm Whale jaws are long and straight. It does not take a scientific eye to notice what is wrong with these jaws.
The section of Sperm Whale jaw in the Royal BC Museum collection.
Both dentary bones are hooked to the left and it looked like the teeth were fairly normal with decent sized sockets. We have no idea what the upper lip looked like – but I assume these jaws just hooked out of alignment and hung out to the side of the animal. What a drag that would have been. The jaws are large – so this animal was able to feed – the teeth towards the back of the jaw probably functioned normally and it certainly could have performed suction feeding to catch fishes and cephalopods.
Strangely enough, there is no information with these jaws to say when and where the animal was caught. Whaling here in BC ended within my lifetime (including the live capture of Orcas as a form of whaling – some would say jailing) – so I can only assume this jaw was collected pre-1970s when whaling stations were still actively processing Sperm Whales.
Sperm Whales with normal straight jaws (Image A-09221 (top) and Image A-09220 (bottom) courtesy of the Royal BC Museum and Archives).
Jaw deformities are not that rare in Sperm Whales – there are several reports published and some of the deformities are shocking – some are stubby, others in a tight spiral like a conispiral snail shell (see: Murie 1865, Thomson 1867, Nasu 1958, Spaul 1964, and Nakamura 1968).
This specimen is not cataloged in the Royal BC Museum Mammalogy collection, there is no record in our database, and no mention in the museum’s annual reports. Perhaps whaling records will mention this animal – I can’t imagine this whale was processed in the ‘fishery’ and the set of jaws saved with no comment made of the deformity. Time to go CSI on this dentary record.
To dig deeper:
MURIE, J. 1865. On deformity of the lower jaw in the cachalot (Physeter macrocephalus, Linn.). Proceedings of the Zoological Society of London, 1865: 396-396.
NAKAMURA, K. 1968. Studies on the sperm whale with deformed lower jaw with special reference to its feeding. Bulletin of the Kanagawa Prefecture Museum of Natural History, 1: 13-27.
NASU, K. 1958. Deformed lower jaw of sperm whale. Scientific Reports of the Whales Research Institute, 13: 211-212.
SPAUL, E. A. 1964. Deformity in the lower jaw of the sperm whale (Physeter catodon). Proceedings of the Zoological Society of London, 142: 391-395.
THOMSON, J. H. 1867. Letter relating to the occasional deformity of the lower jaw of the sperm whale. Proceedings of the Zoological Society of London, 1867: 246-247.
Frost-Free Footwear post
When reptiles and amphibians take shelter from the cold, they seek refuge above freezing, but not too warm – maybe 2 to 4°C. If it is too cold, tissues freeze and for most animals, this is fatal. Death comes from ice crystal growth which essentially shreds body cells at a microscopic level. Some animals like the Wood Frog (Rana sylvatica) are freeze–tolerant, and consequently are our northern most ‘herpetile’ along the coast of the Arctic Ocean/Beaufort Sea.
Wood Frog photographed in a ditch near Winnipeg.
If the refuge is too warm, the animal’s metabolism burns stored fat and the animal loses weight. My father had a pet Hermann’s Tortoise (Testudo hermanni) when he was a child – and in winter, his parents put the tortoise in a box, surrounded it with hay, and placed it in the boiler room to hibernate. The boiler room was too warm for hibernation – the tortoise starved or died of dehydration. Consequently, my grandparents bought a new tortoise each year after burying its ‘hibernating’ predecessor. I am not shore how many tortoises they went through.
Hermann’s Tortoise image from Wikipedia.
On March 5th (2019) I received a set of photos of a frozen adult Wall Lizard found by John Hunter, a Colwood resident. It appeared that the cold, frosty nights of Late February and Early March 2019 had claimed at least one lizard life. The lizard had taken refuge in John’s gardening shoes. Its body was placed on a rock in the garden – presumably nature would deal with the remains. March 6th – the lizard awoke and ran off.
Resurrection? No. The Common Wall Lizard (Podarcis muralis), like the Wood Frog, has a physiological ace up its sleeve. It can freeze up to 28% of its body water and still survive. As long as the cold snap is not too long or too severe, they usually survive without trouble. The mild winters of southern Vancouver Island were almost tailor-made for these invaders.
However, shoes obviously were not ideal shelter from the cold. Shoes keep our feet warm because our feet produce heat – the shoe only slows heat loss to the environment. Has anyone ever said to you, “Here, this blanket will warm you up.” Truth is, a blanket doesn’t provide heat, only slows heat loss – just like winter boots. With no source of heat, and with the open cuff/collar, footwear would act more like a sci-fi cryo-tube than a cozy refuge. At best the shoes shielded the lizard from scavengers.
I left my gardening boots outside last week – they were a bit too muddy to bring indoors. But since lizards are still about 60 meters north of my garden – I don’t think I will find any lost souls lining the insole.
Lettuce Travel post
Lettuce is shipped to Canada regularly. Plastic-wrapped-produce crosses our border every day – it is inspected and then it goes to grocery stores across the province. The lettuce then gets purchased, bagged and taken home – sometimes for sandwiches, salads, or maybe for juicing.
Green goodness at a local grocery store.
This November 27th, a bag of leafy goodness was opened after crossing the international border with a stowaway – a small frog in lettuce from California. It emerged – and was taken to the local SPCA. From there it was sent to me at the Royal BC Museum for identification.
The stowaway was sent to me in a container filled with damp moss.
On first glance this refugee looks like our Pacific Chorus Frog (Pseudacris regilla) which ranges south of BC to California. The taxonomy of the Pacific Chorus Frog is quite contentious though. Historically only one species was defined – P. regilla. In recent years, mitochondrial DNA suggested three species exist in California in what was once a wide-ranging Pacific Chorus Frog. Based on mtDNA, our Pacific Chorus Frog was thought to only range into extreme northwestern California. To the south, the Sierran Chorus Frog (P. sierrae) ranged across central California, and Baja California Chorus Frogs (P. hypochondriaca) were scattered across southern third of that state. If that wasn’t enough to upset a frog’s personal identity, work in 2016 placed the Pacific Chorus Frogs in a new genus Hyliola. Then in 2017, after referring back to a 2014 analysis of nuclear DNA, the three species were once again lumped into Pseudacris regilla. Or is it Hyliola? I bet the frog is confused too.
The range of the three chorus frog species based on mtDNA, from: http://www.californiaherps.com/frogs/maps/pregillamap3species3.jpg
Call me lazy, but if they are all lumped into one species – P. regilla – that makes my life easier. If the Pacific Chorus Frog was split into three species, then either I’d need to take a tissue sample to get an identification (and the frog would not enjoy that), or I’d need to know exactly where the lettuce came from. Odds are grocery records are pretty tight in this era of E. coli-tainted tracheophytes, but I have some doubt we’d ever know exactly where a given bag of lettuce originated.
A Pacific Chorus Frog from just north of the Nighthawk border crossing in the Okanagan.
Let’s just assume we are lumping all the Pacific coast Pseudacris into one species – then this refugee regilla is the same species as our Chorus frogs in BC. If this is the same species, can I just let it go? No way. It is genetically distinct since it comes from so far away, and there always is the risk of disease transmissions posed by exotic frogs. At least this Californian frog didn’t come from a pet shop where it could encounter a range of other exotic frogs and their diseases.
To be honest, I am really impressed that the frog was contained in the first place – people have a habit of releasing stowaways rather than turning them in for examination. Years ago a couple returned home from Mexico and found a red and black snake in their luggage. The snake didn’t seem well, but they released it somewhere in Metchosin. Presumably that snake died, but if it had been a gravid female, it could have deposited 7-10 (or more) eggs, and we’d have an instant population. What species had infiltrated their luggage? I have no idea – it could well have been venomous. When I was an undergrad student, a red and black snake appeared in the pet trade in Winnipeg – it was labeled Honduran Milk Snake and looked like this. I assumed it was harmless based on the old rhyme:
Red-on-Black, Safe for Jack.
Red-on-Yellow, Kill a fellow.
I was wrong – the snake in the pet shop was rear-fanged and bit me. It was my first (and currently only) venomous snake bite. Bottom line is: Better to be safe than sorry. And as a member of IMISWG (Inter-Ministry Invasive Species Working Group) we always say that it is better to not release something, than try to clear out exotic species later. Turn in stowaways to your local SPCA or Natural Resource Officers. It is safer for the environment. Frogs obviously are harmless, but if you think you have something dangerous in your groceries – an Eyelash Viper in a bunch of bananas or a Brown Widow Spider in your Californian cauliflower – call your local Natural Resource office and arrange for a professional to remove the offending animal.
Above all else, don’t let it loose.
Fieldwork 2018
This year’s fieldwork was our 17th in the alpine of the northern B.C. We made collections from six mountains. The area is so vast and remote and access is difficult, thus few if any biological inventories have been undertaken in many large areas. Many peaks and lakes have no names; in fact there are no names on entire mountain massifs. I feel like we are to some degree just ‘scratching the surface’ of what is out there.
We again worked together with the insect and spider experts at the museum. And we followed our typical approach of setting up camp for 2-3 days and collecting specimens of every species we encounter, being intentional to reach as many different habitats as possible.

Typical day of hiking (dotted line) and collecting, striving to reach as many habitats as possible. Upper part of figure documents elevation changes during the day. Three of the four plants in this illustration were encountered only once, highlighting the importance of having several botanists in the field and covering as much ground as possible.
At one mountain, Mt. Whitford, we were joined by two staff and a contract photographer of the Yellowstone to Yukon Conservation Initiative https://y2y.net/about-us and their guest freelance journalist who produces pieces for both CBC and NPR. At a second mountain, south of Tumbler Ridge, we were joined by two staff members of the Tumbler Ridge Geopark, http://tumblerridgegeopark.ca/. What is a Geopark? According to their website “A UNESCO Global Geopark is an area recognized as having internationally significant geological heritage.” These groups are all interested in knowing as much as possible about the biota of these areas and we will share everything we learn with them.
As we have in the past, we contacted the local indigenous groups and informed them of our work and will provide them species lists when the identifications are complete.
We are often asked if we notice any of the effects of climate change during our fieldwork. Treeline is controlled by temperature, not elevation and is highest at the equator – where temperatures are warmer at higher elevations – and becomes lower and lower further north and south. One likely consequence of a warming planet is that forests will advance into the alpine, reducing the available habitat for tundra plants that generally require open, i.e. non-shaded habitats.

Young trees in alpine meadows at 1750 m north of Laurier Pass, Graham-Laurier Provincial Park. Note the dark skies of a storm about to dump
For a number of years I’ve noticed small trees in the alpine and of course wonder if their appearance is related to climate change as a consequence of global warming. I’ve also noticed the absence of dead trees. The absence of dead trees may mean that tree populations in the alpine are relatively young, compared to lower elevation forests where trees have been growing and dying for thousands of years.
Two hypothetical graphs of tree age versus elevation curves. The upper one portrays trees that become gradually younger with elevation gain, much as one might expect if treeline was moving higher gradually. The hypothetical curve on the lower graph describes a more abrupt change, i.e. what one might expect if tree line had been stable for a relatively long period of time, slowly moving to higher elevations, then suddenly and relatively recently, young trees became established higher than before.
But a further question is this: have the young trees in the alpine arrived relatively suddenly and recently because of recent rapid increase in global temperatures, or are they gradually moving upwards due to a long term warming trend that has been taking place ever since the end of the Pleistocene, ca. 13,000 years ago? Dating trees by their growth rings could provide the answer by measuring the ages of trees along an elevation gradient from well below tree-line into the alpine. I suspect this kind of research is underway.
Every year we seem to encounter botanical surprises, either range extensions or species that we haven’t seen before. I like these kinds of discoveries because distribution patterns tell us something about the history of the landscape and when those distribution patterns are found to be different from what was previously known, the background story might change.
One notable collection this year was Dodecatheon frigidum (northern shootingstar) that we collected in northern Graham Laurier Provincial Park, about 200 km south of where it has been collected previously near the Alaska Highway. We’ve visited 8 mountains in the intervening area and have not encountered this species. What does this occurrence mean? Have we merely overlooked it in other areas or is it in fact not present for this 200 km distance?
Another interesting find was Claytonia lanceolata (western spring beauty) which I saw in the alpine for the first time. Previously I had encountered it at lower elevations in Botanie Valley north of Lytton. Indigenous people in the southern interior of BC eat the tubers either fresh or cooked. Perhaps indigenous people in the Tumbler Ridge area also eat the tubers. I haven’t had a chance to ask local people or to investigate the literature.
When is a holotype not a type specimen?
When it was never published in the first place.
The Royal BC Museum fish collection contains a specimen which had been locked securely in one of our type cabinets since the 1980s. It was designated as the holotype for a new species – Sebastes tsuyukii – there was even a manuscript noted on the specimen label (Westreim and Seeb 1989). It sounded legit – and no one checked until recently.
Jody Riley – my ever diligent volunteer – flagged this record when she was re-organising the fish collection. She checked what is in our old paper catalog, checked the electronic database, then looked to see if the actual specimen exists. When Jody hit Sebastes tsuyukii, and found no record of the species online, yet here in her hands was the jar with a big yellow tape label saying Holotype for Sebastes tsuyukii, she knew something was fishy.
 Did the manuscript stall during composition, submission, or revision? Who knows.
Did the manuscript stall during composition, submission, or revision? Who knows.
In the end, we can take this large jar out of the cabinet designated for type specimens, Sebastes tsuyukii now is a nomen nudum (a naked name), and I can delete the species from the taxonomy in our museum database. Some database problems are easy to solve.
But this reminds me to get my fingers in gear and type the type descriptions for species I have yet to publish.
Orca Abscessed post
Nitinat (T12A) was a well known Orca along the BC coast. Born in 1982, he was a fixture along the BC coast and an active participant in the 2002 attack on a Minke Whale in Ganges Harbour, Saltspring Island. This animal – with its characteristically wavy dorsal was found dead off Cape Beale near Bamfield, September 15th, 2016. Funds weren’t available to prepare the entire skeleton, so I had to settle for the skull and jaws.
As you can imagine, the head of an orca would pop the frame of any domestic chest freezer, and it blocked the aisle of the walk-in freezer at the Pacific Biological Station in Nanaimo. It was also no small feat to fork-lift the head into the museum’s van, and then get it back out of the van and wheel it to the museum’s walk-in freezer. It also was a surreal experience driving around with an orca head in the truck. The head is heavy – and slippery – and difficult to tie down – so I drove smoothly to avoid having the head roll around behind me. Imagine explaining to an insurance company how an orca head caused you to lose control of your vehicle?
Nitinat’s head was prepared by Mike DeRoos and Michi Main – their internationally acclaimed business, Cetacea, focuses on cleaning and articulating whale skeletons. While preparing this skull for burial, they noticed that Nitinat had broken teeth. Given that I broke a molar on a frozen Reese’s Piece in a Dairy Queen Blizzard, I could imagine how Biggs Orcas could break a tooth when biting down on a sea lion or elephant seal. Large pinnipeds have dense bones.
Once the skull was cleaned, Mike and Michi found that not only were teeth broken, there also is a nickle-sized hole in the palate and many teeth were abscessed. The hole in the palate is particularly interesting. It has smooth sides and so certainly had healed before Nitinat’s death. Was it a puncture and the source of the infection that caused the distortion of the teeth? Or was it a channel for the abscess to weep into Nitinat’s mouth (not a pleasant thought regardless).
Normal teeth (left) have a long root and recurved crown, with natural wear for their ecotype – but the abscessed teeth were stunning with their broken crown and expanded root. They almost remind me of some squash varieties that are available.
One of the teeth is so swollen that it couldn’t be removed from its distorted socket.
Red lines beside the skull indicate expanded tooth sockets – perhaps age and infection combined to create this effect. The sockets for the abscessed teeth were eroded and far larger than normal sockets (in this non-mammalogist’s opinion). Erik Lambertson made a great scale bar.
Nitinat’s teeth are enough to make anyone who has had a toothache cringe, and a dentist’s eyes pop with fascination. I am just waiting for the day someone requests to see Nitinat as the focus of a pathology research paper. For now, he is a permanent addition to the Royal BC Museum collection and will soon get his official catalog number.
I don’t know if the title of this article is an accurate way to say fork-tailed lizard in German, but the Gabelschwanz-Teufel – the P-38 Lightning (the fork-tailed devil) could take a lot of punishment and still get home at the end of a sortie. A fork-tailed lizard has a parallel story – it has taken a beating and survived.
It is common to find lizards with regenerated tails or tails that are recently dropped – with their tell-tail stump. Sometimes the tip is lost, others about 90% of the tail is lost. The regrown tail segment is never as nice as the original and has different scale patterns and colouration.
This male Wall Lizard photographed by Deb Thiessen, lost its tail near the base and the regenerated tail is obvious. Its meal had a perfect tail.
I have seen fork-tailed, even trident tailed lizards in photos – I remember images like this in the books I poured over earlier in my ontogeny. Had I ever seen one in person? Not until now. During my PhD thesis work, the only fork-tails I thought about were thelodont fishes known from Early Devonian rocks of the Northwest Territories.
This July, Robert Williams, a colleague from University of Leeds in England was here working on Wall Lizards. He was trying to determine if our native Northern Alligator Lizards react in any way to the scent of the European Wall Lizard.
Live animals are not allowed at the Royal BC Museum, so Rob had to perform scent trials in my dining room. The lizards were held in containers in my kitchen – and I thank my wife for her patience.
The work helps give a frame of reference to reactions between the native Sand Lizard in the UK and introduced Wall Lizards, but you’ll have to wait to hear the results. While hunting Wall Lizards on Moss Rocks here in Victoria, Rob caught a fork-tailed specimen.
Since this was such a neat specimen I requested it be saved intact for the Royal BC Museum’s collection. Here is a photo of a fork-tailed Wall Lizard from England, but Rob had to come all the way to the Pacific coast of Canada to catch one.
Lost Soles post
In museum collections, space is critical. We can’t waste space. Every millimeter of shelving is critical. If you can arrange cabinets more efficiently, do it. Can you pack more jars in a given area? Do it. If you can make space. Do it.
I have been on a binge of deaccessioning lately. What is deaccessioning? It is the museum practice of removing accessioned/cataloged specimens from the collection. Once deaccessioned, we either send specimens to other museums where they are relevant, or give them to teaching collections or perhaps to nature centers. Only rotten specimens are destroyed. We try everything we can to re-purpose specimens before we resort to destruction.
This surfperch, Embitoca lateralis, is a rare candidate for destruction. It has been deaccessioned – someone had cranked the clamp too tight years ago and the glass at the apex of lid popped. Alcohol evaporated and by the time it was noticed, it was too late. If the fish in the jar could speak, they’d say, “There’s a fungus among us.”
Deaccessioning allows me to make space in the collection for new material. Since I am trying to keep the Royal BC Museum’s vertebrate collection focused on British Columbia, the eastern North Pacific Ocean and any adjacent territory, specimens with no relevance to this region obviously have my attention. Specimens with incomplete information (or no information), also flare my obsessive nature and are on my deaccession hit list. Space is created on a jar-by-jar basis.
Putting ‘incomplete information’ in everyday terms – if we are going to meet somewhere, you generally expect some level of detail. If I say I want to meet in Tofino in June, what would you say? Imagine now that I didn’t even give you my name – but still wanted to meet in Tofino in June. I am betting you’d put on your best Monty Python-esque King Arthur and say, “You’re a Loony.” Incomplete or missing data is a real issue.
My long suffering volunteer Jody found a jar of flatfish this weekend which had never been cataloged, but was in the collection. It was only a 125 ml jar – so not a huge waste of space. On closer inspection the fishes were identified (Parophrys vetulus, English Sole), there was a location (Tofino), and a date (June 1985).
Where was I in June 1985 – oh yea – just about to graduate from grade 12. Oh the 80s – I am listening to Duran Duran while typing this – RIO – the obvious choice with its maritime theme.
Yep, that was me in 1985.
Parophrys vetulus is a common fish here in BC, so it is likely you can catch them all around Tofino in June – but it would be nice to know which beach relinquished its sole. And when did it happen? Was it at night? Was it a full moon? On the 1st of the month, or mid month? Were they in ankle-deep water or at 10 meters depth? Open beach or a tidepool? Caught by hand or with a net? Inquiring minds may want to know. And with no collector noted in the hand-written label – I can’t even badger someone by email to jog their memory or review old field notes.
These are the lost soles Jody found. Is one of them yours?
To a museum, data is everything. If you collect and preserve a specimen, record as much as you can about the event. If you are giving me your sole, then tell me its secrets.
Lacertophagy post
I have said before that European Wall Lizards (Podarcis muralis) will eat smaller conspecifics – there are a few photos online from elsewhere on Earth – but until now I didn’t have solid evidence of lacertophagy (lizard eating) here on Vancouver Island.
However, this last week, Deb Thiessen took a few videos of a Wall Lizard eating a yearling Wall Lizard on her property just north of Victoria. These are really good videos and clearly show that Wall Lizards can stuff down a huge meal.
Posted by Deb Thiessen on Friday, June 1, 2018
In this first video the smaller Wall Lizard is already dead, and I suspect that the larger lizard killed it. Looks like another lizard had thoughts of stealing the meal. Sure looks like breathing is an issue while stuffing down so large a meal. Snakes solve the problem of eating and breathing by pushing their trachaea (windpipe) out of the mouth so that food does not block air flow.
Posted by Deb Thiessen on Friday, June 1, 2018
The victor looks like a male, and in the second video you can see how quickly it disposes of the tail rather than having that part of the meal hanging out of its mouth for a few days.
Almost all of the victor’s own tail had been lost some time ago. You can always see where its original tail ended and the re-growth takes over – the new tail is never as neatly patterned.
Be glad Wall Lizards aren’t the same size as Varanus prisca, otherwise we’d be on the menu.
Science is Serious post
Or if you are an astronomer, then your science is Sirius. If you are a geologist, then your science is pretty gneiss. Don’t take science for granite.
I have been tracking Wall Lizards now for a while – and I am sure my wife will say lizard tracking has become an obsession – a serious obsession. I look at rock walls as we drive around town. I look for lizards on our weekend hikes. I watch for lacertids when I walk our daughter too and from school. Science is serious.
I have been watching the range expansion of two nicely segregated populations of lizards in Victoria – one population is about 0.63 km SSW from our house west of Hillside Mall, and the other is about 0.24 km north of us near Doncaster School – not that I have measured.
Each year I walk the perimeter of these populations to get an idea how fast lizards disperse in urban environments – again – this is serious science. Stop laughing. I can hear you laughing. Rolling your eyes does not help.
Wall Lizards seem to spread 40 to 100 meters – and it is the young ones that do the dispersing. Why? They race off to new habitat to avoid the cannibalistic tendencies of their parents. Parents with a 40 year old trekkie in the basement may want to consider this option as an incentive to get kids to move out.
Young lizards head for the relative safety of boring lawns – garden areas with lots of structure are occupied by hungry adults. Homeowners sometimes claim their lawn is crawling with young lizards in August – when all the summer’s eggs have hatched. In contrast, adults are relatively sedentary – once they find good sunny, rocky (complex) territory, they tend to move very little from year to year.
Now imagine my surprise when I walked up my driveway last night (May 23rd, 2018) and heard the characteristic rustling sound of a lizard in our food forest (yes, the lawn is gone and we have a food forest – the entire front garden is devoted to plants we can eat, and plants that attract bees to pollinate the plants with edible bits – but I digress). The lizard I found is at least 0.24 km from the nearest known population of lizards in my neighbourhood, and is an adult – with a perfect tail too – must have lived a charmed life free of bird and domestic cat attacks. Did this adult go walkabout? I doubt it.
The new colonist in the food forest at UF1510 (yes, as sci-fi nuts we gave our place a code name Urban Farm1510)…
Furthermore, the lizards nearest to my house are not brightly coloured – in fact they are kind of drab as far as Wall Lizards go. But our new lizard is gorgeous – more like ones from Triangle Mountain or farther north on the Saanich Peninsula.
This male is from Durrance Road – far more colourful than the ones near Doncaster School or Hillside Mall.
Is this a case of seriously good science prank? Was this a drive-by lizarding? Did a neighbour just buy some new garden supplies and a stow-away lizard emerged to find utopia in our food forest? I may never know.
My daughter has named the lizard Zoom. I guess he is there to stay.
Here’s a link to a new paper by: Luke R Halpin, Jeffrey A Seminoff, and myself.
Source: Northwestern Naturalist, 99(1):73-75.
Published By: Society for Northwestern Vertebrate Biology
This new paper provides the first photographs of a Loggerhead Sea Turtle (Caretta caretta) from west of Vancouver Island. The species has been spotted in the region before and as far north as Alaska, but until now, there were no photographs or specimens as solid evidence.
While the photos in this paper are black and white – the original photographs by Luke Halpin are color and van be viewed upon request. PDFs also are available – just send me an email.
British Columbia is now within the range of 4 species of marine turtle. This Loggerhead survived into February of 2015 because of the unusually warm water in the eastern North Pacific Ocean (the Warm Water Blob), whereas Green Sea Turtles (Chelonia mydas) and Olive Ridley Sea Turtles (Lepidochelys olivacea) wash up dead (or near dead) in early winter. Unfortunately, the fate of the Loggerhead from 2015 is unknown.
Nanaimo Invasion post
Years ago after coming off parental leave, I found a series of photographs of Wall Lizards and a Google Earth image of a road intersection marked to show locations for a lizard colony. Quick search in Google Earth showed that this colony was in Nanaimo. I fired off a fast blog article to generate interest and get people looking for Wall Lizards.
It worked. Reports came in.
Jump forward a few years and now that street (Flagstone – site 1) is crawling with lizards according to eyewitnesses. But we now also have another site (2) along the Nanaimo Parkway near Douglas Avenue and Tenth Street. Oh wait, there’s also a third site (3) in the Chase River Estuary Park, and as of this weekend, there’s another (4) – way north of the rest along Arrowsmith Road. The report of the lizards in the Arrowsmith Road area was accompanied by video – there was no doubt as to the identification of those lizards – and that was a big jump from previous known occurrences.
 Two other records – one along Enfer Road near Quennell Lake, and along Leask Road south of Nanaimo have yet to be verified with specimens, photographs or video.
Two other records – one along Enfer Road near Quennell Lake, and along Leask Road south of Nanaimo have yet to be verified with specimens, photographs or video.
There you go Nanaimo, the invasion has picked up pace. Keep your eyes peeled for lizards with a green tint to their scales, minute scales on their back, and generally more delicate proportions than the native Alligator Lizard.
Look at this post to help identify any lizards in your neighborhood.
If you find suspected Wall Lizards – email me at: ghanke@royalbcmuseum.bc.ca
If you find a lizard that is not a Western Skink, Northern Alligator Lizard, or European Wall Lizard – I definitely want to know about it.
Please record the date and street address (or prominent landmark) to pin down exactly where the lizard was seen. A photo would be really helpful to confirm the lizard’s identification. Happy hunting.
The Special Issue of Aquatic Invasions on “Transoceanic Dispersal of Marine Life from Japan to North America and the Hawaiian Islands as a Result of the Japanese Earthquake and Tsunami of 2011” has been published as Volume 13, Issue 1, pages 1-186 (totalling more than 220 pages with supplementary files).
The Special Issue includes the 14 papers (all Open Access) by 39 researchers, including Dr. Henry Choong, Curator, Invertebrate Zoology.
Funding support was provided by the Ministry of the Environment (MOE) of the Government of Japan, through the North Pacific Marine Science Organization (PICES), which made this Special Issue possible.
The Introduction to the Special Issue provides vignette summaries of examples of notable Japanese Tsunami Marine Debris objects, and also details contributions to the knowledge of Japanese and North Pacific marine biota as a result of JTMD research. These contributions include new species, new species records for Japan, and a rediscovered species (last documented in 1929). A final summary table in the Introduction provides examples of molecular genetic contributions to our understanding of JTMD biodiversity.
The JTMD project, which commenced in 2012, and which is now entering its 6th year of research, as we continue to monitor for the potential arrival of living species, 7 years after the tragic disaster of March 11, 2011.
Calder, D.R., Choong, H.H.C., Carlton, J.T., Chapman, J.W., Miller, J.A., and Geller, J. 2014.
Abstract
Fourteen species of hydroids, including two anthoathecates and 12 leptothecates, are reported from the west coast of North America on debris from the tsunami that struck Japan on 11 March 2011. Six species were found on a dock that stranded at Agate Beach, Newport, Oregon, five from a boat at Gleneden Beach, Oregon, four from a dock in Olympic National Park, Washington, and two from a boat in Grays Harbor, Washington. Obelia griffini Calkins, 1899, the most frequently encountered species, was collected on three of the four derelict substrates. Eight of the species are known to be amphi-Pacific in distribution. Of the rest, at least five (S tylactaria s p . ; Eutima japonica Uchida, 1925; Orthopyxis platycarpa Bale, 1914; Sertularella sp.; Plumularia sp.) are not previously known from the west coast of North America. Hydroids of E. japonica occurred as commensals in the mantle cavity of the mussel Mytilus galloprovincialis Lamarck, 1819. Obelia griffini, O. gracilis Calkins, 1899 (not its secondary homonym Laomedea gracilis Dana, 1846) and O. surcularis Calkins, 1899 are taken to be conspecific. Of the three simultaneous synonyms, precedence is assigned to the name O. griffini under the Principle of the First Reviser in zoological nomenclature. The species is sometimes regarded as identical with O. dichotoma (Linnaeus, 1758).
Henry H. C. Choong* and Dale R. Calder
Invertebrate Zoology Section, Department of Natural History, Royal Ontario Museum,100 Queen’s Park, Toronto, Ontario, Canada, M5S 2C6
*Corresponding author
Abstract
The leptothecate hydroid Sertularella mutsuensis Stechow, 1931 is reported on debris from the 2011 Japanese tsunami that came ashore on 5 June 2012 at Agate Beach north of Newport, Oregon. Its discovery on a barnacle (Semibalanus cariosus) from a derelict floating dock originating at Misawa, Honshu, confirms the capability of successful transoceanic dispersal for this species. We compare our specimens to Stechow’s syntype material of S. mutsuensis in collections at the Zoologische Staatssammlung München, and designate a lectotype and paralectotype of the species.
Key words: Leptothecata; hydroid; lectotype; transoceanic dispersal; anthropogenic debris; Oregon coast
The Power of 1 post
It is always satisfying to update taxonomy in the museum’s database or find and correct mistakes. This week I spent some time sorting out details on Royal BC Museum specimens of California Yellowtail (Seriola dorsalis) and Great Amberjack (Seriola lalandi). Turns out that since these fishes were collected, Seriola dorsalis has been sunk, and all our fishes are Seriola lalandi (as noted by Gillespie 1993). This carangid fish is known to move into our waters in warmer years.
While reviewing what we knew about the first BC specimen (979-11312) it became obvious that the Royal BC Museum’s database was missing some information for that fish. Fortunately, this information was easily updated – the original report was published in the Royal BC Museum’s extinct periodical Syesis (see Nagtegaal and Farlinger 1980).
Drawing of Seriola dorsalis – oops lalandi (979-11312) by K. Uldall-Ekman.
In fixing that record, I noticed that some online sources had given incorrect coordinates for this fish. Contrast the capture location of 54°35’N, 131°00’W in Caamaño Passage as reported by Nagtegaal and Farlinger (1980), with online sources which state the fish was caught at 54°35’N, 31°00’W. That missing 1 in the reported longitude determines which ocean is linked this fish.
The takeaway message? Always check the original paper rather than relying on internet sources. Precise data is everything – and in the words of a well known scoundrel: “Without precise calculations we could fly right through a star, or bounce too close to a supernova and that’d end your trip real quick, wouldn’t it.” Or in this case, you’d be landing southwest of Iceland to look for Great Amberjack.
References:
Gillespie, G.F. 1993. An Updated List of the Fishes of British Columbia, and Those of Interest in Adjacent Waters, with Numeric Code Designation.Canadian Technical Report of Fisheries and Aquatic Sciences 1918. 116 p.
Nagtegaal, D.A. and S.P. Farlinger. 1981. First record of two fishes, Seriola dorsalis and Medialuna californiensis, from waters off British Columbia. Syesis 13:206 –207.
I’ve received a steady series of emails this year detailing European Wall Lizard locations here on Vancouver Island, and it’s now April and wall lizards certainly are active. However, an email arrived April 11th which gave me a WTH (What The Herp) moment. The email contained a beautifully focused photo of a new turtle for BC. Then it occurred to me that I’d lost count of how many turtle species have been dumped here – unwanted pets that outlived the interest of their owners.
I really like when people send me photos of things they think are unusual – and this week’s email was no exception. We know that Red-eared Sliders (Trachemys scripta elegans), Yellowbelly Sliders (Trachemys scripta2), and a Map Turtle (Graptemys sp.) have been dumped in Goodacre Lake, and Red-eared Sliders into Fountain Pond, but this new turtle photographed by Deb Thiessen (see below) certainly was not just an odd coloured slider, nor was it another map turtle. As an aside, I haven’t had a chance to catch the Map Turtle in Beacon Hill Park to get a good look at it, but I have seen it at a distance, and ID’ed it based on photos from Darren Copley and James Miskelly. It looks like a False Map Turtle (Graptemys pseudogeographica). I think that’ll be a summer goal, to get good photos of that turtle to be sure which species it represents.
A Peninsula Cooter (Pseudemys peninsularis) from Fountain Lake, Beacon Hill Park, Victoria, BC. Photograph by Deb Thiessen, retired CRD Parks naturalist.
As you can see from Deb Thiessen’s photograph, this new turtle has a large shell for the size of the head, and the stripes on the neck are crisp, and bold yellow offset by black. The bold markings to me suggested Peninsula Cooter (Pseudemys peninsularis). The short claws on its forelimb indicate it is female. Males would have claws double the length of those in the photo. This animal is way outside its normal range – Peninsula Cooters are from Florida.
This animal brings our list of pet turtles to 10 species abandoned in BC ponds and lakes – that we know of. Here is the list I have of turtles that have been found in BC – way out of their native range – and (shockingly) it parallels species available in the pet trade here in BC.
Trachemys scripta (Pond Slider – both T. s. elegans and T. s. scripta)
Pseudemys peninsularis (Peninsula Cooter)
Pseudemys concinna (River Cooter)
Chrysemys picta marginata (Midland Painted Turtle, possibly also Southern Painted Turtles, C. p. dorsalis)
Graptemys pseudogeographica (False Map Turtle)
Emys orbicularis (European Pond Terrapin – always did like the word Terrapin – a bit of nostalgia from my British roots)
Chinemys reevsi (Reeve’s Turtle)
Malaclemys terrapin (Diamondback Terrapin)
Apalone spinifera (Spiny Softshell Turtle)
Chelydra serpentina (Common Snapping Turtle)
Fortunately most turtles are dumped one at a time and do not reproduce. Sadly though, I can’t say the same for the Red-eared Sliders – they now can reproduce successfully here in British Columbia (I have two pets from the first successful clutch found on the south coast of BC, ca. January 11, 2015). Red-eared Sliders now are common in artificial and natural ponds and in lakes here in southwestern British Columbia – and until recently, we were sure that each adult represented an abandoned pet (or maybe the occasional escapee). Now males are finding females. Females are finding decent nesting locations. And eggs are surviving to hatch.
Knowing that sliders can breed here, I stopped to check whether sliders and cooters can hybridize, and it has been suggested to be possible – but no solid proof. And since it is better to be safe than sorry… Does anyone know how to neuter a Cooter?
This time of year, my garden is one big mudslide. Sunny days with a blue horizon are not that common here on Vancouver Island in winter – but when they occur, we certainly enjoy them. So do our slim little European Wall Lizards.
This January and February I collected lizards which were active when the air temperatures were between 5° to 7°C. As a survivor of the Canadian prairies, collecting lizards in winter seems about as strange as an empty room in a museum collection.
I found lizards along Derby Road in my neighborhood, on Moss Rocks, at Gardenworks Nursery in the Blenkinsop Valley – winter lizard activity is nothing new here on Vancouver Island.
Lizards were found in south-facing locations with full sun exposure and when caught, were very warm to the touch. It is obvious that they are effective solar collectors and can elevate their body temperatures well above that of the chilly air – even when it is a bit windy. It is not uncommon to see lizards only exposing their head for a while, then the rest of the body. Perhaps this is a low-risk way to warm blood via blood vessels in the throat before they venture out and deal with intruding conspecifics. I haven’t seen any wall lizards feeding in winter – but that doesn’t mean they don’t. I’ll have to examine museum specimens to see what’s in the stomachs of winter-caught lizards.
An adult European Wall Lizard caught on Derby Road in Victoria, February 26th, 2018.
As of this February, the Royal BC Museum collection has 30 lots of European Wall Lizard specimens representing surface activity for each month of the year. Some people collect trading cards to get a complete set, I collect lizards to get one per month. Wall Lizards are active in winter as far north as Denman Island, and given that range, probably could extend further north of Campbell River in areas with a warm microclimate.
The collection of lizards for each season put a song from 1971 into my head – so I reworded the chorus a bit…
Winter, spring, summer or fall,
All they have to do is crawl,
And I’ll be there, yes I will,
Their spread has to end.
Tristan A. McKnight & Robert A. Cannings
Abstract: Stackelberginia cerberus sp. nov. (Diptera: Asilidae) is described from the Amargosa desert (USA: Nevada) and compared to related taxa. This is the first record of the genus in the Western Hemisphere; other species live in the deserts of central Asia. Stackelberginia Lehr is proposed as the sister taxon to Lasiopogon Loew in the subfamily Stichopogoninae based on morphological characters and a Bayesian species tree estimated from one mitochondrial (COI) and three nuclear protein-coding loci (AATS, PEPCK, wingless). Stackelberginia has the medially divided epandrium and rotated hypopygium of Lasiopogon, but the facial gibbosity is flat, macrosetae of thorax, head, and legs are unusually long, and phenology peaks in late autumn.
Key words: Stichopogoninae, robber fly, assassin fly, species tree, molecular, Palearctic
Joel. F. Gibson
Abstract: The thick-headed flies (Diptera: Conopidae) are rarely observed parasitoids. Confirmed hosts include many species of bees and wasps. Often collected from flowers, conopids may serve as either pollinators or pollinator predators. The last detailed checklist of the Conopidae of British Columbia was published in 1959. An updated checklist for British Columbia, the Yukon, and Alaska is presented based on over 1,000 specimens and specimen records. Geographical distribution, using an ecoprovince approach, is documented for each of 26 species in the region. Host, plant association, and hilltopping behavioural records based on past literature and new observations are also included. An identification key to all species recorded is included.
Key words: parasitoid, biogeography, plant associations, host associations, Nearctic
Colin J. Curry, Joel F. Gibson, Shadi Shokralla, Mehrdad Hajibabaei, and Donald J. Baird
Abstract: We reviewed the availability of cytochrome c oxidase subunit I (COI) sequences for 2534 North American freshwater invertebrate genera in public databases (GenBank and Barcode of Life Data Systems) and assessed representation of genera commonly encountered in the Canadian Aquatic Biomonitoring Network (CABIN) database. COI sequence records were available for 61.2% of North American genera and 72.4% of Insecta genera in public databases. Mollusca (73.9%) and Nematoda (15.4%) were the best and worst represented groups, respectively. In CABIN, 85.4% of genera had COI sequence records, and 95.2% of genera occurring in >1% of samples were represented. Genera absent from CABIN tended to be uncommon or members of groups not routinely used for biomonitoring purposes. On average, 94.1% of genera in well-identified samples had associated sequence data. To leverage the full potential of genomics approaches, we must expand DNA-barcode reference libraries for poorly described components of freshwater food webs. Some genera appear to be well represented (e.g., Eukiefferiella), but deposited sequences represent few sampling localities or few species and lead to underestimation of sequence diversity at the genus level and reduced confidence in identifications. Public COI libraries are sufficiently populated to permit routine application of genomics tools in biomonitoring, and ongoing quality assurance/quality control should include re-evaluation as new COI reference sequences are added or taxonomic hierarchies change. Next, we must understand whether and how established biomonitoring approaches can capitalize on high-throughput sequencing tools. Biomonitoring approaches that use genomics data to facilitate structural and functional assessments are fertile ground for future investigation and will benefit from continued improvement of publicly available sequence libraries.
Key words: COI, invertebrates, biomonitoring, high-throughput sequencing, DNA metabarcoding, identification,
genus, Biomonitoring 2.0
Robert A. Cannings & Russell V. Pym
Archilestes californicus McLachlan (California Spreadwing) is a large damselfly native to western North America, ranging from Washington and Idaho south to New Mexico, Arizona and California and, in Mexico, to Sonora and Baja California Sur (Paulson 2011; Westfall and May 2006). This note records the species for the first time in Canada—from three sites in the southern Okanagan Valley, British Columbia (BC; Figure 1).
Russell Pym saw several males and females at a small, shallow, artificial pond at the end of an artificial stream near the entrance to the Liquidity Winery at 4720 Allendale Road, Okanagan Falls, BC (49.32553°N, 119.54993°W). He observed them from 13:00 to 14:00 PDT on 26 September 2016; one male was photographed (Figure 2). From 16:30 to 17:00 PDT the same day, he recorded a female in knee-high grass, three to four metres from the shore of a dugout pond across the road from Walnut Beach Resort, 4200 Lakeshore Drive, Osoyoos, BC (49.01825°N, 119.43580°W). Cattail (Typha latifolia) and willows (Salix spp.) lined the pond margins.
You and Yew post
Plants make molecules that chemists could never imagine. Chemical poisons that deter herbivores are advantageous for organisms that can’t move. For humans, depending on the dosage, these molecules can be either poisonous or medicinal. The small shrub circled here is Pacific Yew. In 1962 it was discovered that its tissues, especially the bark, contain ‘taxol’ effective in treating cancer. It took many kg of bark (in 1993 34,000 kg were harvested in BC) for a single dose – and killing the tree. Now the drug is synthesized in a lab using precursors from the needles. The seed is surrounded by red, fleshy tissue called an ‘aril’, sometimes incorrectly called a ‘berry’.

The small shrub circled here is Pacific Yew.

The seed is surrounded by red, fleshy tissue called an ‘aril’, sometimes incorrectly called a ‘berry’.
Swordfishes in BC post
In an earlier post I mentioned that Luke Halpin was out surveying marine mammals and birds from the deck of the CCGS John P. Tully, and spotted something totally different west of Brooks Peninsula. The fish was estimated at 3.5-4 meters in length, and was cruising against the current just below the surface.
But until the paper announcing his find was accepted by a scientific journal, I didn’t want to spill the beans and say what he had found. His research paper (Halpin et al. 2018) will be published in the spring issue of the Northwestern Naturalist.
Photo by Luke Halpin, September 5th, 2017
This picture says it all – there is no debating what this fish is – only one species that fits the bill. Swordfish are known north to the southern Kuril Islands in the western Pacific, but Luke’s find is the northern-most record for the species in the eastern Pacific and is conclusive evidence of this species right along our coast.
A Google Earth image showing where the Swordfishes from 2017 and 1983 were found relative to Vancouver Island.
A previous record from 1983 (see Sloan 1984, and Peden and Jamieson 1988) was from just inside of our exclusive economic zone (EEZ) and barely qualified as a BC fish. The 1983 specimen was caught as by-catch at 47°36’N, 131°03’W, during an experimental fishery survey by the M/V Tomi Maru. The rostrum and tail were preserved in the Royal BC Museum’s fish collection (RBCM 983-1730-001). I am guessing the edible bits in between were cut into steaks, and ended up on someone’s dinner table. At least Luke’s Swordfish was left alone and for all we know, is happily cruising south to slightly warmer water.
References:
Halpin, L.R., M. Galbraith, and K.H. Morgan. 2018. The First Swordfish (Xiphias gladius) Recorded in Coastal British Columbia. Northwestern Naturalist, 99(1): XX-XX. (pages not set)
Peden, A.E., and G.S. Jamieson. 1988. New distributional records of marine fishes off Washington, British Columbia and Alaska. Canadian Field-Naturalist, 102(3), 491-494.
Sloan, N.A. 1984. Canadian-Japanese Experiental Fishery for Oceanic Squid off British Columbia, Summer 1983. Canadian Industry Report of Fisheries and Aquatic Sciences No. 152: pp. 42.
We Three Kings post
Keep your eyes peeled for deep-sea fishes while strolling along our shores. In the last month, three King-of-the-Salmon (Trachipterus altivelis) have washed up in the Salish Sea. Two were found in September (21st and 26th) in the Oak Bay area, Victoria. One of these was still swimming when found. A third was found October 3rd in Hood Canal, in Puget Sound. The first Oak Bay specimen will be preserved for the Shaw Centre for the Salish Sea in Sidney, the second was not recovered, and the third will be preserved in the Burke Museum’s collection. The Royal BC museum has 18 Trachipterus specimens, with several of these from the Salish Sea area.
The King-of-the-Salmon from Hood Channel, photographed by Randi Jones.
Is this species new to the region? No. The species ranges from Alaska to Chile, and knowledge of this species pre-dates European arrival on this coast. Is this trio of King-of-the-Salmon a case of post-spawn mortality? A sign of change in our oceans? We don’t know. Actually, when you look at the diversity of marine fishes off our coast, there is a lot of basic biology that we don’t know. We also get Longnose Lancetfishes (Alepisaurus ferox) washing up from time to time, although it has been a few years since I have heard report of a Lancetfish in the Victoria region.
King-of-the-Salmon swim by passing a sine wave down their dorsal fin – they can get a fair bit of speed just by doing that. They can also reverse using the same fin flutter. They slowly turn by putting a curve in the body. However, in the first few seconds of the linked video you can see that they also swim in a more typical fishy way (using eel-like body oscillation) when they need a burst of speed or a really quick turn. If you’d like to see this form of locomotion in person – you can see it in a pet shop. Knife fishes use the same basic locomotion method – except they use their anal fin rather than the dorsal.
Close up of the head of the King-of-the-Salmon showing the premaxillary (red) and maxillary (green) bones extended, photographed by Randi Jones.
Note also in the video that the fish has a very short face compared to the Hood Channel specimen photographed onshore. As with many fishes, the jaws of the King-of-the-Salmon are protrusible – the premaxillary and maxillary bones swing out to create a tube – the gill chamber dilates, and water rushes into the mouth along with the prey. The same sort of suction pump mechanism is used by a wide variety of fishes – from tiny seahorses to giant groupers. Once the prey item is inside the fish’s mouth, the mouth closes, water is released through the gills and the prey is swallowed. The entire sequence is lightning fast – even in pipefishes and seahorses – blink and you miss it. In some fishes, the process is even audible – you can hear a snapping sound when seahorses slurp up crustaceans (and fishes). You can’t hear the same snapping sound when larger fishes engulf their prey, but it is no less dramatic an effect.
In 2014, a Louvar and a Finescale Triggerfish were found in BC – a double-header of interesting southern fishes in our waters. But wait… it looks like 2017 is also a double-header for cool coastal fish.
This summer of 2017 (and in 2016), Basking Sharks were sighted here in BC. I think every Basking Shark is newsworthy given that they were nearly eliminated here in an ill-conceived plot to protect BC fisheries (see Wallace and Gisborne 2006 for that sad story). This year’s Basking Sharks were found in Caamano Sound in July, and near the Delwood Seamounts in August. Was it one roving shark? Or two? Are there others?
This September however, Luke Halpin was out surveying marine birds from the deck of the CCGS John P. Tully, and spotted something totally different west of Brooks Peninsula. The fish is estimated at 3.5-4 meters in length, and was cruising against the current just below the surface.
We are really fortunate that it was sunny and seas were so calm – because his picture leaves no doubt as to the fish’s identification. The best part about the story is that the fish is still out there. Don’t get me wrong, I’d have loved to have the fish as a specimen for the museum’s collection – but then again, it would require a custom vat – three to four meter fishes don’t fit in jars.
This species is known north to the southern Kuril Islands in the western Pacific, but Luke’s find is the northern-most record for the species in the eastern Pacific and is conclusive evidence of this species as a new addition to our coastal fish fauna. Which species did he find? You’ll have to wait until he publishes his observations in a scientific research paper. Consider this a trailer – a teaser – there’s a big fish out there – it is cool… and I am jealous. I would love to see this fish alive.
100 Meter Dash post
The Doncaster population of the European Wall Lizard probably is 6 years old based on conversations I have had with home owners. In the Google Earth image – the white dots are known locations – the green dots are new locations for 2017.
How do I know these are new? Homeowners specifically said they had no lizards in 2016 – but they certainly do now. That’s the power of local knowledge and citizen science. The green dots along Oak Crest Drive were newly reported in the spring of 2017, with at least three adult lizards now known on the property. The two green dots along Cedar Avenue to the northeast are based on sightings of at least three young lizards – probably lizards that hatched this year and got well-clear of their parent’s territory. Cannibalism is a good emigration motivation.
Based on where lizards were known in 2016, these 2017 records represent range extensions from 20 to 100 meters. Compared to their body size, that’s pretty decent dispersal given that adult lizards only grow to 21 cm (those fortunate enough to have a perfect tail), and in many cases, the dispersing lizards are young-of-the-year at 8 or so centimeters in total length.
If younglings continue to bolt at this rate and make a bee-line south, I will have lizards in my garden in 2 years. More realistically, it will be another 3 years before we see them along our raised beds or in our greenhouse – not that I’m counting.
We now have 21 orca specimens at the Royal BC Museum—the latest to arrive was T-171, a 6.07 meter female Biggs Orca which was found near Prince Rupert, October 19th, 2013. She had pinniped skulls, vibrissae (whiskers) and partially digested bones in her gut but was emaciated. Why was she emaciated?
During the necropsy, researchers discovered that T-171 had mid-cervical to lumbar vertebrae with severe overgrowth of the neural arches and lateral processes (noted as spondylosis in the necropsy) – the overgrowth looks roughly like popcorn or cauliflower – and had the effect of interlocking some vertebrae. This likely explains her emaciated state. Was she able to hunt? Was she supported by her relatives?
The skull of T-171 (ventral (palatal) view [left], right side [center], and dorsal view [right]) awaiting its catalog number and final place in the Royal BC Museum collection.
Comparison of T-171’s vertebra (left) with overgrowth of bone vs. the normal vertebra of another Biggs Orca (12844) (right). The two vertebrae are not from the exact same position along the spine, but the difference between the two is still shocking.
Many of T-171’s vertebral centra are eroded and porous – not like those of a healthy animal (12844).
The overgrowth of the neural arches pinched the spinal chord of T-171; compare to a neural arch of 12844 (right). The vertebral malformation must have limited this animal’s mobility. It is hard not to anthropomorphize and imagine the discomfort due to this deformation.
T-171 originally was prepared for exhibit at the Royal Ontario Museum, but they wanted a clean articulated skeleton for exhibit. In contrast, we were interested in T-171 because of its skeletal malformation. To make a short story long, we came to an agreement with the ROM to transfer T-171 to the Royal BC Museum, and since, the ROM has acquired L95 (Nigel), a 20 year old southern resident who was found near Esperanza Inlet, March 30th, 2016.
Which Orca is next? In most cases we have no clue – it is not like we hunt orca just to add them to the collection. And we don’t usually have a production line of specimens in preparation. New specimens are acquired when a body washes up, and we make a snap-decision to cover the cost of specimen recovery and preparation. However, September 15, 2016, T-12A (Nitinat) was found off Cape Beale and towed to Banfield. I was contacted September 16th to see if the Royal BC Museum was interested (obviously that was a YES), and now his massive skull is being prepared. Once degreased, Nitinat’s skull will be added to the Royal BC Museum collection – sometime in 2018 – and made available for scientific research.
As a kid I collected many things – from reptiles and amphibians to model airplanes to Star Wars cards – and now look where I am. I dress in black and white as a Stormtrooper with the 501st legion and collect black and white delphinids – Killer Whales – for the Royal BC Museum. Life sure takes you to unexpected destinations.
A little while back I was musing over a spot on my Wall Lizard map that shows a large expanse east of Highway 17 between Cordova Bay Road to Mt Newton Cross Road that appears to be Wall-Lizard-free turf. Wall Lizards are crawling everywhere just the other side of the highway on Tanner Ridge. Either no one has reported lizards from this area – and it seems unlikely given how many reports I receive each year, or lizards have not been able to cross HWY 17.
Cedar Hill Road in the Southeast Cedar Hill area also seems to be a decent barrier even though it is not a particularly busy road. Lizards have been in that area for about 6 years(as of 2016) and have crossed Derby Road without a problem – but not Cedar Hill Road. Cedar Hill may be just busy enough to limit the survival of adventurous lizards.
It seems interesting that a lizard as fast as the Wall Lizard could not cross – but then again – why would they? Young ones disperse to avoid cannibalism, but perhaps the noise, vibration and sight of passing vehicles is enough to dissuade all but the most suicidal of lizards.
I recently tripped across an article detailing road crossing behaviour in snakes (Andrews and Gibbons 2005). In their study, smaller snakes seemed to avoid crossing roads, whereas larger snakes have no problem with the concept. I wonder if the same is true for Wall Lizards? Interestingly, all snake species they studied crossed perpendicular to the road’s length – an adaptive behaviour minimizing distance and time on the tarmac. Some species froze in place when a car passed – that is maladaptive – and significantly increased an animal’s exposure to vulcanized rubber.
I have not seen Wall Lizards crossing a street – but would be interesting to see if they too cross perpendicular to the curb, and whether they blast across or dart and pause – unintentionally increasing their risk of catastrophic z-axis reduction.
Andrews, K.M., and Gibbons, J.W. 2005. How to Highways Influence Snake Movement? Behavioural Responses to Roads and Vehicles. Copeia 2005(4): 772-782.
Another lizard arrived in BC last week. We can add Brown Anole (Anolis sagrei) to our list of accidental imports – but this certainly is not the first one to have arrived by accident in BC. Many lizards travel the globe as stow-aways. This one travelled here in its egg along with a Snake Plant (also known as the Mother-in-Law’s Tongue). Sansevieria are popular houseplants – Snake Plants are easy to keep and look neat. My wife bought one for our living room – no lizards in our plant though.
Where was the plant from? Who knows. This plant could have come from anywhere. Brown Anole’s have invaded Florida, and southern parts of Georgia, Louisiana, Mississippi, and Alabama. They also have invaded Hawai’i, southern Texas and southern California along with their relative the Green Anole (Anolis carolinensis). The Green Anole is native to the south-eastern United States, and in their native range, Green Anoles may be forced out of their usual habitat by their exotic relatives. Brown Anoles are native to Cuba and the Bahamas. Even if it got loose, this anole would not survive our winter. It was no real threat to our environment or fauna, but does show that the transport of exotic species is ridiculously easy – an egg in the soil in a plant pot. This time we are fortunate. Only one egg was present. Anoles are light-weight arboreal lizards which lay one egg at a time, and they are not parthenogenetic. Anole eggs develop in alternate ovaries at about a two week interval – if I remember correctly. This ensures the female lizard is not excessively encumbered, and for us it meant that only one egg likely was present in the pot (or any other pot at the home hardware store).
Even if it got loose, this anole would not survive our winter. It was no real threat to our environment or fauna, but does show that the transport of exotic species is ridiculously easy – an egg in the soil in a plant pot. This time we are fortunate. Only one egg was present. Anoles are light-weight arboreal lizards which lay one egg at a time, and they are not parthenogenetic. Anole eggs develop in alternate ovaries at about a two week interval – if I remember correctly. This ensures the female lizard is not excessively encumbered, and for us it meant that only one egg likely was present in the pot (or any other pot at the home hardware store).
Brown Anole eggs are a bit bigger than a Tic-Tac candy, so no wonder they are overlooked – they also are buried a centimeter or so in the soil – so they’d be out of sight. As long as the soil was not disturbed, was warm and moist – but not too wet, and the egg was not rolled, the developing embryo would survive transport.
I wonder where this lizard’s brothers and sisters ended up? They could be anywhere. Since the lizard travelled here in an egg, I vote we name it Mork. Na-Nu Na-Nu.
Just tripped across this fish while sorting out odd records in the RBCM fish database.
999-00114-001 – unidentified fish – Family Triglidae (Searobins, Gurnards)
Well, it turns out to be Prionotus stephanophrys – a Lumptail Searobin – and a new family, genus and species for BC. Three other triglid species (two of them are Prionotus species) are known to stray into Atlantic Canada.
This one was caught in 1998 on La Perouse Bank, it was added to the RBCM collection in 1999, and sat there ever since. No one had taken a second look at this specimen – until today. It was completely new to our system and as such, I had to add the genus and species to our database’s taxonomic tree.
Until now, its northern record was off the mouth of the Columbia River – this new(ly rediscovered) record extends this family north about 260 km in the eastern North Pacific Ocean.
The lower three pectoral rays of this fish are almost like fingers – it probably walks along the bottom like other triglids – the walking mechanism makes me think of face-hugger Alien larvae.
I took these photos of Royal BC Museum lizard specimens with my iPhone 4 through the eyepiece of the old dissecting microscope in my lab. Then sent the photos via two emails to office thanks to WiFi – and to think – this is the “low-tech” way of doing things these days. Low-tech – sending files through the air from a hand held device… I have to laugh how technology has changed since I was a kid with my first pet lizards. The nerd in me can’t help but hear James Earl Jones’ voice – “Several transmissions were beamed to your inbox. I want to know what happened to the scans they sent you.”
In earlier blogs I have mentioned scale differences between BC lizards – so I thought I may as well take close-up shots to clearly show the differences. Under a dissecting microscope (diss-secting, not die-secting), you can easily see the shape of the bead-like back scales of the European Wall Lizard (Podarcis muralis). It’s like a microscopic cobblestone pavement. Each scale is about the diameter of a standard sewing pin.
European Wall Lizard (2112)
The larger back scales of the Northern Alligator Lizard (Elgaria coerulescens) are painfully obvious, and each scale has its own raised keel. The keel gives each scale an angular appearance.
Northern Alligator Lizard (1358)
The Pygmy Short-horned lizard (Phrynosoma douglasii) has a really complex squamation with tiny granular scales interspersed between clusters of larger keeled scales. The larger scales are raised into spires above the general scale-scape (the lizard equivalent of landscape).
Pygmy Short-horned Lizard (323)
Western Skinks (Plestiodon skiltonianus) by contrast are painfully even and smooth – yawn. It’s a good thing they have speed-stripes and a bright blue tail to make them stand out in a crowd.
Western Skink (1964)
Western Fence Lizards (Sceloporus occidentalis) have scales each with a trailing spine – characteristic of all Sceloporus species. Some, like the Crevice Spiny Lizard in the United States have really robust spines on their scales, others like the Sagebrush Lizard have tiny spines. Cordylids in Africa take spiny scales to a whole new level.
Western Fence Lizard (705)
Sorry, I forgot a scale bar in the photos, but the images were fairly close to the same magnification.
Geraldine A. Allen1 & Laurie J. McCormick1 & Johanna R. Jantzen1 & Kendrick L. Marr 2 & Becky N. Brown3
Abstract: Phragmites australis (common reed) is a widespread perennial grass of wetland habitats, with cryptic native and introduced subspecies in North America. We determined the relative abundance of the subspecies and the distributions of plastid DNA haplotypes throughout British Columbia, Canada, at the northwestern distribution limit of common reed in North America. Of 203 specimens assigned to subspecies using molecular markers, we identified only 9 plants as the introduced ssp. australis; all remaining samples were the native ssp. americanus. The two subspecies co-occurred at only one locality. We identified four native haplotypes (one widespread in British Columbia and three others more localized) and two introduced haplotypes. Using plants of known haplotype, we assessed the utility of different morphological traits and trait combinations for distinguishing native and introduced subspecies in this geographic region. No single morphological trait was diagnostic, but principal components analysis and identification indices based on combinations of traits consistently separated the native and introduced subspecies in our sample. Two- or three-trait combinations of ligule length, lemma length and stem anthocyanic coloration gave the best separation. These indices could reduce the need for confirmation of the introduced subspecies using molecular tools, facilitating efforts to monitor and control this invasive plant.
You’d think that sharks and rays would be pretty well known along our coast. Did you know that two Hammerhead Sharks have been found off Vancouver Island? Even a Tiger Shark has strayed north to Alaska. Did it swim along the BC coast, or did it take a more direct route from Hawai’i? We’ll never know. However, in 2016 a new shark was added to our fish fauna – the Pacific Angel Shark (Squatina californica) – based on a clear photograph by Mark Cantwell and his detailed description of the dive location.
We have known since 1931 that Angel Sharks ranged north to Seattle, and there is a single record from Alaska. The specimen label for this 35 cm Alaskan female had been lost (Evermann and Goldsborough 1907) and we cannot pin down its collection location with certainty. Until now, we had no Angel Shark records for British Columbia – but it was only a matter of time.
On the 30th of April, 2016, a single adult Angel Shark was sighted by a diver off Clover Point right here in Victoria. The shark’s gender cannot be determined from the photograph since claspers, if present, are not visible. The Angel Shark was found in about 12 meters of water, about 30 meters off the point. The diver estimated the shark’s length at about 1.1 to 1.2 meters in length. The specimen was not collected, but it would have made a fantastic museum specimen.
King and Surry (2016) published the discovery of this shark in BC in a recent issue of the Canadian Field-Naturalist. While this now is not breaking news – in fact it is a year late – people may still want the primary reference to our latest elasmobranch.
PDFs are available here [as a new paper, King and Surry (2016) is available by subscription to The Canadian Field-Naturalist or by contacting the primary author]:
Belted Kingfishers (Megaceryle alcyon) usually take fishes – why else would they be called kingfishers. They sometimes take crustaceans and frogs, and I’d be shocked if they turn their beaks up at big juicy insects. However, mammal predation is quite a dietary shift. Apparently no one explained the meaning of “fisher” to a kingfisher in the southwestern Yukon.
This female obviously read its species description. Looks like she caught a young goldfish. (From: https://en.wikipedia.org/wiki/Belted_kingfisher#/media/File:Belted_Kingfisher_with_prey.jpg)
A paper came out in a recent issue of the Canadian Field-Naturalist (see Jung 2016) detailing the capture of a Western Water Shrew (Sorex navigator) by a Belted Kingfisher. That would make a decent meal and a real energetic boost for the Kingfisher. Jung (2016) mentioned that Belted Kingfishers have been known to take Eastern Water Shrews (Sorex albibarbis), and he (Jung 2013) also reported on a kingfisher trying to subdue a Spotted Bat (Euderma maculatum).
From: https://upload.wikimedia.org/wikipedia/commons/8/8f/Belted_Kingfisher.jpg
Imagine if kingfishers changed tactics to regularly prey on other small animals? Their ecology could converge on that of butcher birds (shrikes). What’s next? Lizards and snakes?(Yes, shrikes impale their prey on thorns (or barbed wire) to age a bit).
Sure glad kingfishers aren’t the size of a Banshee or Leonopteryx from Avatar, or we’d all be at risk when swimming.
Keep your eyes on the sky. And as for that specific Water Shrew, all you can say is: “Hair today, gone tomorrow.”
PDFs are available here:
Was this an odd title? Actually I think the song went,
“On top of spaghetti… all covered with cheese,
I lost my poor meat ball… When somebody sneezed.
It rolled off the table… and onto the floor.
And then my poor meat ball… rolled out of the door.
Wow that was a dredged from deep cephalic crevices…
Anyway, I got a tip from Purnima Govindarajulu, my herpetological counterpart in the Ministry of Environment that she’d seen a European Wall Lizard on Mount Tolmie here in southern Saanich. Given how fast and far Wall Lizards are spreading, it was only a matter of time before they colonized this rock. This pocket of lizards will form another expanding sub-population – pretty-much midway between the single lizard I saw at the University of Victoria and the lizards near Doncaster School.
This morning (April 27th) was nice and sunny, and I hiked up to the summit after dropping my daughter at daycare. What did I find first? A Northern Alligator Lizard. That made me very happy – I don’t see those everyday and this lizard was more than patient with the iphone-wielding twit who wanted its picture.
Then less than 2 meters away were the Wall Lizards – five of them. A meter or so along the road, another Wall Lizard. Up along the southeast corner of the reservoir – another large male Wall Lizard.
Yep, looks like they have found a solid toe-hold in this region. Cedar Hill X Road may make a decent barrier to northward dispersal (not that Wall Lizards aren’t north of there anyway) – but they will easily spread southeast and southwest into gardens adjacent to the park. Note the small scales and green colour on this Wall Lizard’s back, compared with the larger coppery scales on the Alligator Lizard (above).
Keep your eyes on rock gardens, rock walls, woody debris, and any bedrock with decent cracks for shelter. The photo below shows just how slender the Wall Lizards are – this one with an intact tail is the largest lizard I have caught to date (21.2 cm total length). After checking the RBCM’s herps database, I see that the only months where I haven’t caught Wall Lizards are January and February – too bad that this spring was consistently cold and wet. I have missed my chance to get a full year’s worth of lizards in 2017.
Somnolent Sculpins post
Yesterday I worked with Chris O’Connor from our Learning Department – we took some children on a tidepool tour. The main point was to chat about museum collections and things we record or measure when we are out sampling. We didn’t go crazy catching fishes, only taking 3 Tidepool Sculpins (Oligocottus maculosus) in the end. But we talked about our role as museum researchers, and why we take more than 1 specimen (if possible) to get an account of variation within and between species.
You can see slight differences between these fishes – even an injury – just like the subtle, or not so subtle differences we see in each other.
The three fishes will be added to the Royal BC Museum’s ichthyology collection, but before that, they are fixed in 10% Formaldehyde. Researchers used to drop fishes directly into Formaldehyde – many fishes died horrible deaths. When I accidentally get Formaldehyde in a cut – it stings intensely – I couldn’t imagine being dunked directly into that chemical.
Today we are more humane, and give fishes an overdose of anaesthetic before immersion in Formaldehyde. They are dead before they are fixed, and are preserved with a relaxed posture. The primary anaesthetic I use is 2-Phenoxy-Ethanol, but it is hard to get without ordering from a chemical supply company, and the chemical is a suspected carcinogen. I still have about 500 ml of the stuff – so I will use up what I have. Do I really want to buy more? Maybe not.
Do we have safer options? Yes, Clove Oil is a good anaesthetic if mixed as an emulsion in a small volume of 99% Ethanol. But you have to carry a jug of 99% Ethanol everywhere you go – that may not go over well at a Police check-stop. The up-side to this chemical mix is that you smell spicy at the end of the day if you accidentally spill some on yourself.
People have tried Alka-Seltzer tablets. They fizz and release CO2, which knocks-out fishes – but the process is slow and some fishes (those like catfish that gulp air to survive in low oxygen conditions) are resistant and survive way too long in a stressful condition.
A few months ago I tried using Oragel (20% Benzocaine) on European Wall Lizards – colleagues had found it worked well on amphibians. They put Oragel along the spine of an amphibian and it soaks into the skin; I give lizards an oral dose. It renders bullfrogs and wall lizards unresponsive in 20 seconds to a minute. Oragel seems to be a convenient anaesthetic for these invasive herpetiles.
Yesterday, I told the tidepool group that we’d be performing an experiment – I tried Oragel for the first time on the 3 sculpins we caught. As I hoped – less than 20 seconds and the fishes were out cold. 2-Phenoxy-Ethanol takes about the same time on similar sized fishes.
The beauty of Oragel is that it is readily available, and if you run out, you can stop by the nearest pharmacy. It also is safe – we use it on sore teeth or gums. Perfect – it works fast on specimens and is safe for the researcher.
Perhaps someone needs to do a larger scientific study to see how effective over-the-counter Oragel is on larger fishes. Maybe this is an effective over-the-counter tool for preserving new museum specimens.
A Big Shark in BC post
A specimen with no data is not worth keeping. A specimen with vague data is not worth keeping either. The Royal BC Museum’s ichthyology collection contains a vertebral centrum with cartilaginous remnants of its respective haemal arch and neural arch from a shark that washed up November 5th, 1975 (only a few months after Jaws was released in cinemas). It was cataloged as 976-00052-001 in the fish collection (with a variant of the catalog number listed as a previous number ~ B.C.P.M. #97652). Our electronic database only had a collection date for this centrum (no location, no collector).
Flip to our original paper catalog, and we find that there is indeed a collection location: Ahousaht Village, Flores Island – but this never got translated to our electronic database. The paper catalog states that the shark washed up on a beach – but there was no latitude and longitude provided for the record beyond 49°N, 125°W. If you plot the western-most limit of 125°W, it is nowhere near Flores Island – so the location is questionable. Ahousaht Village’s nearest beach is at about 49°16’N, 126°03’W.
Worse yet, the vertebral centrum indicates that this was a big shark – we don’t have a lot of big sharks here…
Great White Shark (Carcharodon carcharias) reaches 6 meters
Pacific Sleeper Shark (Somniosus pacificus) reaches 5-6 meters
Basking Shark (Cetorhinus maximus) reaches at least 9 meters
The shark centrum in the Royal BC Museum collection is about 7.3 cm in diameter – it spans most of the palm of my hand. This must have come from a decent-sized shark. Was it a small Basking Shark? A large Great White? A large Sleeper Shark? It’s not ‘reptilian’ so we can rule out Cadborosaurus (whew). Hang on, Cadborosaurus’ so-called “type specimen” was a photograph of a digested basking shark – Hmmm…
It is a shame no one bothered to take a skin sample – the scales may have been diagnostic. What about teeth? A sample of teeth – even one tooth – would have been enough to identify this fish. Sadly though, nothing remains other than this centrum and a bit of cartilage. It was fixed in formaldehyde and stored in isopropanol – so I think we can forget sending a chunk to Guelph for DNA barcoding. DNA barcoding wasn’t a thing back in 1975, so tissue samples were not preserved for future analysis.
If no one in Ahousaht has a photo of this shark on the beach, or some teeth stashed away, all I have to say is , “Sorry Charlie, the Royal BC Museum wants specimens with good data.”
A Winter so Harsh post
This winter has been cold here in Victoria – relatively speaking. We have had lots of rain, several rounds of snow – and I even had to shovel my driveway and sidewalk. Actually I have had to shovel several times this winter. The rest of the country is not all that sympathetic to the wintery-woes of its Pacific Islanders.
One odd feature of Victoria is that Anna’s Hummingbirds are present year-round – because people feed them. Without artificial feeding stations, they likely would migrate south in autumn with the Rufus Hummingbird and return each spring. It still strikes me as strange to see a hummingbird in winter – given that I moved here from Winnipeg.
In my neighbour’s yard there is Holly bush that is a regular nesting site for our resident male Anna’s Hummingbird – the spot must be coveted because the prickly leaves are a great deterrent to would-be nest thieves.
This nest from 2005 was near the junction of Government Street and Niagra Street in James Bay – also in a Holly bush.
Our hummingbird – yes we are possessive even though we don’t feed hummingbirds in winter – is a regular visitor to our veggie garden and flowers in summer. It stayed this winter even though it was snowy and cold. Someone nearby must have a hummingbird feeder.
Not all Anna’s Hummingbirds were so lucky this year. Today I received a nest containing two feathered nestlings which were snuggled together in their soft little lichen-cup nest. This is certainly an early nesting attempt – they are known to nest from February to August, but nesting this early in the spring is a big risk.
The fate of the female is a mystery (males don’t raise their young). Did she hit a window? Run short of food and die? Did a free-range domestic cat get her? These two nestlings were in a sheltered spot alongside a house here in Victoria, but without a parent, they didn’t last long. Natural selection can be as cold as this winter.
In 2006 I spent a month at sea on the CCGS W.E. Ricker, collecting hundreds of deep sea fishes during a Tanner Crab Survey. Most fishes were identified the traditional way using anatomical features, but we didn’t have an extensive library on board, so many ‘field’ identifications were wrong. Such is life on the high seas when you are rushed to process samples.
Several snailfishes and of course the poorly known Flabby Whalefishes were only identified to genus. One snailfish with its distinctive pelvic girdle resembling a pair of bat’s wings – was simply labeled as “Batwing.” It was a few years later while sorting out some of the samples, that I tripped across a paper by David Stein (1978) describing our “Batwing” species in detail – Osteodiscus cascadiae. Keep in mind that the last comprehensive book on BC fishes – Pacific Fishes of Canada – was published in 1973… I was 6 years old. Pacific Fishes of Canada needs an update – it is woefully out of date.
This week I have been cataloging the last of the fishes caught on the 2006 Tanner Crab Survey – Screech – I know what you are thinking. A decade has passed since these fishes were caught. I am not a slacker – well, some would argue that – but there are many reasons why I am only now sorting and cataloging the last of the Tanner Crab specimens. Forgive me if progress is slow.
Many of the specimens we collected in 2006 had a small plug of tissue removed for DNA Barcoding. Three specimens (DNA barcode field tags from left to right, G5036, INV792, and 0738-Bo2), from Queen Charlotte Sound and west of the northern end of Vancouver Island were identified as Careproctus canus. If this is correct, they are the first for British Columbia.
The same can be said for specimens (barcode field tags from left to right, R5826 and G5026), both from Queen Charlotte Sound which were identified as Careproctus attenuatus. If correct, they are the first of their kind for BC, and both species C. canus and C. attenuatus, are way-south of their known ranges in the Aleutian Islands. We also caught one other snailfish identified as Paraliparis melanobranchus (15943) – if correct, it is the second specimen for the RBCM.
When I got down to the last few unidentified fishes to catalog in the RBCM database, I found that they had tags from the DNA Barcoding project. Obviously I looked up the molecular identification, but I have to wonder whether a genetic sequence was used to identify these new snailfishes, or whether the DNA barcoding team used our field identifications. We certainly do not carry an exhaustive library at sea, and we do our best to identify fishes with what we have at our finger-tips while the decks are heaving and rolling. Since I don’t trust my own eye regarding snailfishes – these noteworthy records need to be verified – and I think I’ll send them to a snailfish expert that I know just south of the border.
However, two specimens of Gyrinomimus (lovingly known as Flabby Whalefish) were identified as G. grahami (barcode tags, left to right INV0718 and R5828), and both were from west of the northern end of Vancouver Island. They don’t look much better in person. We left these specimens identified to genus because we had no literature for Flabby Whalefishes on board. As a result, I know the species-level identification did not come from me – and had to be based on molecular information. YAY, Gyrinomimus grahami (15942, 15935) is new to BC.
These interesting records alone justify the time taken to collect and send DNA samples to Guelph for the barcoding project. I may not be a gene-jockey, but if the identifications of these fishes are correct, we will rack up another three new species for BC, boost our knowledge of biodiversity, finally have two of our whalefish specimens o-fish-ally identified. Now to compare the newly identified whalefish specimens to the other 10 jar-loads of specimens to see if we have one or more species in our collection.
Thanks all you DNA barcoders – particularly Dirk Steinke who was out with us in 2006 – couldn’t have done this without you.
Abstract
In Canada, there are no native catfish west of the continental divide and until recently, the list of extant exotic catfishes in British Columbia only included introduced Black Bullhead (Ameiurus melas) and Brown Bullhead (Ameiurus nebulosus). We report that a single Yellow Bullhead (Ameiurus natalis) was collected from Silvermere Lake in the Lower Fraser River drainage. This represents the first record of the Yellow Bullhead in western Canada, and its introduction likely was accidental with a shipment of Largemouth Bass (Micropterus salmoides) rather than dispersal from Washington. Warm, eutrophic, weedy habitat in the Fraser Delta provides ample habitat for Yellow Bullheads and other exotic fishes. A Blue-eyed Panaque (Panaque suttonorum), a loricariid catfish found in 1995 in Shawnigan Lake, Vancouver Island, probably represents a single, illegally released aquarium fish, as does a large Silver Pacu (Piaractus cf. P. brachypomus), which was found in Green Lake on Vancouver Island in 2004.
ABSTRACT
Polymerolepis whitei Karatajūtė-Talimaa, 1968 was described based on isolated polyodontode scales recovered from the Ukraine, and originally was thought to be heterostracan (Agnatha). Additional scales with neck canals were described years later, and as a result, P. whitei was reclassified as a bradyodont holocephalan because it had scales similar to those of Listracanthus Newberry & Worthen, 1870. Until now, no articulated body fossils were known, and so the classification of this taxon has remained uncertain and based only on the original author’s opinion. New specimens of P. whitei from the Mackenzie Mountains, Northwest Territories, Canada, show articulated scale patches from the head, with the best specimen showing part of an anal fin, caudal peduncle, and caudal fin. This new material confirms that the original account of scale variation was accurate, but also that P. whitei possesses an anal fin spine, a feature that, until recently, was thought to be a synapomorphy of acanthodian fishes among Palaeozoic fishes. Several primitive chondrichthyans (Obtusacanthus Hanke & Wilson, 2004; Lupopsyroides Hanke & Wilson, 2004; Kathemacanthus Gagnier & Wilson, 1996; Seretolepis Karatajūtė-Talimaa, 1968; Doliodus Traquair, 1893; Antarctilamna Young, 1982, and also problematic taxa such as Gyracanthides Woodward, 1902, and now Polymerolepis Karatajūtė-Talimaa, 1968), are known from articulated remains and show a fin-spine complement like that of acanthodian fishes. They also have placoid scales or polyodontode scales that grew by areal rather than superpositional accretion. These taxa blur the distinction that exists in historic literature between acanthodians and early chondrichthyans.
ABSTRACT
New anatomical details are described for the acanthodian Lupopsyrus pygmaeus Bernacsek & Dineley, 1977, based on newly prepared, nearly complete body fossils from the MOTH locality, Northwest Territories, Canada. New interpretations of previously known structures are provided, while the head, tail, and sensory lines of L. pygmaeus are described for the first time. The pectoral girdle of L. pygmaeus shows no evidence of pinnal and lorical plates as mentioned in the original species description. Instead, the dermal elements of the pectoral region appear to comprise a single pair of prepectoral spines which rest on transversely oriented procoracoids, and large, shallowly inserted, ornamented pectoral fin spines which contact both the procoracoids and scapulocoracoids. The scales of L. pygmaeus lack growth zones and mineralized basal tissue, and superficially resemble scales of thelodonts or monodontode placoid scales of early chondrichthyans, and not the typical scales of acanthodians. However, L. pygmaeus possesses perichondrally-ossified pork-chop shaped scapulocoracoids, a series of hyoidean gill plates, and scale growth that originates near the caudal peduncle; these features suggest a relationship to acanthodians. Prior to this study, both authors conducted separate cladistic analyses which resulted in differing tree positions for L. pygmaeus and its relationships within the Acanthodii. However, both analyses did agree that there is no evidence allying L. pygmaeus to the traditional “climatiid” acanthodians contrary to previous historical classifications.
Abstract
Mid- to Late Palaeozoic sharks and holocephalans display a wide range of armour, with bodies that range from sleek, pelagic forms to slow-swimming, chimaeroids or ray-like bottom dwellers. Despite this Late Palaeozoic diversity, there still is an expectation that early chondrichthyans will be anatomically like later species. Recent discoveries from eastern Canada (Doliodus problematicus), and several heavily spined fishes from the MOTH locality in the Northwest Territories, including Kathemacanthus and Seretolepis, described here, challenge this expectation. These fishes show scale and endoskeletal features thought to be characteristic of chondrichthyans, yet they have paired fin spines, anal fin spines, and in some cases rows of prepectoral and prepelvic spines as would be expected from primitive acanthodians. Kathemacanthus and Seretolepis do not fit neatly within the cur- rent taxonomy, demonstrating that previous distinctions between acanthodians and chondrichthyans, including scale-based criteria, fail to account for the diversity being discovered in the fossil record.
Abstract:
Two new acanthodian taxa are described. The ischnacanthid Xylacanthus kenstewarti is based on large, dentigerous jaws, and Granulacanthus joenelsoni is based on isolated spines. The isolated remains of these species are similar in that they both possess pustulose denticles or tubercles, either on the mesial ridge (X. kenstewarti) or on the fin spines (G. joenelsoni). Jaws of X. kenstewarti are similar in size to those of Xylacanthusminutus, Ischnacanthus kingi, and I. wickhami and smaller than those of X. grandis. The jaws of X. kenstewarti are most similar to those of X. minutus, but are distinguished from this and other ischnacanthid species by a tapering patch of pustulose denticles that is widest midway along the jaw, mesial denticles that are simple blisterlike structures, the monocuspid, striated primary teeth that are subcircular in cross section, and a posterodorsal process that is enlarged. The spines of G. joenelsoni have distinctive tubercular ornamentation. Tubercles, or nodular ornaments on fin spines, are characteristic of primitive acanthodians, but the slender shape of the spines, the low number of spine ribs, and the fine striations posterior to the main ribs of each spine suggest that G. joenelsoni is a relatively advanced acanthodian. Xylacanthus kenstewarti and G. joenelsoni are from the Silurian (Wenlock or Ludlow) of the southern Mackenzie Mountains. Xylacanthus kenstewarti represents the earliest representative of the genus, the earliest unequivocal remains of a gnathostome from the Mackenzie Mountains, and extends the known geographical range of the genus from the Mackenzie Mountains east to Spitsbergen.
Abstract
An acanthodian, Tetanopsyrus lindoei gen. et sp. nov., is described. All specimens are from Lochkovian of northwestern Canada. The body is covered with unornamented, flat scales, with two finely noded dorsal spines, finely noded anal, pelvic and pectoral spines, a high scapulocoracoid, and toothless jawbones with large, flat, crushing surfaces. Tetanopsyrus lacks pectoral dermal plates and intermediate pre-pelvic fin spines. Tetanopsyrus is classified in the new family Tetanopsyridae, and possible relationships of the family to diplacanthids are discussed.
Abstract
Specimens of two new fish species were collected from the Lower Devonian ichthyofauna of the Mackenzie Mountains, Northwest Territories, Canada. These two species are interesting in that they have monodontode scales, lack teeth, and have an unossified axial, visceral, and appendicular endoskeleton. These characteristics have been suggested to be primitive for jawed fishes. However, the new taxa have combinations of median and paired fin spines which are similar to those of acanthodian fishes. The new taxa show no obvious characteristics to suggest relationship to any particular group of acanthodians, and for the moment, we will not try to determine their relationships, but to use them as outgroups in an analyses of relationships within the class Acanthodii. Our cladistic analysis results suggest that climatiiform fishes are basal relative to acanthodiform and ischnacanthiform taxa. However, in contrast to previously published analyses, the order Climatiiformes appears paraphyletic relative to the other two acanthodian orders. Lupopsyrus pygmaeus is placed as the basal-most acanthodian species, Brochoadmones milesi, Euthacanthus macnicoli, and diplacanthids are relatively derived “climatiiform” fishes, and the heavily armored condition in Climatius reticulatus and Brachyacanthus scutiger appears as a uniquely derived state and not primitive for all acanthodians. In addition, Cassidiceps vermiculatus and Paucicanthus vanelsti seem to be related to acanthodiform fishes based on fin spine structures. Cassidiceps vermiculatus originally was placed with climatiiform fishes in the original description. Given our character coding, we identified several primitive characteristics which were retained in relatively derived acanthodian taxa.
Abstract
A mesacanthid acanthodian, Promesacanthus eppleri n. gen., n. sp., is described based on specimens collected from the Lower Devonian (Lochkovian) Manon-the-Hill locality of the Mackenzie Mountains, Northwest Territories, Canada. The head and body resemble that of other mesacanthids, but unlike all other acanthodiforms, this new taxon has a small prepectoral spine anterior to the pectoral fin spine. This new mesacanthid also possesses ornamented, blade-like hyoidean gill covers, enlarged lobate head scales, fin spines with ribs and fine striations, a scapulocoracoid with a triangular coracoid portion and a dorsal blade which is elliptical in cross section, procoracoids that articulate with a rounded fossa on the anteromedial face of the scapulocoracoids, and jaws which articulate at a simple, single joint. Mesacanthids are thought to be basal among acanthodiforms and are grouped based on a phenetic argument and their shared retention of features which likely are primitive for acanthodiforms (most notably, enlarged head scales, blade-like hyoidean gill covers, and a single pair of prepelvic spines). Based on overall similarity, P. eppleri n. gen., n. sp. appears most similar to Mesacanthus mitchelli, but the relationships of P. eppleri n. gen., n. sp. within the Mesacanthidae have yet to be determined with a cladistic analysis.
Abstract
The acanthodian Paucicanthus vanelsti gen. et sp. nov. is described from six body fossils from Lower Devonian (Lochkovian) rocks of the southern Mackenzie Mountains, Northwest Territories, Canada. This new species is unique among acanthodians in that it lacks both pectoral and pelvic fin-spines. In the absence of fin-spines, the leading edges of the pectoral and pelvic fins are reinforced by enlarged scales. The anatomy of the acanthodiform Traquairichthys pygmaeus is similar to P. vanelsti in that both lack pelvic fin-spines, although T. pygmaeus also lacks pelvic fins. Similarly, the acanthodian Yealepis douglasi lacks both paired and median fin-spines, and its anatomy resembles that of P. vanelsti based only on the loss of paired fin-spines. The lack of paired and (or) median fin-spines in these three taxa contrasts with the widely held view that acanthodian fins all were preceded by spines. The anatomy of P. vanelsti also is similar to that of the acanthodian Brochoadmones milesi in that both have a completely unossified endoskeleton, slightly elevated pectoral fins, and deep, compressed bodies. The median fin-spines of P. vanelsti have an anterior leading edge rib followed by a field of fine striations. This striated ornamentation coupled with few leading edge ribs also is seen on fin-spines of Cassidiceps vermiculatus and primitive acanthodiform acanthodians (e.g., Mesacanthus and Lodeacanthus species). I tentatively suggest that this fin-spine ornament indicates relationship between P. vanelsti, acanthodiform acanthodians, and C. vermiculatus. However, a cladistic analysis is required to test whether or not the characteristics such as fin-spine loss, unossified endoskeleton, elevated pectoral fins, deep compressed bodies, and (or) median fin-spine ornamentation are synapomorphies within the Acanthodii or evolved convergently within the class.
Abstract
New anatomical details are described for the acanthodian Brochoadmones milesi based on nearly complete body fossils from Lochkovian rocks at MOTH, Mackenzie Mountains, Northwest Territories, Canada. The body and caudal peduncle are deep, and a prominent nuchal hump is present before the dorsal fin origin. The caudal fin is correspondingly deep and ventrally, the caudal fin lies close to and is partly joined to the slender anal fin. A delicate pectoral fin trails the flattened pectoral-fin spine where previously known specimens showed only a fin spine resembling a bivalve shell. Seen for the first time in any vertebrate, each of the six pairs of prepelvic spines supports a small, scale-covered finlet. Both prepelvic spines and scalecovered finlets increase in size posteriorly. The series of paired prepelvic finlets originates ventral to the branchial chamber and anteroventral to the pectoral fin, and extends posteriorly as far as the pelvic fins. The scales of the body and fins are thin and flat, without obvious evidence of ossified basal tissue or entry point for vascular tissue. The main lateral-line canal passes dorsal to the branchial chamber and terminates at the trailing edge of the caudal fin web. Lateral-line scales are thicker than body scales and show concentric growth zones. Scales from the dorsal midline of the caudal fin are also thicker, showing few superpositional growth zones in the mesodentine of the crown together with what appears to be cellular basal tissue. The structure and position of the pectoral spine and fin, the extremely thin body scales, the slender anal fin, and the prepelvic finlets are all unique and appear to be autapomorphic features compared to those of other acanthodians. Brochoadmones milesi is derived relative to other fishes traditionally classified in the Climatiiformes. Kathemacanthus rosulentus is removed from the Brochoadmonoidei, leaving only B. milesi in a monotypic suborder.
Abstract
An undescribed genus and species of pachyosteomorph arthrodire, Squamatognathus steeprockensis gen. et sp. nov., from the Middle Devonian (Eifelian) Elm Point Formation in Manitoba is described. It was found in the LaFarge Quarry at Steep Rock, Manitoba, and is represented by the anterior portion of a large, right inferognathal with a large terminal cusp, similar to inferognathals of the family Dinichthyidae. It has unique sculpture on the lingual surface not reported from any other dinichthyid arthrodire.